DNA POLYMERASES A Structural Approach C del Barrio

DNA POLYMERASES: A Structural Approach C. del Barrio, L. Embún, B. García, G. Miró, R. Pinacho, D. S. Rivera

Table of contents • INTRODUCTION • STRUCTURAL DESCRIPTION • DNA SYNTHESIS PATHWAY • DNA-ENZYME INTERACTIONS • ACTIVE SITE • EVOLUTIVE FEATURES • CONCLUSIONS
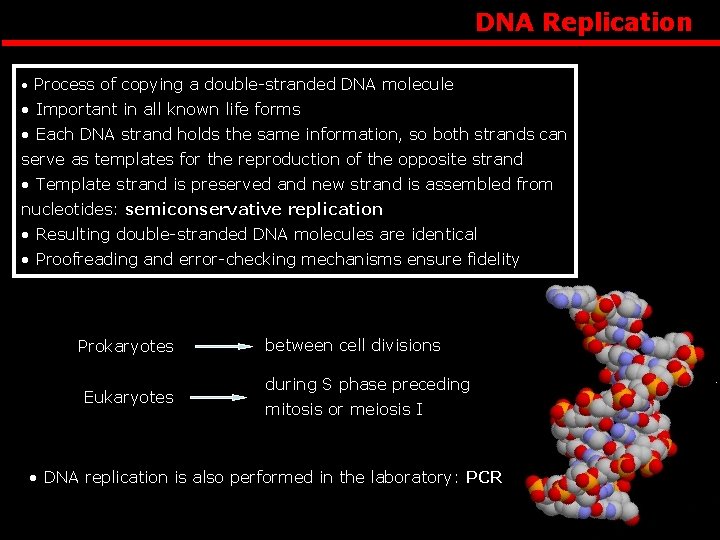
DNA Replication • Process of copying a double-stranded DNA molecule • Important in all known life forms • Each DNA strand holds the same information, so both strands can serve as templates for the reproduction of the opposite strand • Template strand is preserved and new strand is assembled from nucleotides: semiconservative replication • Resulting double-stranded DNA molecules are identical • Proofreading and error-checking mechanisms ensure fidelity Prokaryotes Eukaryotes between cell divisions during S phase preceding mitosis or meiosis I • DNA replication is also performed in the laboratory: PCR
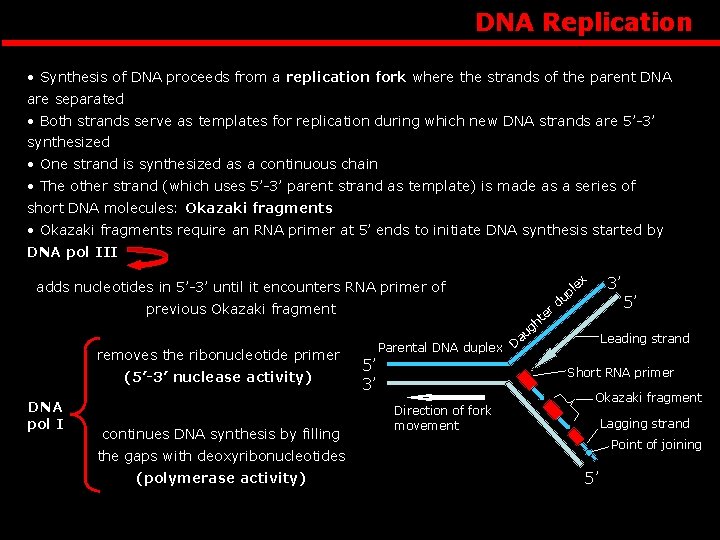
DNA Replication • Synthesis of DNA proceeds from a replication fork where the strands of the parent DNA are separated • Both strands serve as templates for replication during which new DNA strands are 5’-3’ synthesized • One strand is synthesized as a continuous chain • The other strand (which uses 5’-3’ parent strand as template) is made as a series of short DNA molecules: Okazaki fragments • Okazaki fragments require an RNA primer at 5’ ends to initiate DNA synthesis started by DNA pol III ex adds nucleotides in 5’-3’ until it encounters RNA primer of previous Okazaki fragment removes the ribonucleotide primer (5’-3’ nuclease activity) DNA pol I continues DNA synthesis by filling r te 3’ l up d h g au Parental DNA duplex D 5’ 3’ Leading strand Short RNA primer Direction of fork movement Okazaki fragment Lagging strand Point of joining the gaps with deoxyribonucleotides (polymerase activity) 5’ 5’

