Do Now Complete the table Count number of
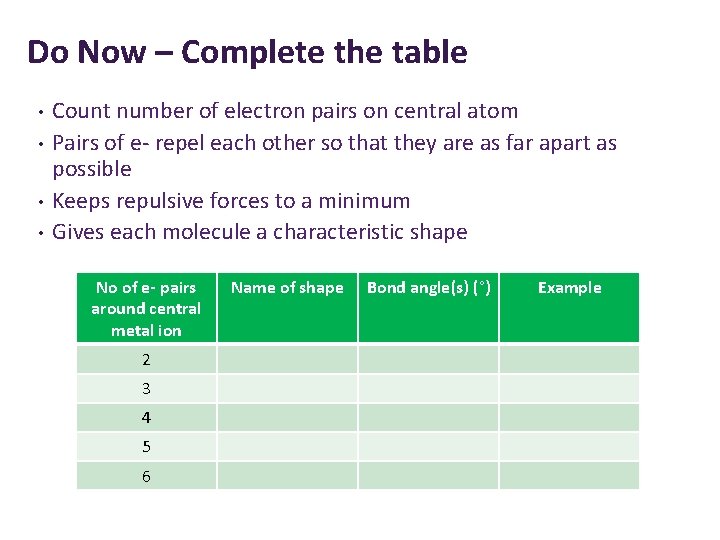
Do Now – Complete the table • • Count number of electron pairs on central atom Pairs of e- repel each other so that they are as far apart as possible Keeps repulsive forces to a minimum Gives each molecule a characteristic shape No of e- pairs around central metal ion 2 3 4 5 6 Name of shape Bond angle(s) (°) Example
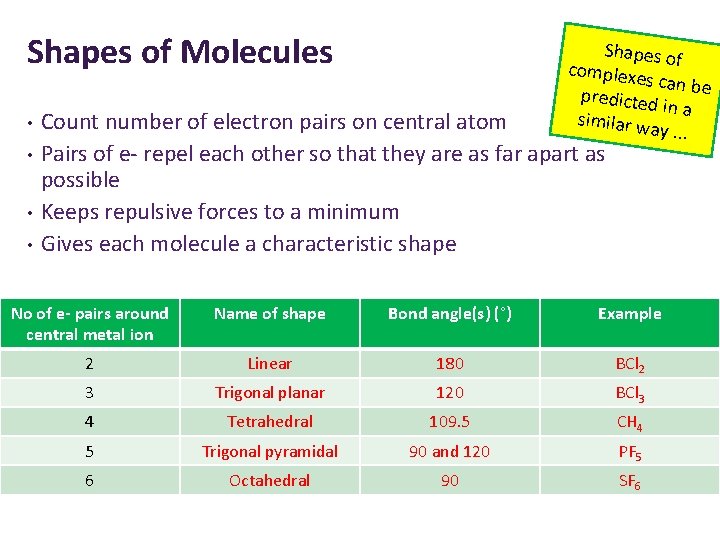
Shapes of Molecules • • Shapes o complexe f s can be predicted in a similar w ay. . . Count number of electron pairs on central atom Pairs of e- repel each other so that they are as far apart as possible Keeps repulsive forces to a minimum Gives each molecule a characteristic shape No of e- pairs around central metal ion Name of shape Bond angle(s) (°) Example 2 Linear 180 BCl 2 3 Trigonal planar 120 BCl 3 4 Tetrahedral 109. 5 CH 4 5 Trigonal pyramidal 90 and 120 PF 5 6 Octahedral 90 SF 6

