Dmitri Mendeleev order elements by atomic mass 1834

Dmitri Mendeleev • order elements by atomic mass 1834 - 1907 • saw a repeating pattern of properties • Periodic Law – When the elements are arranged in order of increasing relative mass, certain sets of properties recur periodically • used pattern to predict properties of undiscovered elements • where atomic mass order did not fit other properties, he re-ordered by other properties
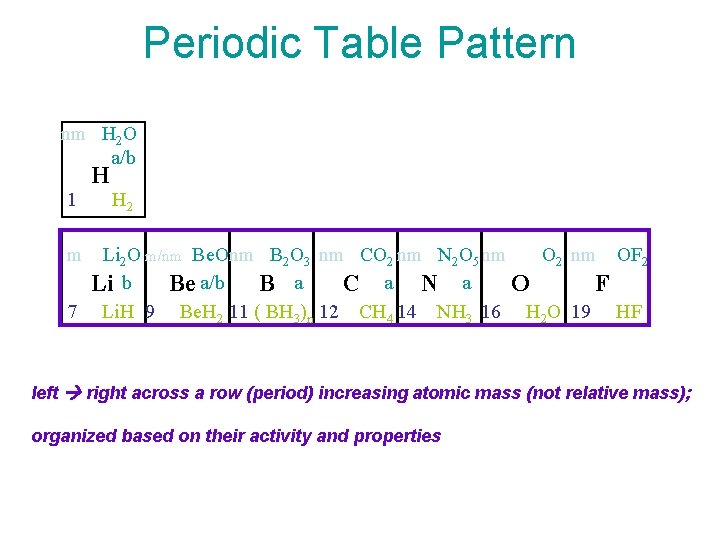
Periodic Table Pattern nm H 2 O a/b H 1 H 2 m Li 2 O m/nm Be. Onm B 2 O 3 nm CO 2 nm N 2 O 5 nm O 2 nm OF 2 Li b Be a/b B a C a N a O F 7 Li. H 9 Be. H 2 11 ( BH 3)n 12 CH 4 14 NH 3 16 H 2 O 19 HF left right across a row (period) increasing atomic mass (not relative mass); organized based on their activity and properties
