Dmitri Mendeleev Russian Invented periodic table Organized elements

Dmitri Mendeleev Russian Invented periodic table Organized elements by properties Arranged elements by atomic mass • Predicted existence of several unknown elements • Element 101 • • Dmitri Mendeleev
Dmitri Mendeléev
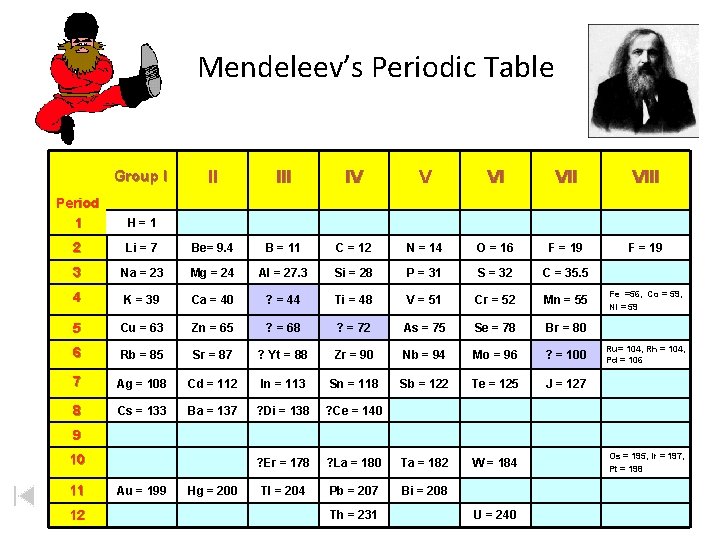
Mendeleev’s Periodic Table Group I II IV V VI VIII F = 19 Period 1 H=1 2 Li = 7 Be= 9. 4 B = 11 C = 12 N = 14 O = 16 F = 19 3 Na = 23 Mg = 24 Al = 27. 3 Si = 28 P = 31 S = 32 C = 35. 5 4 K = 39 Ca = 40 ? = 44 Ti = 48 V = 51 Cr = 52 Mn = 55 5 Cu = 63 Zn = 65 ? = 68 ? = 72 As = 75 Se = 78 Br = 80 6 Rb = 85 Sr = 87 ? Yt = 88 Zr = 90 Nb = 94 Mo = 96 ? = 100 7 Ag = 108 Cd = 112 In = 113 Sn = 118 Sb = 122 Te = 125 J = 127 8 Cs = 133 Ba = 137 ? Di = 138 ? Ce = 140 ? Er = 178 ? La = 180 Ta = 182 W = 184 Tl = 204 Pb = 207 Bi = 208 Fe =56, Co = 59, Ni = 59 Ru= 104, Rh = 104, Pd = 106 9 10 11 12 Au = 199 Hg = 200 Th = 231 U = 240 Os = 195, Ir = 197, Pt = 198
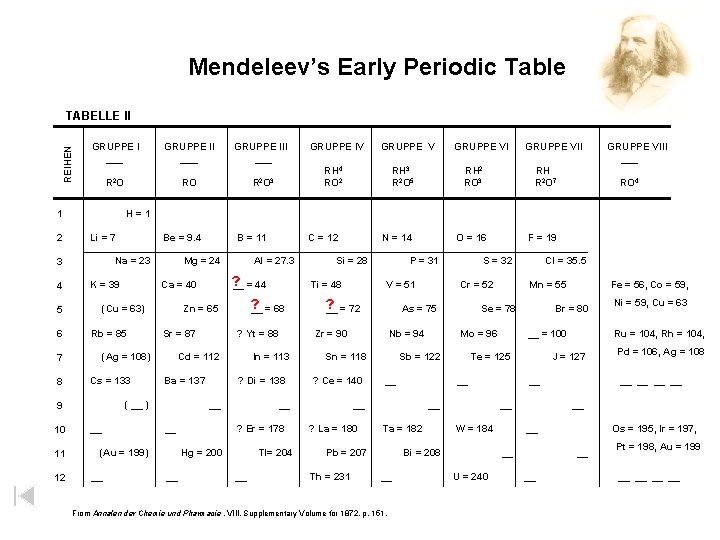
Mendeleev’s Early Periodic Table REIHEN TABELLE II GRUPPE I ___ Li = 7 K = 39 11 12 RH 3 R 2 O 5 Cs = 133 Sr = 87 GRUPPE VI RH 2 RO 3 In = 113 ? Di = 138 __ __ (Au = 199) __ ? Yt = 88 Ba = 137 __ Si = 28 GRUPPE VII RH R 2 O 7 ? Er = 178 Tl= 204 __ V = 51 GRUPPE VIII ___ RO 4 Cr = 52 Nb = 94 Sn = 118 ? Ce = 140 From Annalen der Chemie und Pharmacie , VIII, Supplementary Volume for 1872, p. 151. __ W = 184 Pd = 106, Ag = 108 __ __ U = 240 Ni = 59, Cu = 63 Ru = 104, Rh = 104, J = 127 __ Bi = 208 __ __ = 100 __ Ta = 182 Fe = 56, Co = 59, Br = 80 Te = 125 __ Pb = 207 Mn = 55 Mo = 96 __ Cl = 35. 5 Se = 78 Sb = 122 __ ? La = 180 F = 19 S = 32 As = 75 Zr = 90 Th = 231 O = 16 P = 31 ? __ = 72 __ Hg = 200 N = 14 Ti = 48 ? __ = 68 Cd = 112 ( __ ) __ Al = 27. 3 Zn = 65 (Ag = 108) C = 12 ? __ = 44 Ca = 40 Rb = 85 9 10 GRUPPE V RH 4 RO 2 B = 11 Mg = 24 (Cu = 63) 7 8 Be = 9. 4 Na = 23 5 6 RO R 2 O 3 GRUPPE IV H=1 3 4 GRUPPE III ___ R 2 O 1 2 GRUPPE II ___ Os = 195, Ir = 197, __ __ Pt = 198, Au = 199 __ __
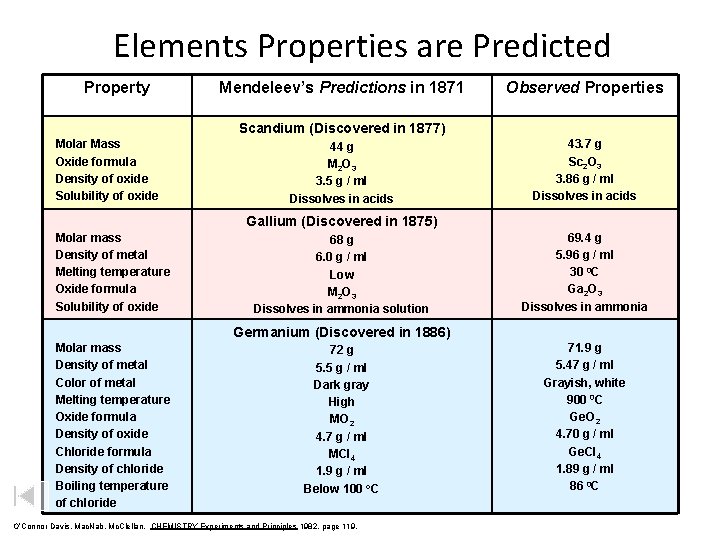
Elements Properties are Predicted Property Mendeleev’s Predictions in 1871 Observed Properties Scandium (Discovered in 1877) Molar Mass Oxide formula Density of oxide Solubility of oxide 44 g M 2 O 3 3. 5 g / ml Dissolves in acids 43. 7 g Sc 2 O 3 3. 86 g / ml Dissolves in acids Gallium (Discovered in 1875) Molar mass Density of metal Melting temperature Oxide formula Solubility of oxide 68 g 6. 0 g / ml Low M 2 O 3 Dissolves in ammonia solution 69. 4 g 5. 96 g / ml 30 0 C Ga 2 O 3 Dissolves in ammonia Germanium (Discovered in 1886) Molar mass Density of metal Color of metal Melting temperature Oxide formula Density of oxide Chloride formula Density of chloride Boiling temperature of chloride 72 g 5. 5 g / ml Dark gray High MO 2 4. 7 g / ml MCl 4 1. 9 g / ml Below 100 o. C O’Connor Davis, Mac. Nab, Mc. Clellan, CHEMISTRY Experiments and Principles 1982, page 119, 71. 9 g 5. 47 g / ml Grayish, white 900 0 C Ge. O 2 4. 70 g / ml Ge. Cl 4 1. 89 g / ml 86 0 C

