Dmitri Mendeleevs Periodic Table of the Elements Gruppe
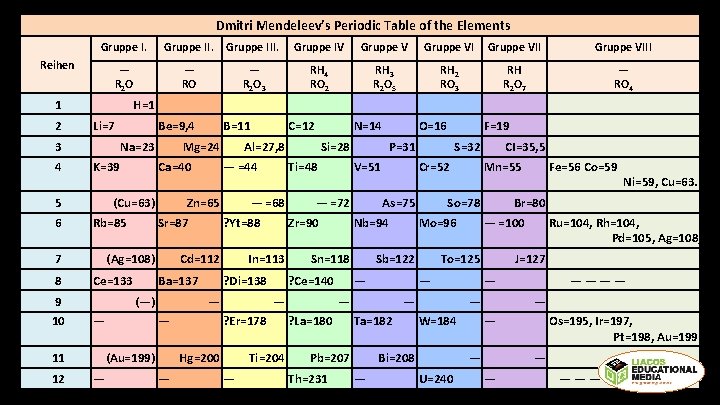
Dmitri Mendeleev’s Periodic Table of the Elements Gruppe III. Gruppe IV Gruppe VIII — R 2 O 3 RH 4 RO 2 RH 3 R 2 O 5 RH 2 RO 3 RH R 2 O 7 — RO 4 Reihen 1 2 3 4 5 6 H=1 Li=7 Na=23 K=39 9 10 C=12 N=14 O=16 F=19 Mg=24 Al=27, 8 Si=28 P=31 S=32 CI=35, 5 Ca=40 — =44 Ti=48 V=51 Cr=52 Mn=55 Fe=56 Co=59 (Ag=108) Ce=133 Cd=112 Ba=137 (—) — 11 12 B=11 Ni=59, Cu=63. (Cu=63) Zn=65 — =68 — =72 As=75 So=78 Br=80 Rb=85 Sr=87 ? Yt=88 Zr=90 Nb=94 Mo=96 — =100 Ru=104, Rh=104, Pd=105, Ag=108, 7 8 Be=9, 4 ? Di=138 — — (Au=199) — In=113 ? Ce=140 — ? Er=178 Hg=200 — Sn=118 Ti=204 — Sb=122 — — — ? La=180 — Ta=182 Pb=207 Th=231 To=125 — — W=184 Bi=208 — J=127 — — — U=240 ———— Os=195, Ir=197, Pt=198, Au=199. — — ————
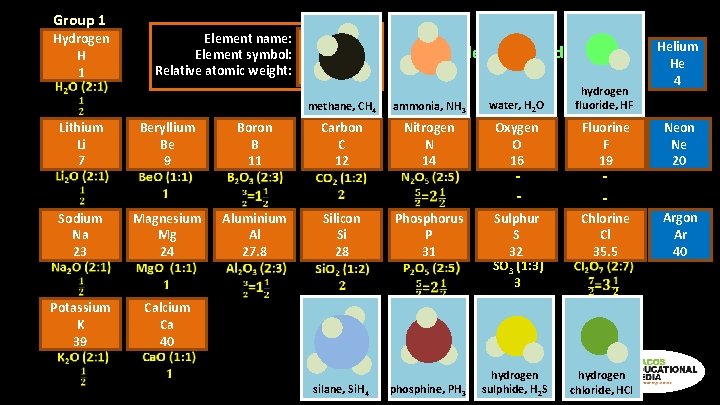
Group 1 Hydrogen H 1 Lithium Li 7 Element name: Element symbol: Relative atomic weight: Beryllium Be 9 Boron B 11 Hydrogen H 1 Mendeleev’s Periodic Table 1870 s methane, CH 4 ammonia, NH 3 water, H 2 O hydrogen fluoride, HF Carbon C 12 Nitrogen N 14 Oxygen O 16 Fluorine F 19 Neon Ne 20 Chlorine Cl 35. 5 Argon Ar 40 - Sodium Na 23 Magnesium Mg 24 Potassium K 39 Calcium Ca 40 Aluminium Al 27. 8 Helium He 4 Silicon Si 28 silane, Si. H 4 Phosphorus P 31 phosphine, PH 3 Sulphur S 32 SO 3 (1: 3) 3 hydrogen sulphide, H 2 S - hydrogen chloride, HCl
- Slides: 2