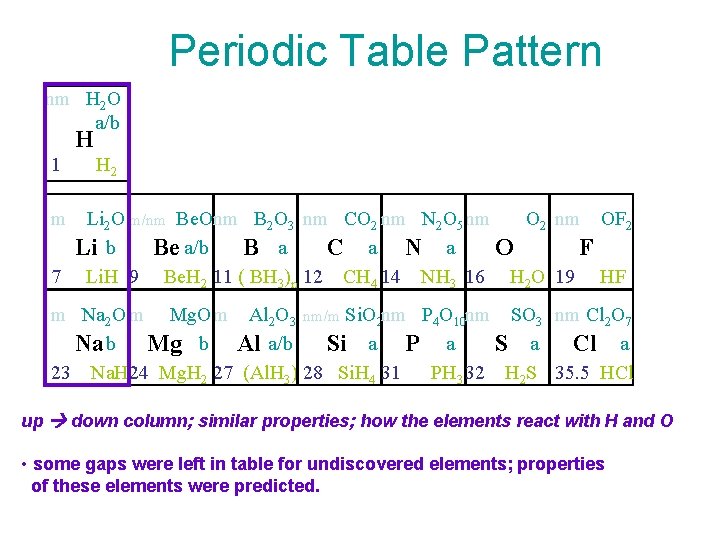
Periodic Table Pattern nm H 2 O a/b H 1 H 2 m Li 2 O m/nm Be. Onm B 2 O 3 nm CO 2 nm N 2 O 5 nm O 2 nm OF 2 Li b Be a/b B a C a N a O F 7 Li. H 9 Be. H 2 11 ( BH 3)n 12 CH 4 14 NH 3 16 H 2 O 19 HF m Na 2 O m Mg. O m Al 2 O 3 nm/m Si. O 2 nm P 4 O 10 nm SO 3 nm Cl 2 O 7 S a Cl a Na b Mg b Al a/b Si a P a 23 Na. H 24 Mg. H 2 27 (Al. H 3) 28 Si. H 4 31 PH 3 32 H 2 S 35. 5 HCl up down column; similar properties; how the elements react with H and O • some gaps were left in table for undiscovered elements; properties of these elements were predicted.
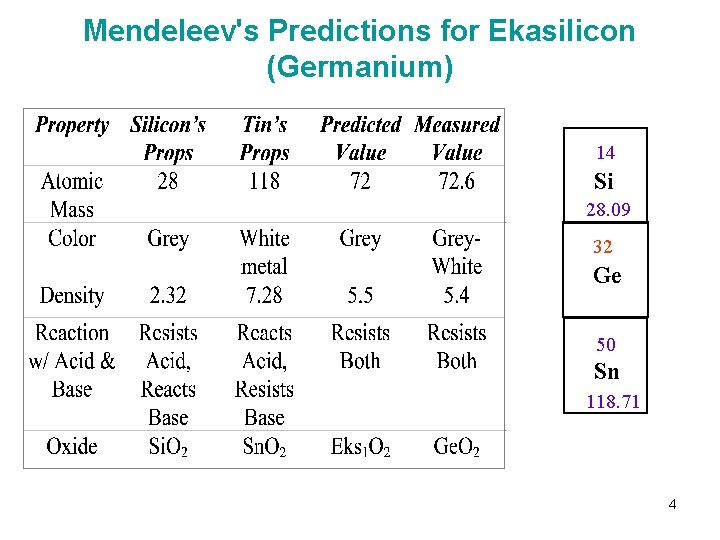
Mendeleev's Predictions for Ekasilicon (Germanium) 14 Si 28. 09 32 Ge 72. 61 50 Sn 118. 71 4
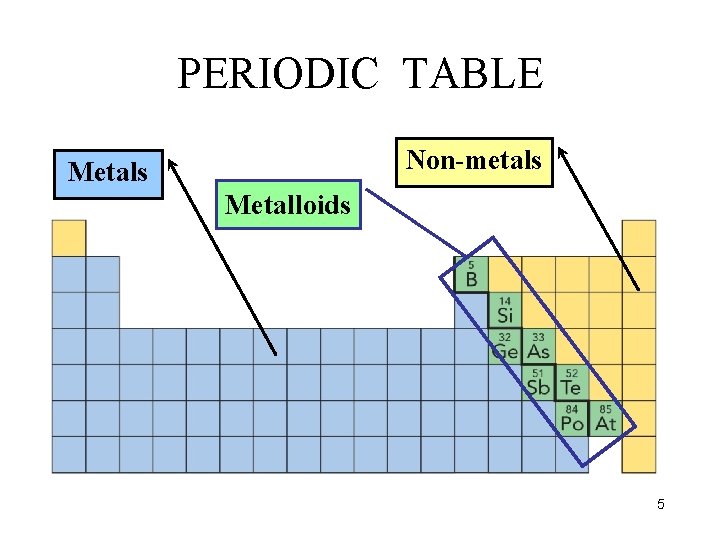
PERIODIC TABLE Non-metals Metalloids 5

