DO NOW Write question answer in notebook How

DO NOW • (Write question & answer in notebook) • How many electrons can go into: • The first energy level? • The 2 nd energy level? • The 3 rd energy level? th • The 4 energy level?

The Atom Protons and electrons are attracted to each other because of opposite charges Define electrostatics the branch of physics dealing with electric phenomena not associated with electricity in motion. Electrostatic Basics:

Electrostatic Basics: - - repel - + attract + + repel

The Wave-like Electron The electron propagates through space as an energy wave. To understand the atom, one must understand the behavior of electromagnetic waves. Louis de. Broglie
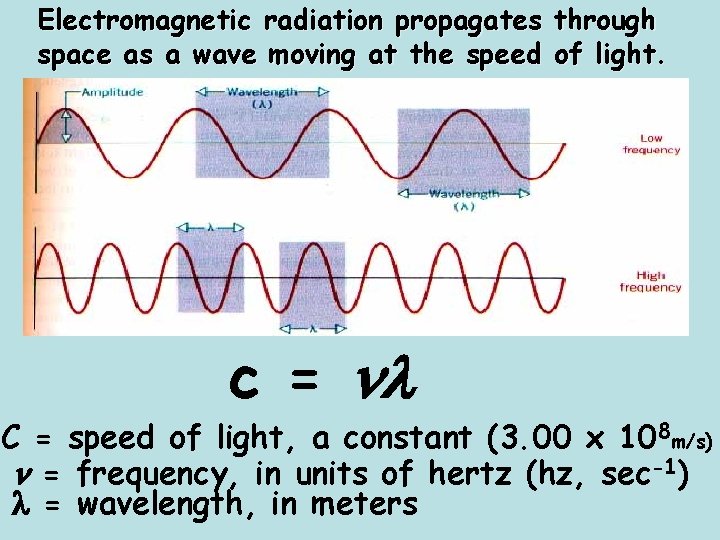