DNA Replication OTHER IMPORTANT ENZYMES INVOLVED IN DNA REPLICATION • Helicase Separates the double-helical configuration • Topoisomerase Catalyzes and guides unknotting of DNA (topological unlinking of the 2 strands) • SSB / SSBP Bind to single-strands, keeping them separated and allowing DNA replication machinery to perform its function • RNA primase Performs new RNA primer synthesis No known DNA pol can initiate the synthesis of a DNA strand without initial RNA primers • DNA ligase Its nick sealing joins new Okazaki fragment to the growing chain

DNA polymerase families Family Main members Features Are they crystallized? A Prokaryotic DNA pol I mitochondrial pol γ phage pols T 3, T 5 and T 7 Found primarily in organisms related to prokaryotes Yes [92 entries] B Phage pols T 4 and T 6 herpes virus pol archeal pol “Vent” mammalian pols α, δ and ε Present in phages, viruses, archea and eukaryotes Many of these pol function replicate the host genome Yes [17 entries] X Mammalian pols β, λ and μ Function during DNA repair Yes [117 entries] RT RTs from retroviruses eukaryotic telomerases Use of a RNA template to synthesize the DNA strand Yes [158 entries] most MMLV and HIV Pol III Bacterial DNA pols Replicate the majority of bacterial genomes Yes [135 entries] Umu. C/Din. B Pols η, ι and κ Rev 1 (terminal deoxycytidyl transferase) TLS pols, which have low fidelity on undamaged templates and replicate through damaged DNA Yes [17 entries]

DNA polymerase I lineage Class Fold Superfamily Multi-domain proteins (alpha & beta) Folds consisting of 2 or more domains belonging to different classes DNA/RNA polymerases 3 morphological domains (“palm”, “thumb” and “fingers”) All members conserve the “palm” domain DNA/RNA polymerases “Palm” domain has a ferrodoxin-like fold, related to that of an adenylyl cyclase domain Family DNA polymerase I Protein domain DNA polymerase I (Klenow fragment) Species Escherichia coli Termophilus aquaticus 6 members

DNA polymerase I domains polymerase 3’ 3’-5’ exonuclease 5’-3’ exonuclease DNA template 5’ 5’ 3’ primer C N 928 518 324 1 residue number Klenow fragment distance between polymerase active site and exonuclease binding site = 30 Å
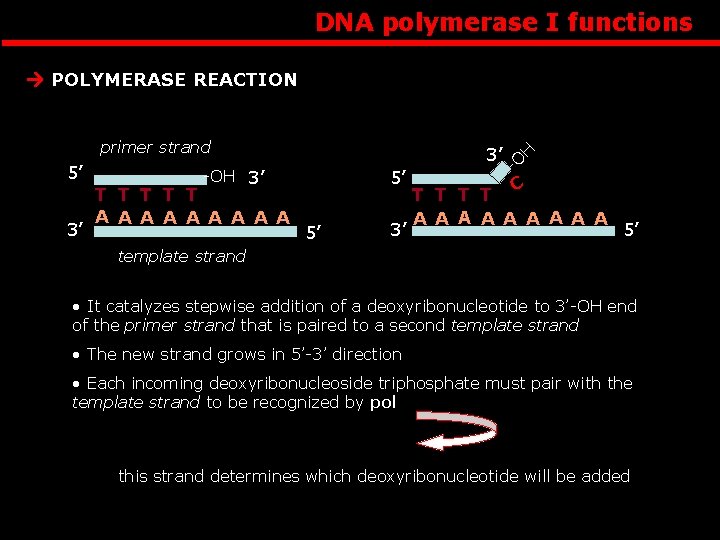
DNA polymerase I functions primer strand 3’ -OH 3’ T T T A A A A A 5’ 5’ 3’ -O 5’ 3’ H POLYMERASE REACTION T T C A A A A A 5’ template strand • It catalyzes stepwise addition of a deoxyribonucleotide to 3’-OH end of the primer strand that is paired to a second template strand • The new strand grows in 5’-3’ direction • Each incoming deoxyribonucleoside triphosphate must pair with the template strand to be recognized by pol this strand determines which deoxyribonucleotide will be added

DNA polymerase I functions 3’-5’ EXONUCLEASE ACTIVITY 3’ 5’ A A A T T G T T T hydrolysis site 5’ 3’ 3’ • One domain catalyzes hydrolysis of nucleotides at the 3’ end of DNA chains • To be removed, a nucleotide must have a free 3’-OH terminus and must not be part of a double helix 5’-3’ EXONUCLEASE ACTIVITY 3’ 5’ A A A T T G T T T 5’ 5’ 3’ hydrolysis site • The second domain hydrolyzes DNA starting from the 5’ end of DNA • It can occur at the 5’ terminus or at a bond several residues away from it • The cleaved bond must be in a double-helical region