PROPERTIES OF METALS & NON-METALS Metals Non-metals • Mostly solid • Can be solid, liquid or gas • Have shiny appearance • Have dull appearance • Good conductors of heat & electricity • Poor conductors of heat & electricity • Malleable & ductile • Brittle (if solid) • Lose electrons • Gain or share electrons 6

Metals Non-metals

METALLOIDS q Metalloids are elements that possess some properties of metals and some of non-metals. q The most important metalloids are silicon (Si) and germanium (Ge) which are used extensively in computer chips. 8

Metalloids Properties of Silicon shiny conducts electricity brittle
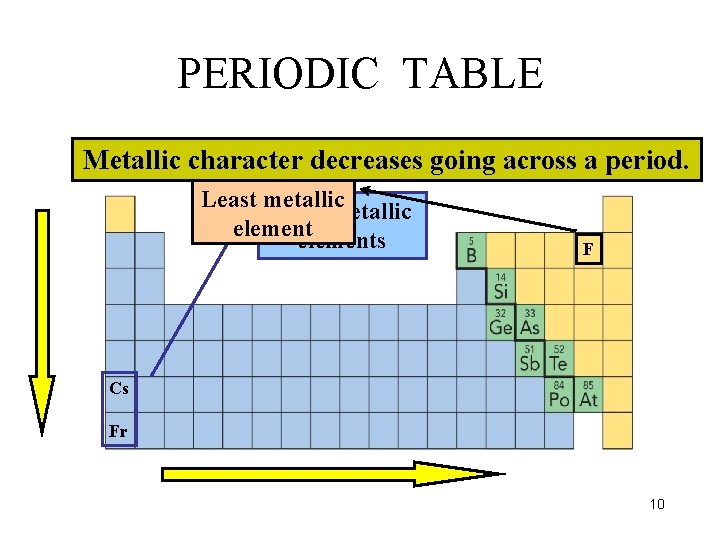
PERIODIC TABLE Metallic character decreases increases going down acrossaagroup period. Least metallic Most metallic elements F Cs Fr 10

PERIODIC TABLE q Seven elements exist as diatomic molecules. q All others exist as monatomic (single atom). 11
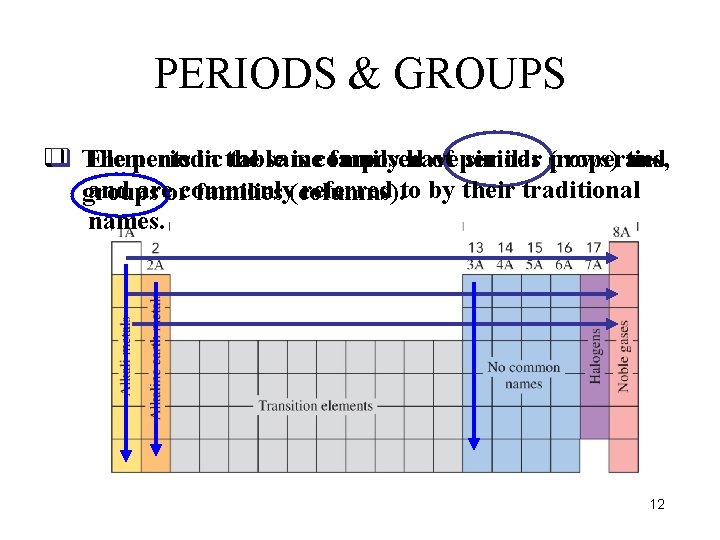
PERIODS & GROUPS q q The Elements periodic in the table same is composed family have of periods similar (rows) properties, and areorcommonly referred to by their traditional groups families (columns). names. 12
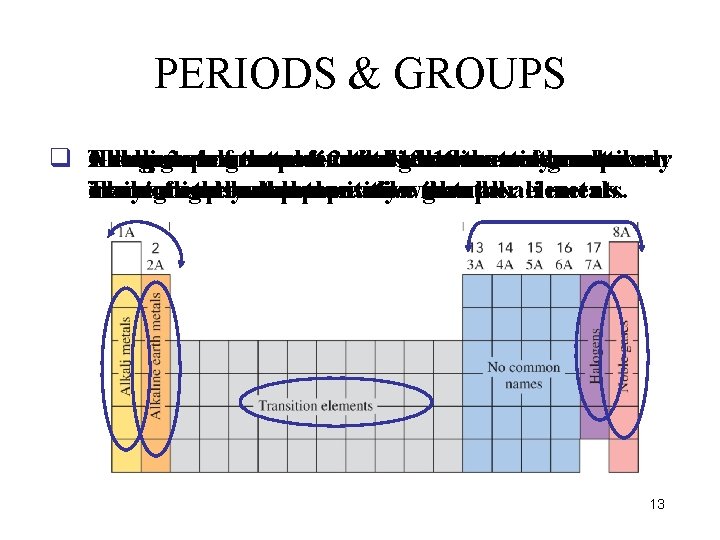
PERIODS & GROUPS q Group The group Elements Alkali Noble Halogens gases metals 2 elements in are ofare groups metals the areun-reactive most soft are 1 -2 inmetals called reactive between and 13 -18 gases alkaline-earth that nonmetals, the are that are main very referred aregroup commonly reactive. and metals. to occur as main-group They used in These elements nature in often metals light are only react or bulbs. called are as representative explosively compounds. less transition reactivewith metals. than groups. other alkali elements. metals. 13

