DO NOW 1What are the four variables used
DO NOW: 1)What are the four variables used to describe a gas? 2) If the volume of a gas decreased, the pressure of the would (increase/decrease)

Day ote N – 2 s Unit: Gas Laws Boyle’s and Charles’s Laws

After today you will be able to… • Explain the effect on gas properties using Boyle’s and Charles’s laws • Calculate an unknown pressure, temperature, or volume by solving algebraically
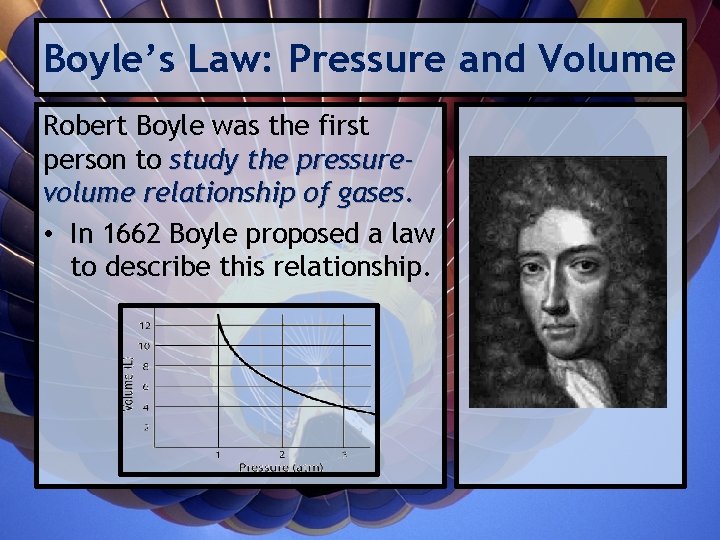
Boyle’s Law: Pressure and Volume Robert Boyle was the first person to study the pressurevolume relationship of gases. • In 1662 Boyle proposed a law to describe this relationship.

“For a given mass of a gas at constant temperature, the volume of a gas varies inversely with pressure. ”

Boyle’s Law: Pressure and Volume We can simplify this relationship by the formula: P 1 V 1 = P 2 V 2 Where, P 1, P 2 = pressure in any unit (atm, k. Pa, mm. Hg), BUT they must match! V 1, V 2 = volume in any unit (usually, L or m. L), BUT they must match!

Boyle’s Law: Example A gas has a volume of 30. 0 L at 150 k. Pa. What is the volume of the gas at 0. 252 atm? P 1= 150 k. Pa x 1 atm P 1 V 1=P 2 V 2 = 1. 5 atm 101. 3 k. Pa V 1= 30. 0 L P 2= 0. 252 atm V 2= ? (1. 5 atm) (30. 0 L) = (0. 252 atm) (V 2) V 2 = 180 L
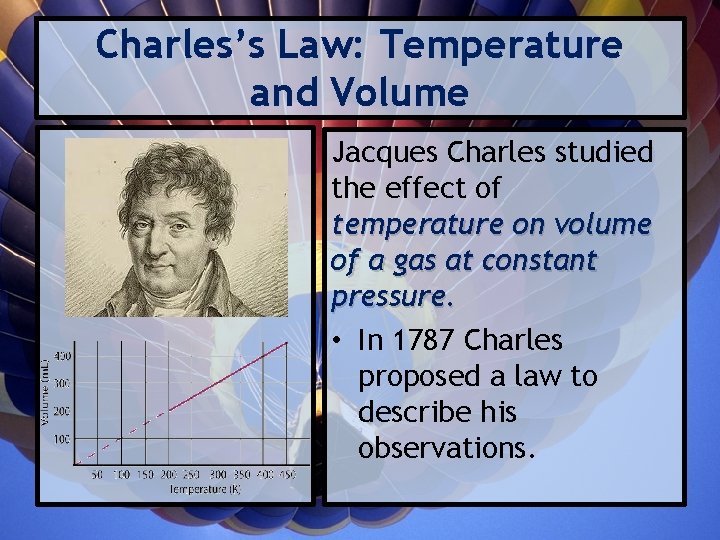
Charles’s Law: Temperature and Volume Jacques Charles studied the effect of temperature on volume of a gas at constant pressure. • In 1787 Charles proposed a law to describe his observations.

“For a given mass of a gas, at constant pressure, the volume of the gas is directly proportional to its temperature in Kelvin. ”

DEMONSTRATION!

Charles’s Law: Temperature and Volume We can simplify this relationship by the formula: V 1 V 2 = T 1 T 2 Where, V 1, V 2 = volume in any unit (L or m. L), BUT they must match! T 1, T 2 = temperature is always in Kelvin! (Recall, just add 273 + °C)

Charles’s Law: Example A gas has a volume of 4. 0 L at 27°C. What is its volume at 153°C? V 1= 4. 0 L T 1= 27°C +273= 300 K V 2= ? T 2= 153°C+273= 426 K V 1 V 2 = T 1 T 2 (4. 0 L) (300 K) = (V 2) (426 K) V 2 = 5. 7 L

DEMONSTRATION!

Complete your worksheet!

Questions? Complete WS 2
- Slides: 15