Taq DNA polymerase I: structure and domains

Why do we use Taq polymerase I instead of the one from E. coli? RMS: 1. 75 Taq polymerase I (Klentaq fragment) [3 KTQ] E. coli polymerase I (Klenow fragment) [1 D 8 YA]

Taq polymerase I N-term C-term PDB: 1 CMW

Taq polymerase 5’-3’ exonuclease domain Klentaq fragment PDB: 1 CMW Linker region (links Klentaq to N-term fragment)

5’-3’ exonuclease domain N-term resolvase-like domain C-term SAM fold PDB: 1 CMW

Klentaq fragment Fingers Thumb Palm Linker region (links to N-term fragment) 3’-5’ exonuclease -like domain PDB: 3 KTQ

Klentaq fragment Fingers Thumb Palm 3’-5’ exonuclease -like domain PDB: 3 KTQ

DNA polymerase I Klenow fragment LARGE DOMAIN N C 14 9 13 12 8 7 • α+β Type with 6 -stranded antiparallel β sheet • Connection between β strands 9 and 12 makes a long excursion that builds up one side of the DNA-binding cleft • It contains helices L-Q as well as the antiparallel hairpin of β strands 11 and 12

DNA polymerase I Klentaq fragment LARGE DOMAIN PDB: 3 KTQ

DNA pol Klenow fragment SMALL DOMAIN nucleotide binding site C 5 2 3 4 1 • α/β Type with 1 antiparallel βstrand • 5 -stranded βsheet with 2 connecting helices and 1 helix (C) at the carboxy terminus (E. coli) UNIQUE TOPOLOGY FOR A MIXED β SHEET

Highly conserved regions

Highly conserved regions Region 1 Region 2 Motif A Motif B Region 6 Motif C

Motif A “DYSQIELR” Asp 610 Hydrophobic residues

Motif B “RRxh. Khh. NFGhh. Y” Arg 659 Lys 663 Gly 668 Tyr 671

Motif C His 784 “HDE” Asp 785

PATHWAY OF DNA SYNTESIS Part 1: DNA binding

Pathway of DNA Synthesis PPi d. NTP E+TP E-TP 1 1 2 3 4 E-TP-d. NTP 2 E-TPn+1 -PPi 3 E-TPn+1 4 Polymerase (E) binds with template-primer (TP) Appropriate d. NTP binds with polymerase-DNA complex Nucleophilic attack results in phosphodiester bond formation Pyrophosphate (PPi) is released rate limiting step phosphodiester bond formation conformational change preceding nucleotide incorporation dynamic interactions between polymerase with its nucleic acid and d. NTP substrates

Pathway of DNA Synthesis PPi d. NTP E+TP E-TP 1 E-TP-d. NTP 2 E-TPn+1 -PPi 3 E-TPn+1 4 Polymerases undergo 4 significant conformational changes: • During DNA binding step • Subsequent to d. NTP binding step and immediately preceding chemical catalysis • Subsequent to nucleotide incorporation during PPi release • During translocation towards new primer 3’-OH terminus

First step: polymerase binds to template Template Region 1 PDB: 2 KTQ

Open conformation 1 REGION 1 2. 40 Å 3. 21 Å DNA PDB: 2 KTQ

Low d. NTPs concentration 2 Tyr 671 DCT OPEN CONFORMATION Stacking interaction 3 1 st base 2. 40Å 90º PDB: 2 KTQ 1 st base

DCT PDB: 2 KTQ

When d. NTPs concentration increases. . . CLOSED CONFORMATION RMS: 0. 75 Open conformation (2 KTQ) Closed conformation (3 KTQ)

Open conformation (2 ktq. pdb) Closed conformation (3 ktq. pdb) 40º 11 -12 Å O-helix

Closed conformation O-helix Arg 587 Arg 659 His 639 Lys 663 Gln 613 Tyr 671 Asp 610 Asp 785 PDB: 3 KTQ