Important Groups - Hydrogen • nonmetal • colorless, diatomic gas – very low melting point & density • reacts with nonmetals to form molecular compounds – HCl is acidic gas and H 2 O is a liquid • reacts with metals to form hydrides (negative ion of hydrogen = H-) – metal hydrides react with water to form H 2 (Metal-H + H 2 O H 2 + metal-OH) (Na. H + HOH H 2 + Na. OH)

Important Groups – IA, Alkali Metals • hydrogen usually placed here, though it doesn’t belong lithium • soft, low melting points, low density • flame tests ® Li = red, Na = sodium yellow, K = violet potassium • very reactive, never find uncombined in nature • tend to form water soluble rubidium compounds cesium 15

Important Groups – IIA, Alkali Earth Metals • harder, higher melting, and denser than alkali metals • flame tests ® Ca = red, Sr = red, Ba = yellow-green • reactive, but less than corresponding alkali metal • form stable, insoluble oxides from which they are normally extracted • oxides are basic = alkaline earth • reactivity with water to form H 2, beryllium magnesium calcium strontium barium

Important Groups – VIIA, Halogens • nonmetals • F 2 & Cl 2 gases; Br 2 liquid; I 2 solid • all diatomic • very reactive • react with metals to form ionic compounds • HX (X = halogen) are all acids – HF weak < HCl < HBr < HI fluorine chlorine bromine iodine

Important Groups – VIIIA, Noble Gases • all exist as gases at room temperature, – very low melting and boiling points • very unreactive, practically inert • very hard to remove electron or add an electron
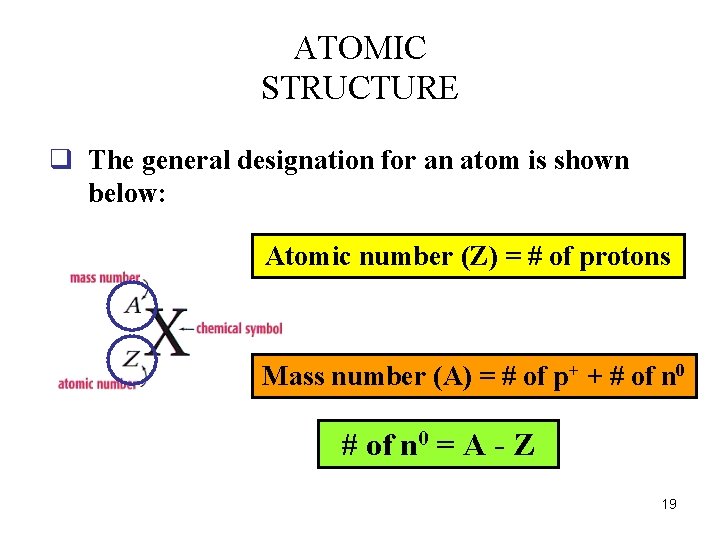
ATOMIC STRUCTURE q The general designation for an atom is shown below: Atomic number (Z) = # of protons Mass number (A) = # of p+ + # of n 0 = A - Z 19

