Do Now Before the bell rings Pick up

Do Now Before the bell rings • Pick up today’s handouts • Get your interactive notebook • Sit at any desk • Place your States of Matter homwork on your desk with your name on it After the bell • Guess Who? • I will give you 3 -5 facts about someone in the classroom. You must guess who the person is.

Agenda Do Now (5 min) q q Remediation Topic (10 min) q Warm Up/Lab Discussion (5 min) q Finish notes: States of Matter (5 min) q Lab Discussion (10 min) q Independent Practice (20 min) q Exit Slip (5 min) q Intro to Physical/Chemical (20 min) q. Closeout (2 min)

Remediation Topic: How? • C – None • H – Raise hand • A – Identifying Lab Equipment • M – None • P – Answering questions to the right, recording responses on left page, discussing with group each prompt What? 1. What is it used for? What is its purpose in the lab? 2. What unit does it measure in? 3. What type of substances do you use with it? 4. What is it called?

Announcements • Quiz on scientific method is in the gradebook • Any non-takers… • Unit 1 Test was changed to Thursday/Friday next week • Now…you need your first reference sheet

Warm Up How? • C – Whispers • H – Raise hand • A – Answer with group • M – None • P – Record responses on left What? (3 min) • Below is the heating curve of water, but I hit my head last night and can’t remember how to label it. page, head up, discussing thoughts and providing input to the discussion • Using your notes and your observations from our lab, redraw the graph and label: • Solid, Liquid, Gas, Melting and Vaporization
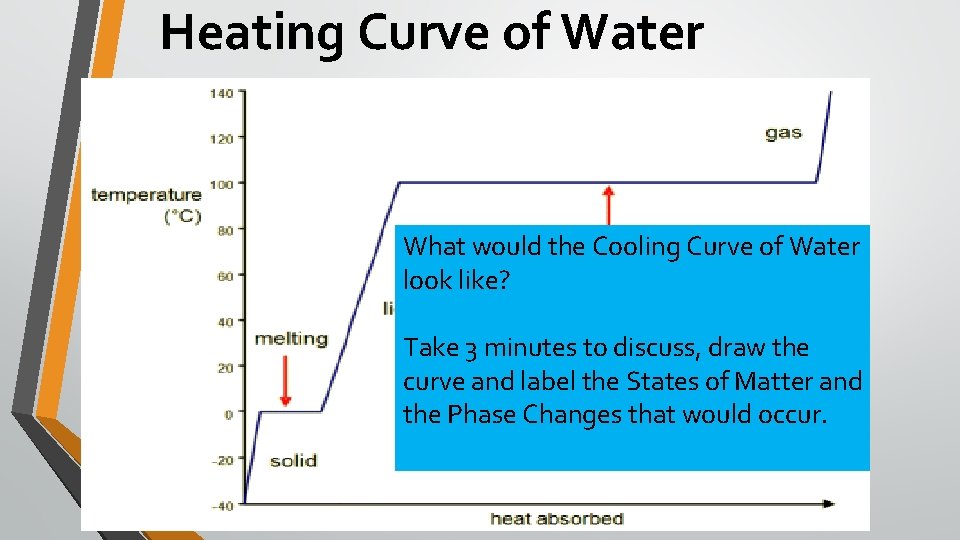
Heating Curve of Water What would the Cooling Curve of Water look like? Take 3 minutes to discuss, draw the curve and label the States of Matter and the Phase Changes that would occur.

Class Discussion: Heating Curve How? • C – One person at a time • H – Raise hand • A – Discussing key takeaways from lab • M – None, in seat • P – Taking notes from discussion, raising hand, volunteering input, head up, alert, supportive of peers What? • At one point in your lab, the temperature stayed relatively constant even though heat was being added. • Where did the energy go? • Why didn’t the temperature increase?

th 4 state of matter? • What happens when you raise the temperature to extreme levels…. does it stay a gas forever? • Plasma! • Plasma is different from a gas, because it is made up of groups of positively and negatively charged particles. • Like other gases has an indefinite shape and indefinite volume. • Examples? • Fluorescent bulbs • Lightening
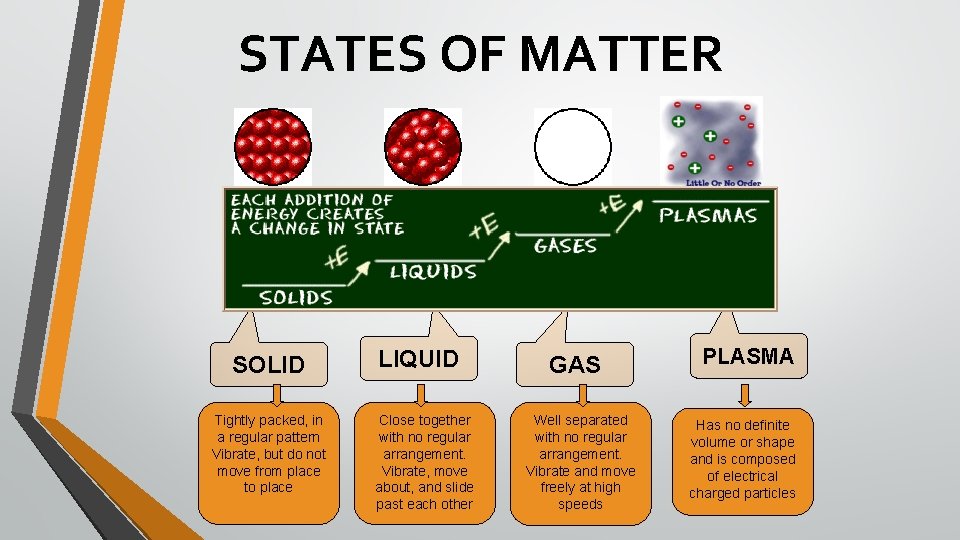
STATES OF MATTER SOLID Tightly packed, in a regular pattern Vibrate, but do not move from place to place LIQUID Close together with no regular arrangement. Vibrate, move about, and slide past each other GAS Well separated with no regular arrangement. Vibrate and move freely at high speeds PLASMA Has no definite volume or shape and is composed of electrical charged particles