Modern Periodic Table

Periodic Table of the Elements 1 2 3 H He 1 2 Li Be B C N O F Ne 3 4 5 6 7 8 9 10 Al Si P S Cl Ar 13 14 15 16 17 18 Na Mg 11 4 K 19 5 7 Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 12 38 Cs Ba 55 56 Fr Ra 87 88 * W 25 43 26 44 Re Os 75 76 27 28 29 47 30 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Po At Rn 84 85 86 Rf Db Sg Bh Hs Mt 104 105 106 107 108 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103

Modern Periodic Table • Henry G. J. Moseley • Determined the atomic numbers of elements from their X-ray spectra (1914) • Arranged elements by increasing atomic number • Killed in WW I at age 28 (Battle of Gallipoli in Turkey) 1887 - 1915

Introduction to the Periodic Table • Elements are arranged in seven horizontal rows, in order of increasing atomic number from left to right and from top to bottom. • Rows are called periods and are numbered from 1 to 7. • Elements with similar chemical properties form vertical columns, called families, which are numbered from 1 to 18. • Groups 1, 2, and 13 through 18 are the main group elements. • Groups 3 through 12 are in the middle of the periodic table and are the transition elements. • The two rows of 14 elements at the bottom of the periodic table are the lanthanides and actinides.
- Slides: 9