Hydrophobic pocket Tyr 671 Phe 667 Tyr 671 PDB: 3 KTQ 4. 11Å

Hydrogen bonding d. NTP ~3Å Hydrogen bonds PDB: 1 QSS

Structure d. NTP Base Pentose Triphosph ate α γ β PDB: 1 QSS

Electrostatic Interaction α- phosphate Arg 587 5, 6Å + PDB: 1 QSS

Electrostatic Interaction Gln 613 4, 07Å + - β phosphate PDB: 1 QSS

Electrostatic Interaction Lys 663 + - 5, 02Å β phosphate Β- PDB: 1 QSS

Electrostatic Interaction + - His 639 ~5Å γ phosphate PDB: 1 QSS

Electrostatic Interaction - + Arg 659 2, 45 Å γ phosphate PDB: 1 QSS

Phe 667 4, 23Å Stacking interactions Tyr 671 3, 19Å Base PDB: 1 QSS

Metal-mediated interactions triphosphate Mg Asp 785 Asp 610 Catalytic site PDB: 3 KTQ

Taq polymerase: Active site Catalytic Triad: 3 active site carboxylates Motif A Asp 610 Motif C Asp 785 Glu 786 GLU 786 ASP 785 Equivalence in Escherichia coli: Asp 705 Glu 710 Asp 882 Glu 883 2 viable triads!! ASP 610 PDB: 3 KTQ

Taq polymerase: Active site Mg A Catalytic Triad: Mg B PDB: 3 KTQ

Taq polymerase: Active site Pα Coordination of the Mg B Pβ Pγ Asp 785 Tyr 611 Asp 610 PDB: 3 KTQ

Taq pol : Active site Coordination of the Mg B PO 4β PO 4α PO 4γ Asp 785 Asp 610 Tyr 611 PDB: 3 KTQ

Taq pol : Active site PO 4 α OH 2 O Asp 785 OMg. A O- Coordination of the Mg A OH 2 O 3’OHPDB: 3 KTQ OAsp 610

Distance between atoms Asp 610 Asp 785 Tyr 611 Pa Pb Pg Mg. A Asp 610 3 2, 77 3, 2 5, 29 2, 86 2, 09 Asp 785 3, 04 3, 14 2, 85 4, 38 2, 12 Tyr 611 4, 61 3, 24 2, 86 2, 24 Pa 3, 4 3, 53 2, 39 Pb 3, 54 2, 214 Pg 2, 228 Mg. A Asp 610 Asp 785 HOH 3003 HOH 3125 Pa 3'OH Mg. B Asp 610 3, 85 3, 4 3, 73 3, 2 ? 2, 37 Asp 785 4, 84 3, 62 2. 81 ? 2, 48 HOH 3003 3, 65 3, 02 ? 2, 54 HOH 3125 4, 312 ? 2, 2 Pa ? 2, 24 3'OH ? Mg. B 3, 78 Mg. A

Taq pol : Active site Coordination of the Mg A: Distances 3. 8 A 2. 2 A PDB: 3 KTQ

Structural Superposition Polymerases I from Thermus aquaticus, E. Coli, B. Stearothermofilus and Bateriophage T 7 Cluster: (1 T 7 P_phage & 3 KTQ_Taq 1 D 8 Y_KF 1 XWL_Bst) Sc 7. 04 RMS 2. 28 PDB: 3 KTQ, 1 D 8 Y, 1 XLW, 1 T 7 P

Structural Superposition Score = 7, 75 RMSD=1, 86 *alignfit PDB: 3 KTQ, 1 D 8 Y, 1 XLW

Evolution within the same genus

Evolution among eukaryotic organisms

Conclusions • These enzimes have a characteristic structure, similar to a hand with 3 differentiated domains: palm, fingers and thumb. • Distinct conformational changes lead to the appropriate DNA sinthesis pathway. • The active site is the most conserved part of the sequence and its mechanism is based on metal coordination complexes. • In other enzimes with polymerase activity, like RT, the 3 Ddisposition and sequence are different, but similar mechanisms have been kept. • Structure and sequence are highly and significantly conserved among some polymerases, determining its importance in different organisms.

Preguntas tipo PEM Pregunta 1 Señale la respuesta FALSA, referente a las familias de DNA polimerasas: a) La DNA polimerasa I procariota forma parte de la familia Umu. C/Din. B b) La familia RT contiene las retrotranscriptasas de los retrovirus c) Los miembros de la familia X funcionan durante la reparación del DNA d) La familia Pol III contiene la mayoría de DNA polimerasas bacterianas e) Existen 6 familias de DNA polimerasas

Preguntas tipo PEM Pregunta 2 ¿Qué dominio/s de la DNA polimerasa I de E. coli constituye/n el fragmento Klenow? a) Dominio polimerasa b) Dominio 3’-5’ exonucleasa c) Los dos anteriores d) Dominio 5’-3’ exonucleasa e) Todos los anteriores