Cheat method. . . • In general for monodentate ligands. . . • 2 ligands = linear (most silver complexes) 4 ligands = tetrahedral (large charged ligands (Cl-) (exception: some are square planar [cis-platin] – see later) • • 6 ligands = octahedral (small uncharged ligands, H 2 O, NH 3)
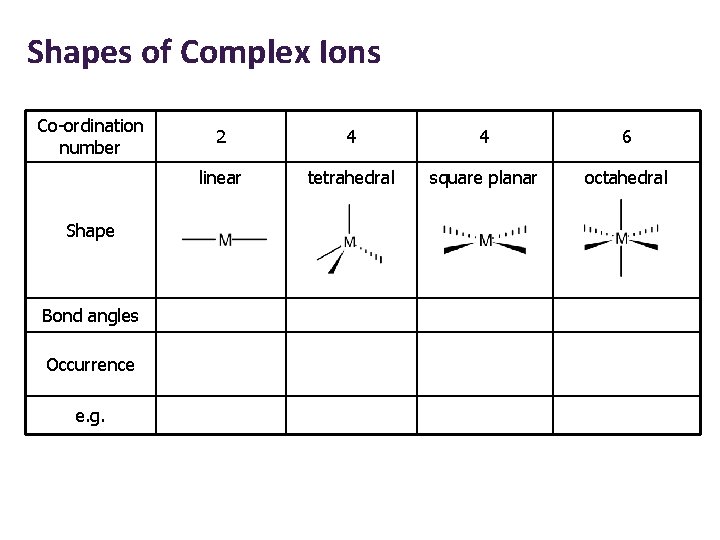
Shapes of Complex Ions Co-ordination number Shape Bond angles Occurrence e. g. 2 4 4 6 linear tetrahedral square planar octahedral
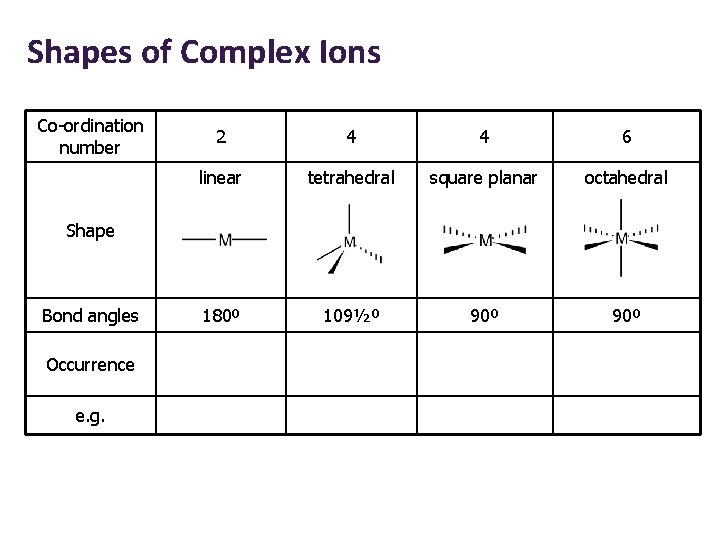
Shapes of Complex Ions Co-ordination number 2 4 4 6 linear tetrahedral square planar octahedral 180º 109½º 90º Shape Bond angles Occurrence e. g.
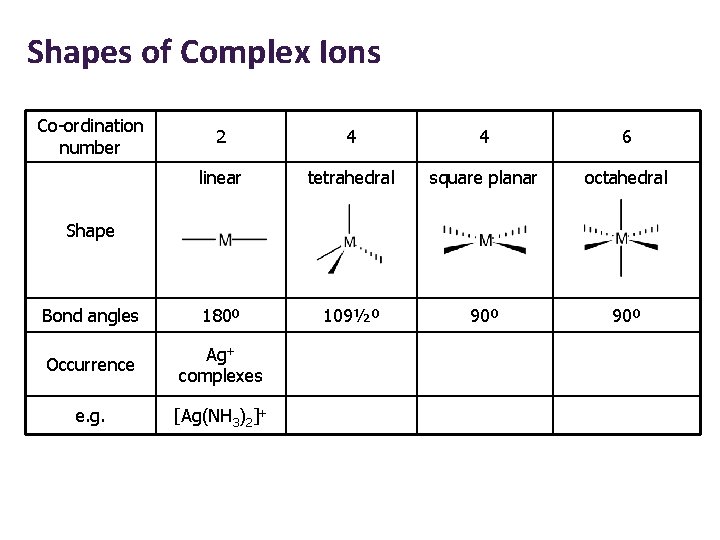
Shapes of Complex Ions Co-ordination number 2 4 4 6 linear tetrahedral square planar octahedral Bond angles 180º 109½º 90º Occurrence Ag+ complexes e. g. [Ag(NH 3)2]+ Shape
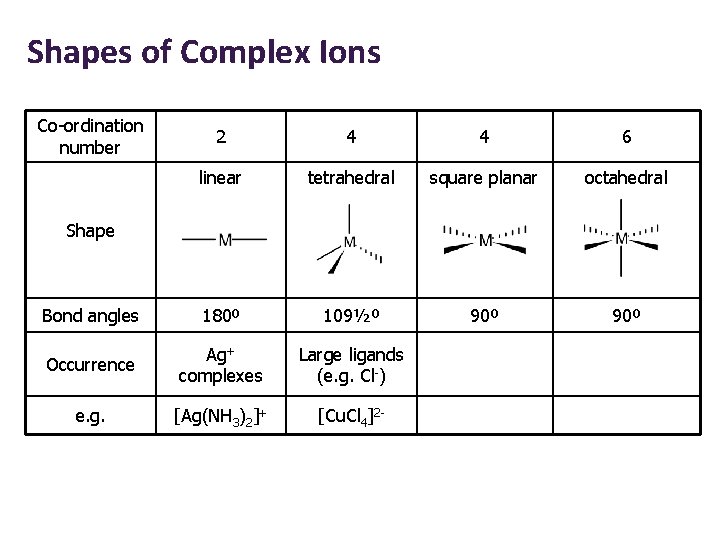