Electromagnetic radiation propagates through space as a wave moving at the speed of light. c = C = speed of light, a constant (3. 00 x 108 m/s) = frequency, in units of hertz (hz, sec-1) = wavelength, in meters
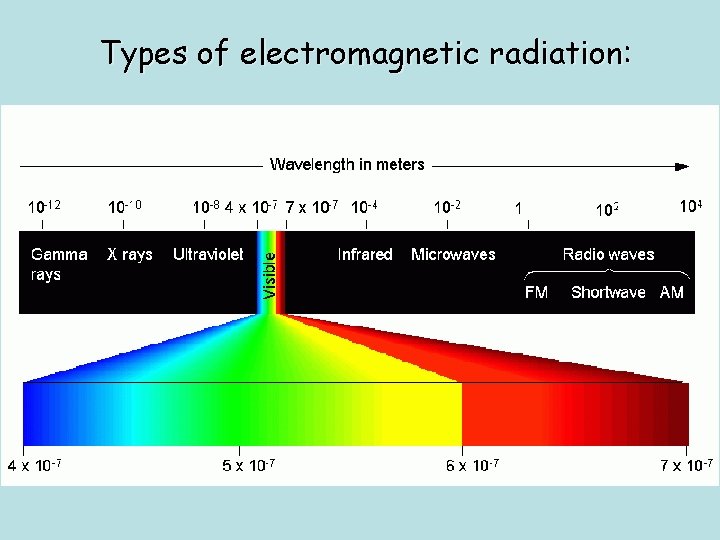
Types of electromagnetic radiation:
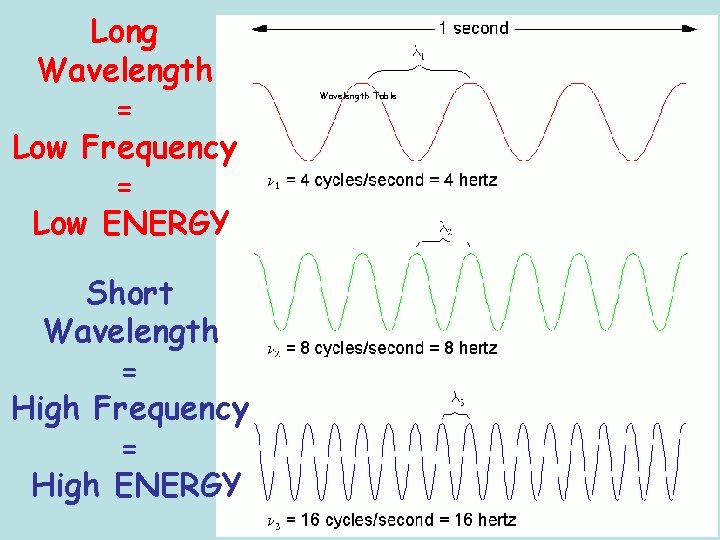
Long Wavelength = Low Frequency = Low ENERGY Short Wavelength = High Frequency = High ENERGY Wavelength Table
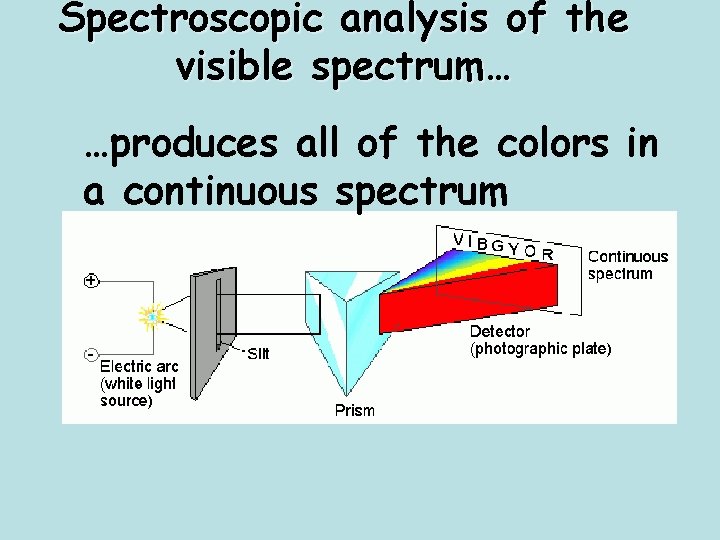
Spectroscopic analysis of the visible spectrum… …produces all of the colors in a continuous spectrum
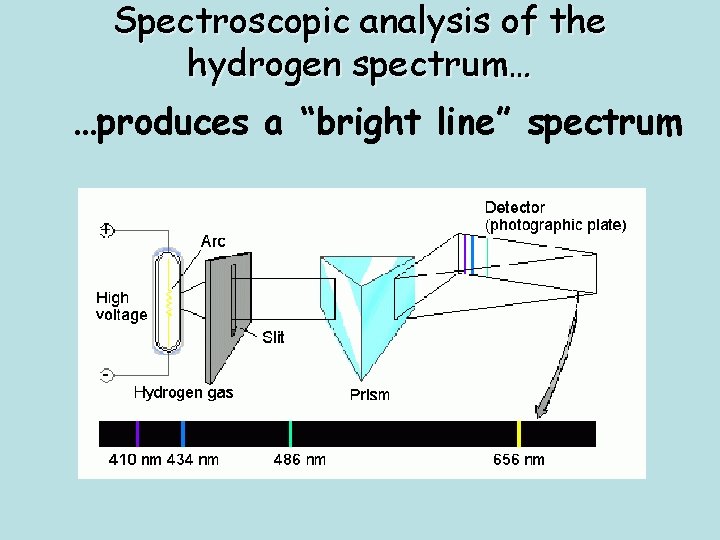
Spectroscopic analysis of the hydrogen spectrum… …produces a “bright line” spectrum
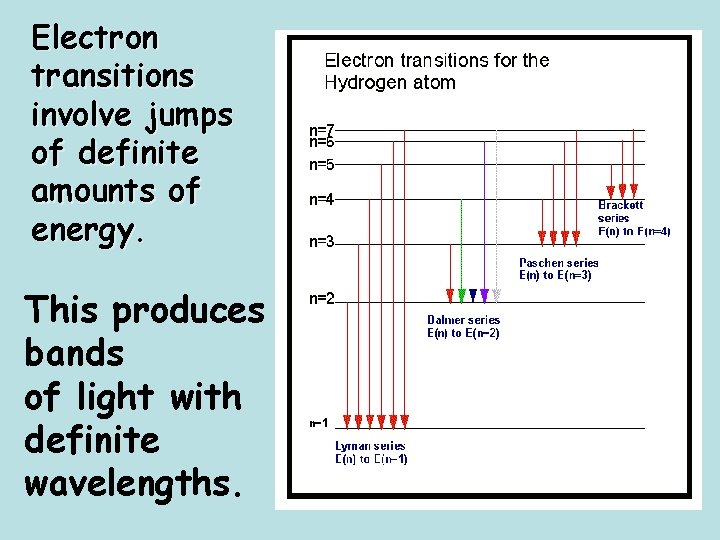
Electron transitions involve jumps of definite amounts of energy. This produces bands of light with definite wavelengths.