Charged Atoms • The number of protons determines the element! – all sodium atoms have 11 protons in the nucleus • In a chemical change, the number of protons in the nucleus of the atom doesn’t change! – no transmutation during a chemical change!! – during radioactive and nuclear changes, atoms do transmute • Atoms in a compound are often electrically charged, these are called ions
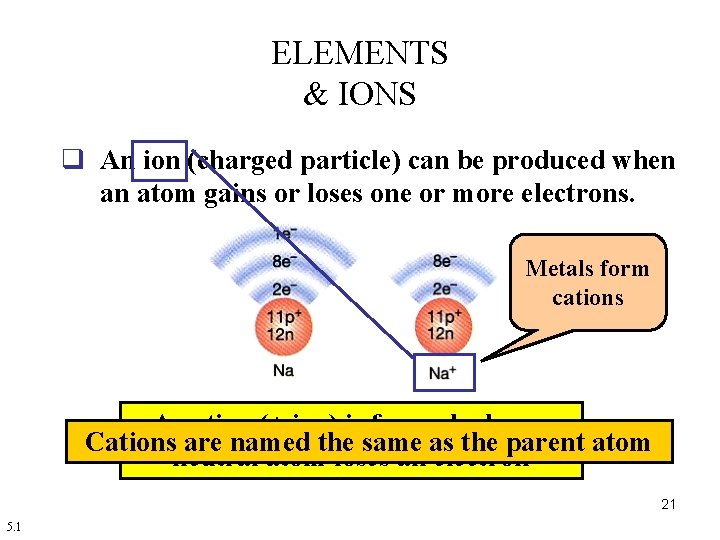
ELEMENTS & IONS q An ion (charged particle) can be produced when an atom gains or loses one or more electrons. Metals form cations A cation (+ ion) is formed when a Cations are named the same as the parent atom neutral atom loses an electron 21 5. 1
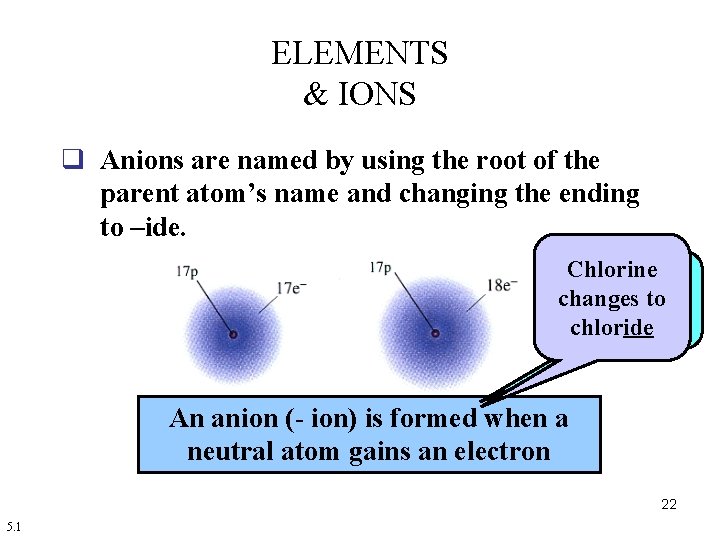
ELEMENTS & IONS q Anions are named by using the root of the parent atom’s name and changing the ending to –ide. Chlorine Non-metals changes to form anions chloride An anion (- ion) is formed when a neutral atom gains an electron 22 5. 1

Ion Charge & the Periodic Table • • Every element wants to be like the noble gases wants 0 or 8 valence electrons valence e- (outer electron shell used for bonding) He has zero valence e-
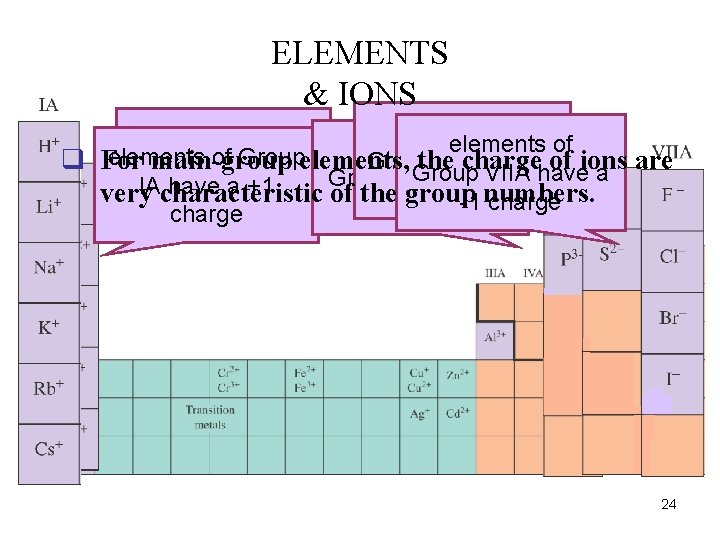
ELEMENTS & IONS elements of elements of Group q For. Group IIA have a main-group elements, the charge of ions are Group VIA have a Group VIIA have a Group VA have a IA have a +1 -2 charge very characteristic of the group numbers. +2 charge -1 charge -3 charge 24