Preguntas tipo PEM Pregunta 3 Señale la respuesta VERDADERA, en relación a la topología de los dominios del fragmento Klenow: a) El dominio grande es del tipo α+β b) El dominio pequeño es del tipo α/β c) Las dos anteriores d) La lámina β mixta del dominio pequeño constituye una topología única e) Todas las anteriores

Preguntas tipo PEM Pregunta 4 Respecto a la vía de síntesis de DNA, ¿qué paso/s es/son limitante/s en la reacción? 1. Unión de la polimerasa al cebador molde (template primer) de DNA 2. Cambio conformacional de la polimerasa, previo a la incorporación de d. NTPs 3. Liberación de pirofosfato (PPi) 4. Formación del enlace fosfodiéster a) 1, 2 y 3 b) 1 y 3 c) 2 y 4 d) 4 e) 1, 2, 3 y 4

Preguntas tipo PEM Pregunta 5 Señale la respuesta FALSA, referente al fragmento klenow de la DNA polimerasa I: a) El fragmento klenow se encuentra en la región N terminal de la DNA polimerasa I. b) El fragmento klenow contiene el dominio exonucleasa 3’-5’ y el dominio con actividad polimerasa. c) El dominio con actividad polimerasa tiene una morfología similar a una mano derecha con tres dominios bien diferenciados: palma, pulgar y dedos. d) Cuando hablamos de la Taq polimerasa I, el fragmento klenow pasa a denominarse fragmento klentaq. e) El dominio palma es el más conservado.

Preguntas tipo PEM Pregunta 6 Sobre la estructura de la DNA polimerasa I, señala la respuesta VERDADERA: a) El dominio 5’-3’ exonucleasa tiene una estructura típica de barril beta y se encuentra englobado en el dominio pulgar. b) El dominio 5’-3’ exonucleasa se compone de dos dominios: un dominio N terminal tipo resolvasa, y otro dominio C terminal con un plegamiento tipo SAM. c) Los dominios palma, pulgar y dedos no son relevantes en la interacción con el DNA y por tanto no participan en la reacción de formación del enlace fosfodiéster. d) El dominio 3’-5’ exonucleasa del fragmento klenow no está formado por ninguna lámina beta. e) El dominio pulgar del fragmento klenow presenta un plegamiento tipo sándwich de dos capas.

Preguntas tipo PEM Pregunta 7 Sobre las regiones conservadas de la DNA pol, señala la respuesta VERDADERA: a) Las regiones más conservadas son los dominios A, B y C. b) El dominio A contiene la Asp 610, que juega un papel importante en la interacción con el DNA. c) Las dos anteriores son ciertas. d) Algunos aminoácidos del motivo B y del motivo C tienen un papel muy importante en la estabilización del d. NTP entrante. e) Todas las anteriores son ciertas.

Preguntas tipo PEM Pregunta 8 Respecto a la conformación abierta de la DNA polimerasa señala la respuesta FALSA: a) La Tyr 671 del dominio dedos ocupa el lugar que correspondería a la primera base del DNA. b) La Tyr 671 establece puentes de hidrógeno con la primera base del DNA. c) La primera base del DNA experimenta un giro de 90º respecto al eje de la hélice. d) El d. NTP se apila sobre el primer par de bases adyacente a la Tyr 671.

Preguntas tipo PEM Pregunta 9 ¿En qué mecanismo se basa el centro activo de una DNApolimerasa? a) En un complejo de coordinación octahédrico en torno a iones metálicos divalentes. b) En una coordinación octagonal entre residuos de la polimerasa y dos átomos de Mg++. c) En la unión covalente con el sustrato, en este caso el DNA. d) En una interacción forzada con el sustrato mediada por puentes salinos. e) Ninguna de las anteriores.

Preguntas tipo PEM Pregunta 10 ¿Por qué se caracterizan evolutivamente las DNA-polimerasas? a) Por el grado de conservación estructural de los dominios A, B y C del centro activo y por su mecanismo de acción. b) Por la elevada identidad de secuencia y conservación estructural de los dominios A, B y C del centro activo y por su mecanismo de acción. c) Por el grado de conservación de secuencia de los dominios A, B y C del centro activo y por su mecanismo de acción. d) Por adoptar una conformación espacial fija, gracias a la presencia de residuos hidrofóbicos en el bolsillo catalítico. e) No se han hallado evidencias de que estos enzimas estén relacionados entre sí evolutivamente.
- Slides: 67