TODAY YOU WILL LEARN • HOW TO BETTER VISUALIZE WHERE ELECTRONS ARE • HOW TO WRITE ELECTRON CONFIGURATIONS

Bohr Model of Atom • Planetary Model of atom – put electrons into fixed paths around nucleus called orbits • Assigned atoms to fixed energy levels • Moving from one energy level to another = requires a fixed quantum of energy • Quantum Leap – jump from one step to another
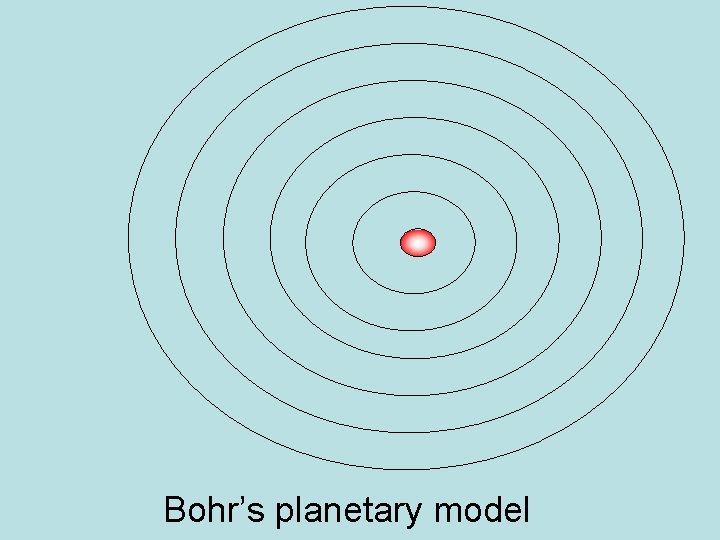
Bohr’s planetary model

Schrodinger Wave Equation Erwin Schrodinger This is an equation for the probability of a single electron being found along a single axis (this one shows x axis)