Structure of the Nucleus (1877 -1956) • Frederick Soddy discovered that the same element could have atoms with different masses, which he called isotopes – there are 2 isotopes of chlorine found in nature, one that has a mass of about 35 amu and another that weighs about 37 amu • The observed mass is a weighted average of the weights of all the naturally occurring atoms – the atomic mass of chlorine is 35. 45 amu

Isotopes • Atomic mass - The average mass for a given element • all isotopes of an element have the same number of protons • isotopes of an element have different masses • isotopes of an element have different numbers of neutrons • isotopes are identified by their mass numbers – protons + neutrons
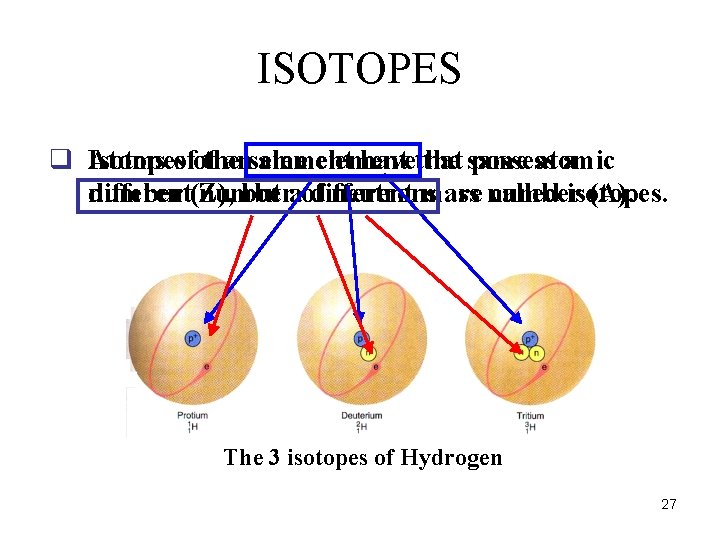
ISOTOPES q Atoms Isotopesofofthe ansame element havethat the same possess atomic a different(Z), number but aof different neutrons mass are number called isotopes. (A). The 3 isotopes of Hydrogen 27

ISOTOPES & ATOMIC MASS q The mass of an atom is measured relative to the mass of a chosen standard (carbon-12 atom), and is expressed in atomic mass units (amu). q The average atomic mass of an element is the mass of that element’s natural occurring isotopes weighted according to their abundance. q Therefore the atomic mass of an element is closest to the mass of its most abundant isotope. 28

4. 8 Isotopes: Natural Abundance Isotopes of neon Naturally occurring neon contains three different isotopes: Ne-20 (with 10 protons and 10 neutrons), Ne-21 (with 10 protons and 11 neutrons), and Ne-22 (with 10 protons and 12 neutrons).

Example 1: Determine the number of protons, electrons and neutrons in a chlorine atom. # of protons = 17 (Z) # of electrons = 17 (= p+) # of neutrons = 18 (35 - 17) 30

Example 2: Which two of the following are isotopes of each other? Isotopes of an element have the same atomic number, but a different mass number 31

Example 3: Based on the information below, which is the most abundant isotope of boron (atomic mass = 10. 8 amu) ? Isotope 10 B 11 B Mass (amu) 10. 0 11. 0 Atomic mass of an element is closer to the mass of the more abundant isotope 32

CALCULATING MASS FROM ISOTOPIC DATA Isotope Mass (amu) Abundance (%) 107 Ag 106. 91 51. 84 109 Ag 108. 90 48. 16 (106. 91) (0. 5184) = 55. 42 amu (108. 90) (0. 4816) = 52. 45 amu 107. 87 amu 33

Mass Number is Not the Same as Atomic Mass • the atomic mass is an experimental number determined from all naturally occurring isotopes • the mass number refers to the number of protons + neutrons in one isotope – natural or man-made

THE END 35
- Slides: 35