Do Now On the back of yesterdays lab

Do Now �On the back of yesterday’s lab sheet, state whether you think this is an open or closed system, and why.

Open vs. Closed Systems �Open system – Things (energy, heat, particles, matter) can come in and out �Closed system – Nothing comes in or out

Warm-up �What do the following all have in common?

What doesn’t belong? �Dozen �Couple �Gross �Baker’s Dozen �Ten

The Mole

Definition �Mole- unit standing for 6. 02 x 1023 particles 602, 000, 000, 000 particles Avogadro’s Number

Very Similar to a Dozen! �A dozen eggs would make a HUGE omelet! �A mole of eggs would make an omelet big enough to cover the Earth thirty times!

Why such a BIG unit? �Atoms and Molecules are TINY! �A drop of water contains 10 trillion water molecules!

Moles and Particles � 6. 02 x 1023 makes a chemist’s life a lot easier �It’s impossible to keep track of one atom so scientist discovered this number that gives us the molar mass of an atom �Molar mass is the mass of 6. 02 x 1023 particles of a given substance Mass is given in grams

We don’t always have exactly 1 mole, though. When we have different amounts of moles, we can convert between atoms and moles
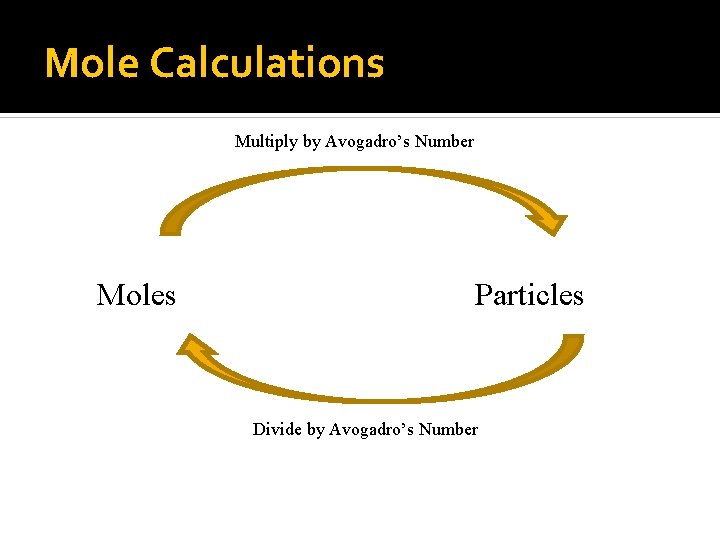
Mole Calculations Multiply by Avogadro’s Number Moles Particles Divide by Avogadro’s Number

Example 1 �A lab experiment calls for 2. 00 moles of Calcium. How many particles of Calcium are required? �How can we solve this? Dimensional Analysis! ▪ ID what we’re given and what the question’s asking for and set up the T-Chart 23 2 moles Calcium 6. 02 x 10 particles 1 mole Calcium

Example 2 �You have 12. 04 x 1023 atoms of sulfur. many moles of sulfur do you have? How

You Try It! �How many moles are 1. 20 x 1025 atoms of phosphorous?

You Try It! �How many moles of magnesium is 3. 01 x 1022 atoms of magnesium?

You Try It! �How many atoms are in 0. 750 moles of zinc?

Exit Slip �You have 1. 0 x 1026 atoms of sodium. many moles do you have? How

Converting from grams mole Mole grams Calculating molecular mass Molecular Mass

Molar Mass Luckily 6. 02 x 1023 particles, or a mole, adds up to the same number as the avg. atomic mass-but in grams So a mole of magnesium has a mass of 24. 305 grams!

Molar mass: mass of 1 mole of in grams 1 mole C atoms = ___________ 1 mole of C atoms has a mass of_____ g. Molar mass of O = ______ Molar mass of Li = ______ For any element, the __________________________________ 3. 2