Quantum Mechanical Model of the Atom Mathematical laws can identify the regions outside of the nucleus where electrons are most likely to be found. These laws are beyond the scope of this class…
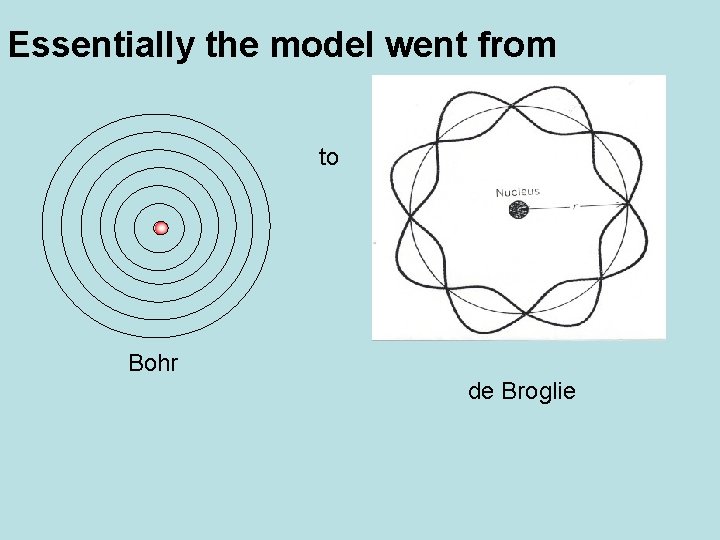
Essentially the model went from to Bohr de Broglie
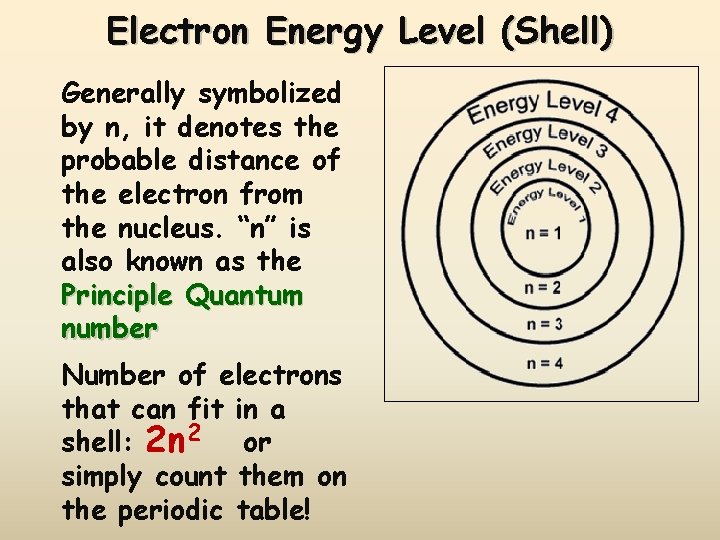
Electron Energy Level (Shell) Generally symbolized by n, it denotes the probable distance of the electron from the nucleus. “n” is also known as the