Shapes of Complex Ions Co-ordination number 2 4 4 6 linear tetrahedral square planar octahedral Bond angles 180º 109½º 90º Occurrence Ag+ complexes Large ligands (e. g. Cl-) e. g. [Ag(NH 3)2]+ [Cu. Cl 4]2 - Shape
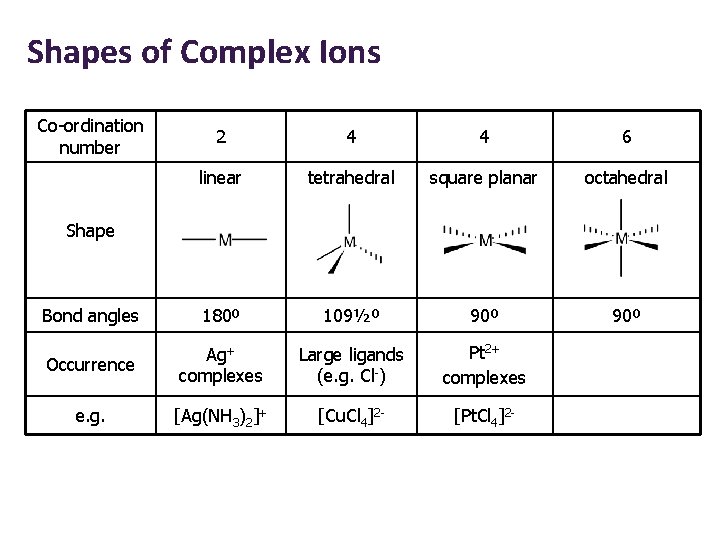
Shapes of Complex Ions Co-ordination number 2 4 4 6 linear tetrahedral square planar octahedral Bond angles 180º 109½º 90º Occurrence Ag+ complexes Large ligands (e. g. Cl-) Pt 2+ complexes e. g. [Ag(NH 3)2]+ [Cu. Cl 4]2 - [Pt. Cl 4]2 - Shape
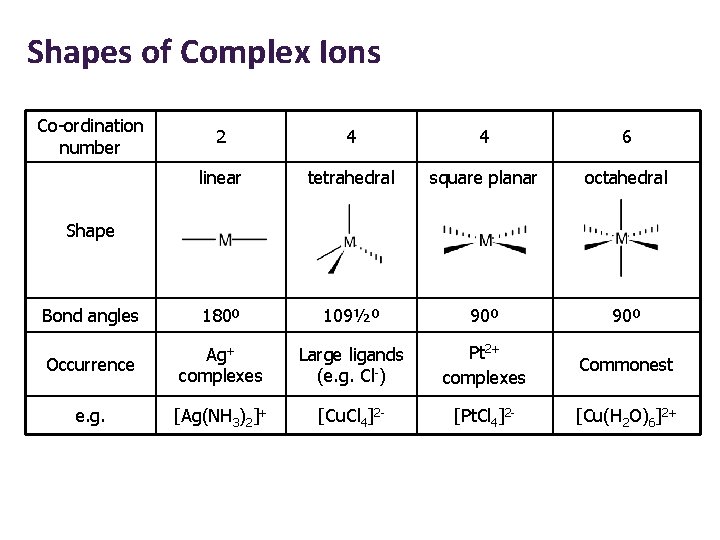
Shapes of Complex Ions Co-ordination number 2 4 4 6 linear tetrahedral square planar octahedral Bond angles 180º 109½º 90º Occurrence Ag+ complexes Large ligands (e. g. Cl-) Pt 2+ complexes Commonest e. g. [Ag(NH 3)2]+ [Cu. Cl 4]2 - [Pt. Cl 4]2 - [Cu(H 2 O)6]2+ Shape
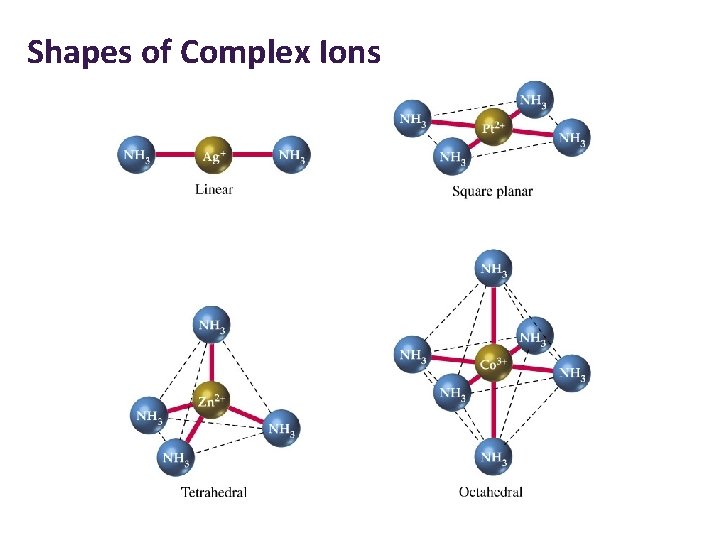
Shapes of Complex Ions

Octahedral Complexes • • • Most common ligands – water, ammonia and hydroxide ion Only uses one lone pair to form a coordinate bond Six ligands – six-fold