Activity: Independent Practice How? • C – None • H – Raise hand • A – Answering questions as a group to classify matter • M – None • P – Recoding answers on left page, head up, reading and thinking through all questions, discussing with group What? • 10 minutes Discuss for 5 minutes Verify & Review

Exit Slip How? • C – No talking • H – Raise hand • A – Taking exit slip • M – In seat • P – Completing exit slip without notes and turning in What? (5 min) • If you finish early, summarize what you learned at the bottom of your notes OR answer today’s guiding question

Physical and Chemical Properties/Changes Objectives: 1. Identify physical and chemical properties of some materials 2. Identify examples of physical and chemical properties 3. Predict if a given property/change is physical or chemical

Guiding Question How do chemical changes impact our everyday lives?

Properties: Physical v. Chemical Physical • Can be observed without changing the substance into another substance. • Examples: • Color • Shape • Odor • Density • Melting/Boiling Point • Malleability • Elasticity Chemical • Indicates how a substance reacts with something else • The substance is chemically changed when observing it • Examples: • Combustibility • Flammability • Ability to rust • Rotting

Properties: Physical v. Chemical Examples: Physical Examples: Chemical

Changes: Physical v. Chemical Physical • Definition: A change in which matter changes form, but does not change composition. (an appearance change only) • Clues: • Change state (solid, liquid, gas) • Shape changes (crinkling, tearing) • Size changes (crushing, smashing) • No color change (except when dye added) • Dissolving Chemical • Def: : When pure substances mix and change into entirely new/different substances. • Clues: • Color change • Heat or light is given off • Gas Production (not from boiling) ( EX: fizzing) • Odor change • Precipitate (e. g. powder) forms • Usually irreversible • Energy Change (usually seen as a temperature change)

Changes: Physical v. Chemical Example: Physical Examples: Chemical

Example Problem Given the particle diagram representing four molecules of a substance: Which particle diagram best represents this same substance after a physical change has taken place? A chemical change?

Metals vs. Nonmetals

Closeout • Reminder: Your Unit 1 Test is Thursday/Friday • Homework is due next class • Submit any burning questions to the box • Next class we will finish investigating Physical/Chemical and review for the test.

Agenda Do Now (5 min) q q Remediation Topic (10 min) q Warm Up (5 min) q INM (25 min) q Lab (20 min) q Shot Put (10 min) q Exit Slip (5 min) q Closing (2 min)

Warm Up How? • C – Whispers • H – Raise hand • A – Answer with group, • M – None • P – Record responses on reverse side of page. What? (5 min) 1. Draw the heating curve of Sulfuric Acid (H 2 SO 4) a. Melting Point = 11˚ b. Boiling Point = 330˚ 2. Label a. 3 States of Matter b. Where Kinetic Energy changes 3. Answer: What is the Celsius scale based on?

Lab: Investigating P/C Changes How? • C- Low conversation about instructions, directions, and observations • H – Lab group raises hand in the air • A – Completing lab investigation, 5 minutes per station • M – At assigned station, with assigned group • P – Following instructions at each station, recording reflections and responses in notebook What? • At each station, describe each substance before beginning any procedure and include 4 physical properties (Ex: shape/size, state of matter, odor, color) • 5 minutes per station

Class Discussion: Heating Curve How? • C – One person at a time • H – Raise hand • A – Discussing key takeaways from lab • M – None, in seat • P – Taking notes from discussion, raising hand, volunteering input, head up, alert, supportive of peers What? • At two points in your lab, the temperature stayed relatively constant even though heat was being added. • Where did the energy go? • Why didn’t the temperature increase?

Activity: Shot Put • C – None How? • H – Raise hand • A – Answering questions as a group to classify matter • M – None • P – Recoding answers on left page, head up, reading and thinking through all questions, discussing with group What? • Earn 3 shots by answering the questions in each section. • 10 minutes

Exit Slip How? • C – No talking • H – Raise hand • A – Taking exit slip • M – In seat • P – Completing exit slip without notes and turning in What? (5 min) • If you finish early, summarize what you learned at the bottom of your notes OR answer today’s guiding question

Homework & Closure • What did we learn today? Don’t forget to stretch!!!! • Complete the 4 stretch questions
- Slides: 27