Principle Quantum number Number of electrons that can fit in a shell: 2 n 2 or simply count them on the periodic table!

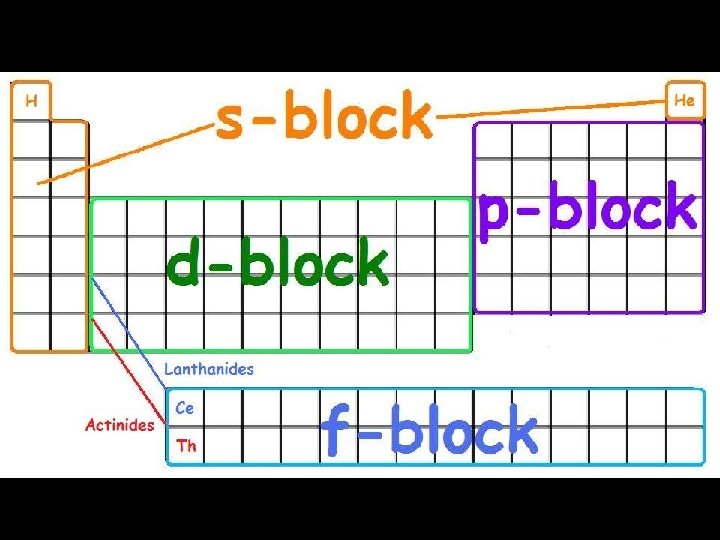
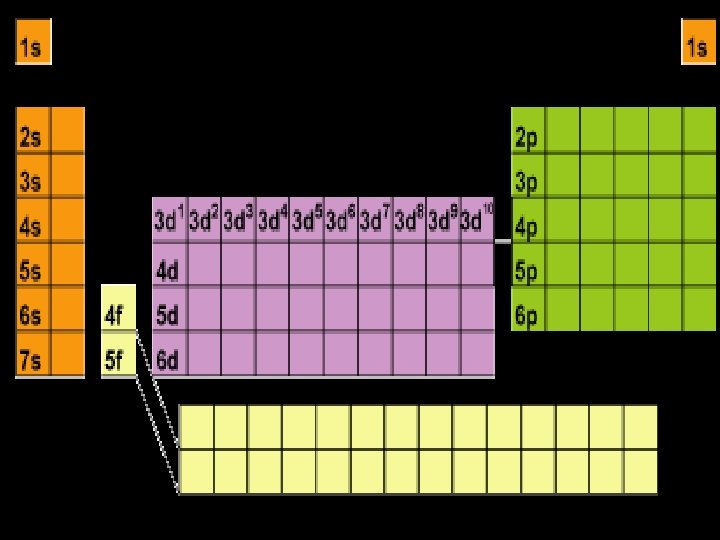
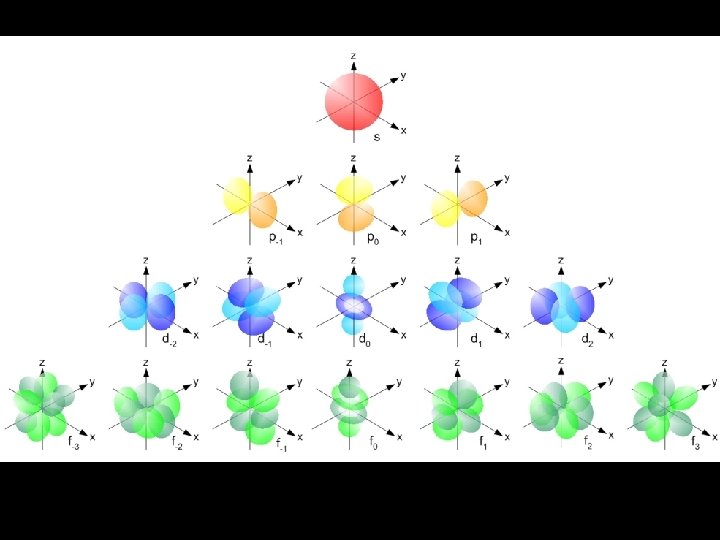
Energy Sublevels (subshells) A sublevel is a region within an energy level where there is a probability of finding an electron. Orbital shapes are defined as the surface that contains 90% of the total electron probability. Make map now.