coordination Electron pair donor in each ligand is an element from Period 2 (N and O) This means the ligands are approximately equal size Name the following: a) b) c) [Mn(H 2 O)6]2+ [Fe(H 2 O)4(OH)2] [Al(OH)6]3 - 1) Wha t is unusu al about t he final com plex? 2) Draw the shape of each complex once nam ed

Tetrahedral Complexes • Less common • Chloride ions act as ligands • Chlorine is a Period 3 element therefore ions are bigger than water, ammonia and hydroxide • Cannot fix 6 chloride ions around the central metal ion • Can fit 4 • [Cu. Cl 4]2 -

Linear Complexes • Less common • Reactive ion present in Tollen’s reagent (ammoniacal silver nitrate) • Silver behaves differently to the other transition metals looked at • Due to being in Period 5, not Period 4 • [Ag(NH 3)2]+

Shapes of Complex Ions Task 1 from booklet (p 5). . . For each of the following complexes: • • • Draw the complex Name the shape Show bond angles Give the metal oxidation state Give the co-ordination number 1) [Ag(CN)2]- 4) [Co(en)2 Cl 2 ]+ 2) [Cr(NH 3)6]3+ 5) [Pt(NH 3)2 Cl 2] 3) [Ni(en)3]2+ 6) [Fe(C 2 O 4)3]4 -
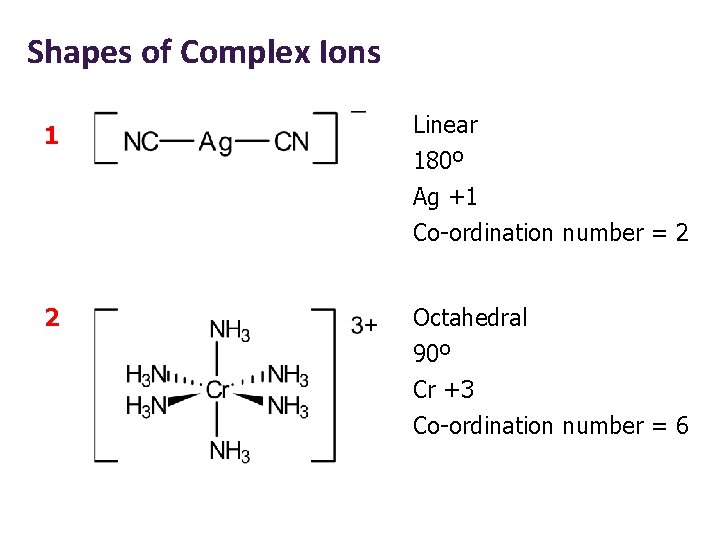
Shapes of Complex Ions 1 Linear 180º Ag +1 Co-ordination number = 2 2 Octahedral 90º Cr +3 Co-ordination number = 6
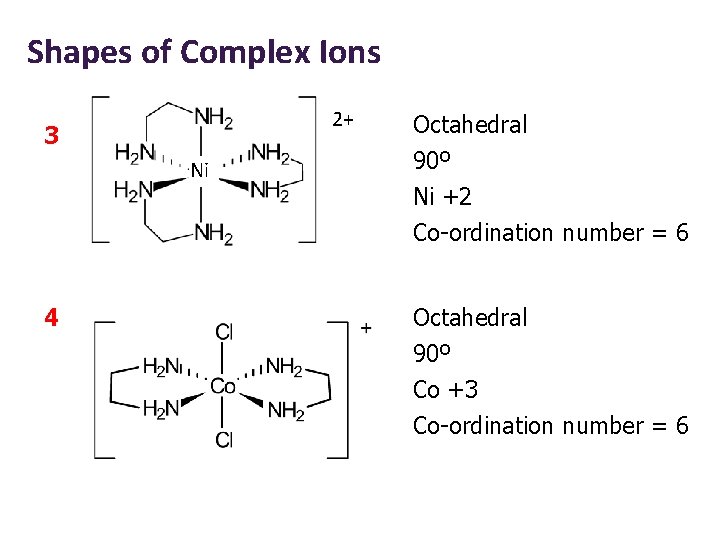
Shapes of Complex Ions 2+ 3 Ni Octahedral 90º Ni +2 Co-ordination number = 6 4 Octahedral 90º Co +3 Co-ordination number = 6
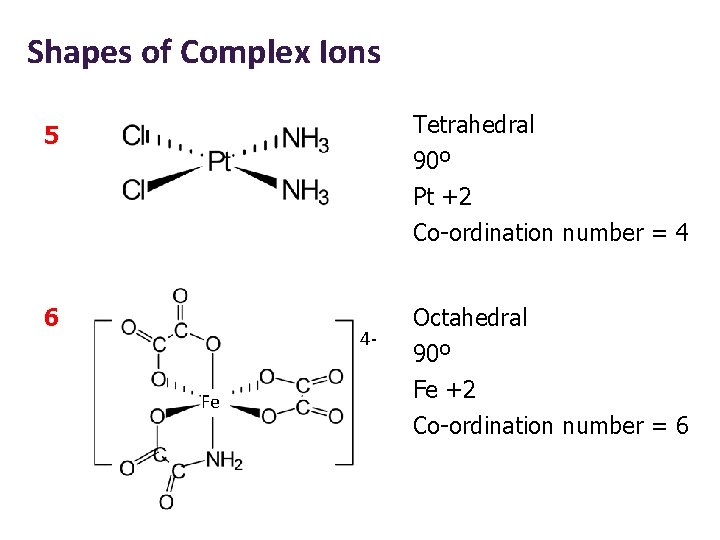
Shapes of Complex Ions Tetrahedral 90º Pt +2 5 Co-ordination number = 4 6 4 Fe Octahedral 90º Fe +2 Co-ordination number = 6

Summary Questions 1. A complex that forms between cobalt ions and nitrate ions (NO 2 -) has the abbreviated formula [Co(NO 2)6]3+. Predict its shape and the oxidation number of the transition metal ion. 2. A complex forms between nickel (II) ions and chloride ions. Predict its shape, name and formula.
- Slides: 18