What is the mass of one mole of: S C Hg Cu Fe 3. 2
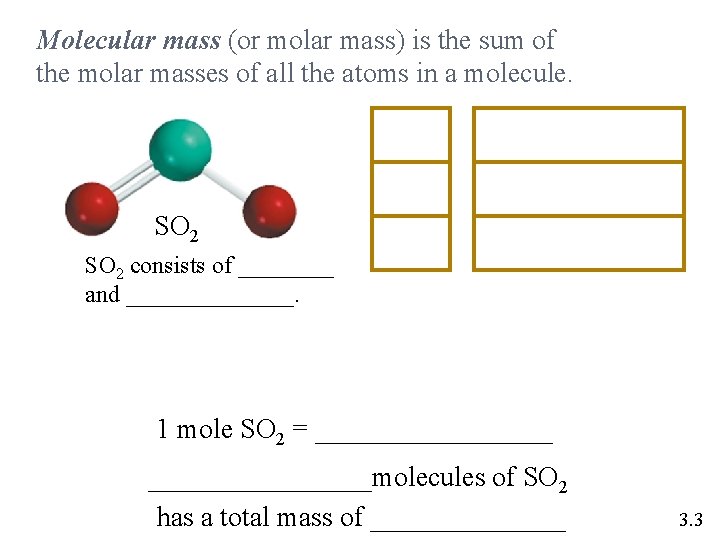
Molecular mass (or molar mass) is the sum of the molar masses of all the atoms in a molecule. SO 2 1 S 32. 06 g/mol SO 2 64. 06 g/mol SO 2 consists of ____ and _______. 1 mole SO 2 = _________molecules of SO 2 has a total mass of _______ 3. 3

When I bought my fly kicks… �If I had 20 Crowns, and the cost of one pair of kicks is 4 crowns for 1 pair, how many pairs could I have? What’s my conversion factor? What unit am I given? What unit is the question asking for? 20 Crowns 1 pair 4 Crowns Now. . . (20/4) pairs 5 pairs

�If I had 20 grams of helium, and one mole of helium is 4 grams, how many moles of helium could I have? What’s my conversion factor? What unit am I given? What unit is the question asking for? 20 grams He 1 mole He 4 grams He (20/4) moles He 5 moles He

Grams Mole Conversion 1 mole of Zinc has a mass of 65. 39 g. Conversion factor: OR How many moles of Zn are in 85 grams?
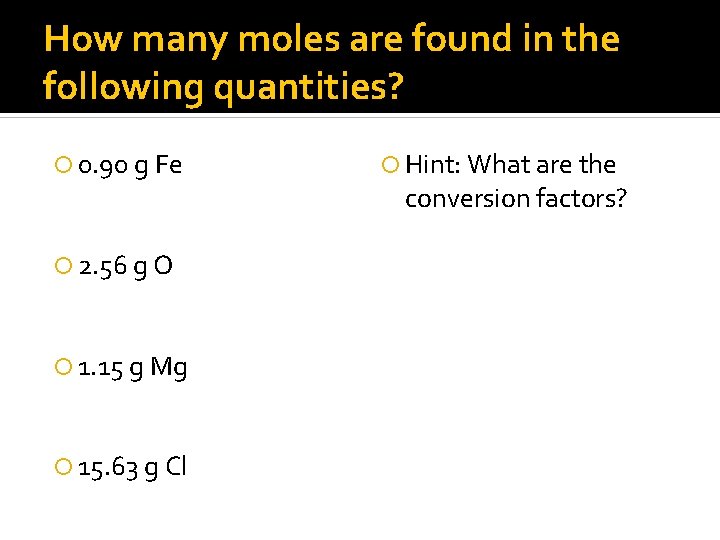
How many moles are found in the following quantities? 0. 90 g Fe 2. 56 g O 1. 15 g Mg 15. 63 g Cl Hint: What are the conversion factors?

Mole Gram Conversion 1 mole of Zinc has a mass of 65. 39 g. Conversion factor: 1 mole Zn 65. 39 g Zn OR 65. 39 g Zn 1 mole Zn If I had 2 moles of Zn, how many grams of Zn do I have? 2 moles Zn 65. 39 g Zn 1 mole Zn 130. 78 g Zn

Find the mass in grams of the following quantities: 1. 35 mol Fe 24. 5 mol O 0. 876 mol Ca 1. 25 mol H Hint: What are the conversion factors?

How many grams are found in 2. 3 moles of C 3 H 8? How many molecules? 2. 3 moles C 3 H 8 44 g C 3 H 8 2. 3 x 44 g C 3 H 8 101. 2 g C 3 H 8 1 mole C 3 H 8 2. 3 moles C 3 H 8 6. 02 x 1023 molecules C 3 H 8 1 mole C 3 H 8 2. 3 x (6. 02 x 1023 molecules C 3 H 8) 13. 2 x 1023 molecules C 3 H 8
- Slides: 29