Write the electron configurations (no shortcuts!) • • • Lithium (Li) - 3 electrons 2 1 1 s 2 s Oxygen (O) - 8 electrons 2 2 4 1 s 2 s 2 p Neon (Ne) - 10 electrons 2 2 6 1 s 2 s 2 p

Write the electron configurations (no shortcuts!) • • • Magnesium (Mg) – 12 electrons 2 2 6 2 1 s 2 s 2 p 3 s Chlorine (Cl) – 17 electrons 2 2 6 2 5 1 s 2 s 2 p 3 s 3 p Scandium (Sc) – 21 electrons 2 2 6 2 1 1 s 2 s 2 p 3 s 3 p 4 s 3 d

Write the electron configurations (no shortcuts!) • Titanium(Ti) - 22 electrons • Vanadium(V) - 23 electrons

Write the electron configurations (no shortcuts!) • Titanium(Ti) - 22 electrons 2 2 6 2 2 • 1 s 2 s 2 p 3 s 3 p 4 s 3 d • Vanadium(V) - 23 electrons • 1 s 22 p 63 s 23 p 64 s 23 d 3
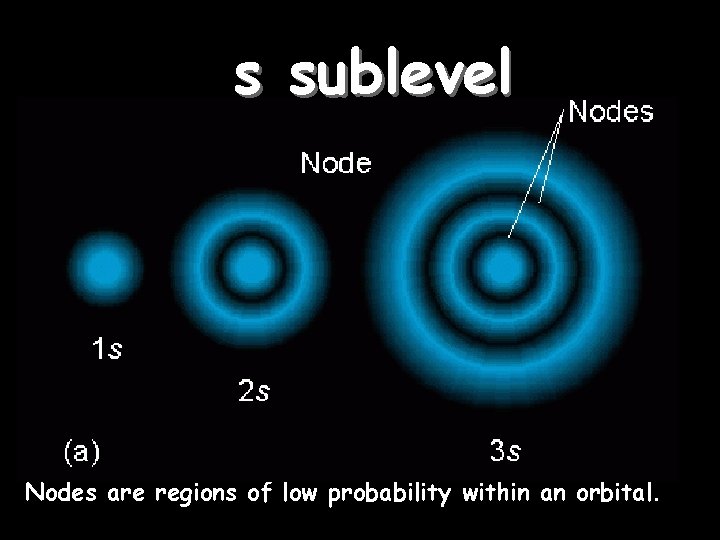
s sublevel Orbitals of the same shape (s, for instance) grow larger as n increases… Nodes are regions of low probability within an orbital.
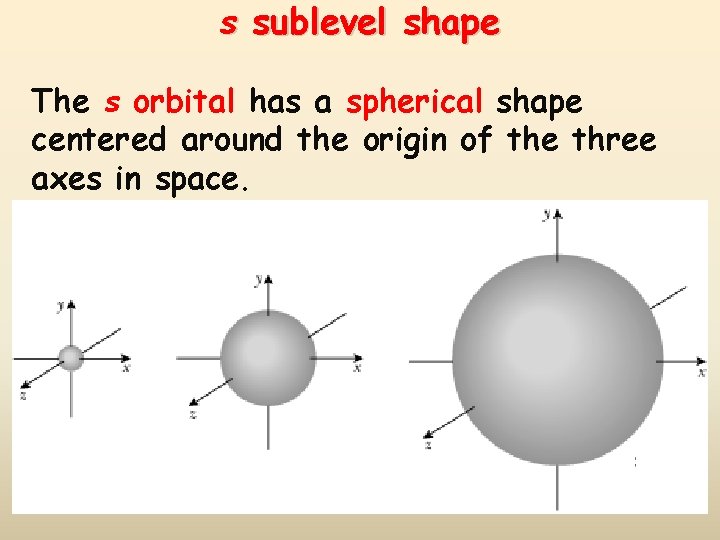
s sublevel shape The s orbital has a spherical shape centered around the origin of the three axes in space.
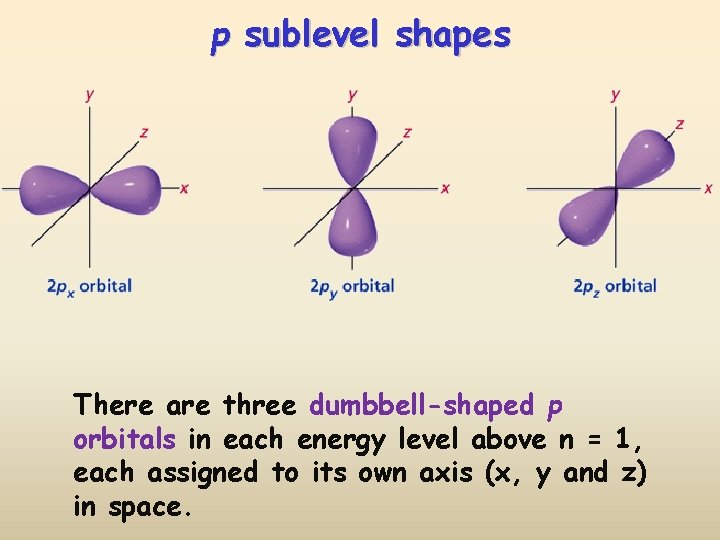
p sublevel shapes There are three dumbbell-shaped p orbitals in each energy level above n = 1, each assigned to its own axis (x, y and z) in space.

d sublevel shapes Things get a bit more complicated with the five d orbitals that are found in the d sublevels beginning with n = 3. To remember the shapes, think of “double dumbbells” …and a “dumbbell with a donut”!
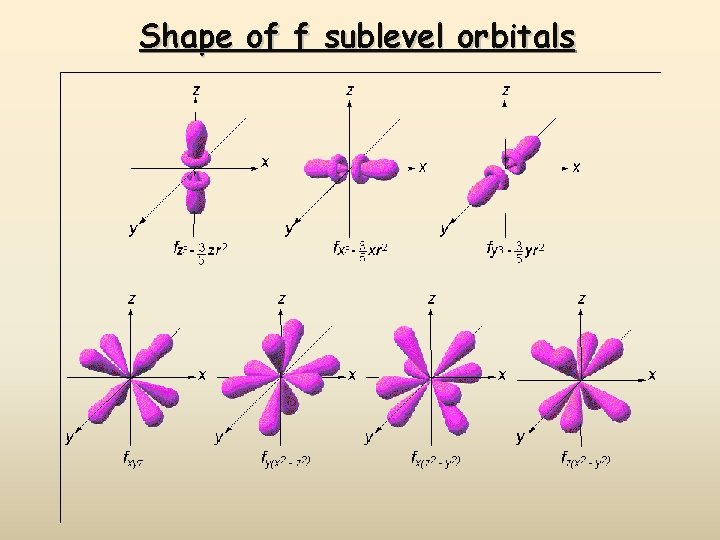
Shape of f sublevel orbitals
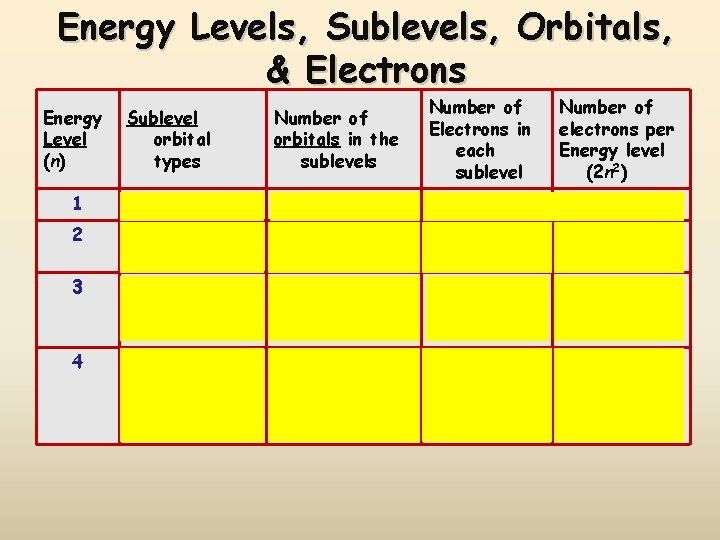
Energy Levels, Sublevels, Orbitals, & Electrons Energy Level (n) Sublevel orbital types Number of orbitals in the sublevels Number of Electrons in each sublevel Number of electrons per Energy level (2 n 2) 1 s 1 2 2 2 s p 1 3 2 6 8 3 s p d 1 3 5 2 6 10 18 4 s p d f 1 3 5 7 2 6 10 14 32

3 Rules for electron configurations: 1) Aufbau Principle – electrons fill lowest energy level orbitals first (lower energy = more stable!) • The word aufbau is German for “building up” • *Note: Atoms always do things to become the most stable!

2) Hund’s Rule – • Electrons will space out evenly. • Want highest number of unpaired electrons with the same spin. • Electrons want the most space between electrons with same energy! • Why? ? ?

Pauli Exclusion Principle Each orbital can hold a MAXIMUM of two electrons! Wolfgang Pauli

Pauli Exclusion Principle Two electrons occupying the same orbital must have opposite spins Wolfgang Pauli

Electron Spin Electron spin describes the behavior (direction of spin) of an electron within a magnetic field. Possibilities for electron spin:

Summarize electron the 3 rules for configuration
![Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1 Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1](http://slidetodoc.com/presentation_image_h2/6556aff5a34cee27b631f3ef78163587/image-38.jpg)
Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1 ____ 1 s Beryllium ____ 2 p ____ 2 s ____ 2 p ____ [He]2 s 2 p 2 ____ 2 s ____ 2 p ____ 1 s 22 p 3 [He]2 s 2 p 3 ____ 2 s ____ 2 p ____ 1 s 22 p 4 [He]2 s 2 p 4 ____ 2 s ____ 2 p ____ 1 s 22 p 5 [He]2 s 2 p 5 ____ 1 s Neon ____ 2 s 1 s 22 p 2 ____ 1 s Fluorine ____ [He]2 s 2 p 1 ____ 1 s Oxygen ____ 2 p 1 s 22 p 1 ____ 1 s Nitrogen ____ [He]2 s 2 ____ 1 s Carbon ____ 2 s 1 s 22 s 2 ____ 1 s Boron Noble gas notation ____ 2 s ____ 2 p ____ 1 s 22 p 6 [He]2 s 2 p 6 ____ 1 s ____ 2 s ____ 2 p ____
- Slides: 38