DME DME reacts to form alkenes DME adsorbs
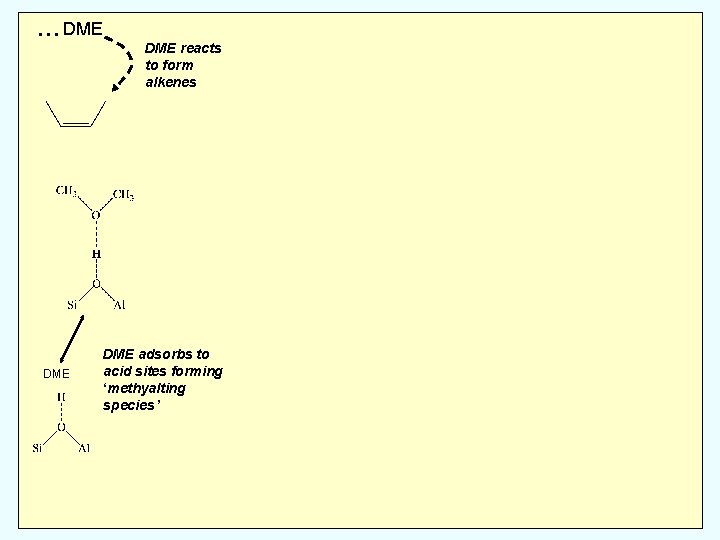
… DME DME reacts to form alkenes DME adsorbs to acid sites forming ‘methyalting species’
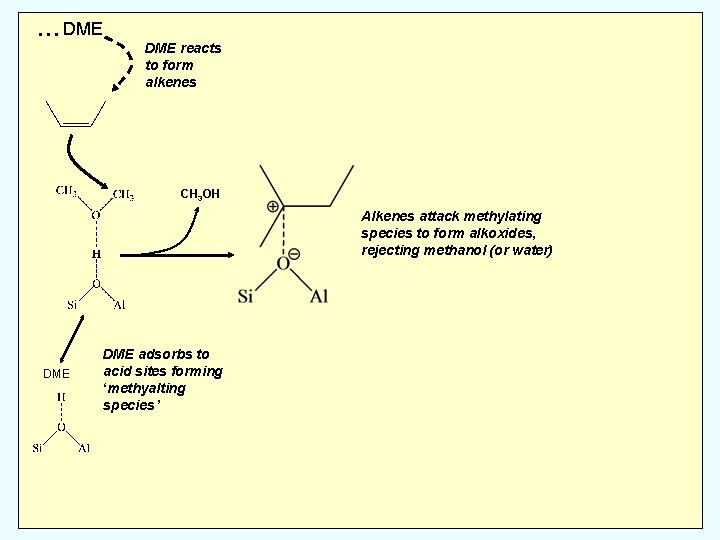
… DME reacts to form alkenes CH 3 OH Alkenes attack methylating species to form alkoxides, rejecting methanol (or water) DME adsorbs to acid sites forming ‘methyalting species’
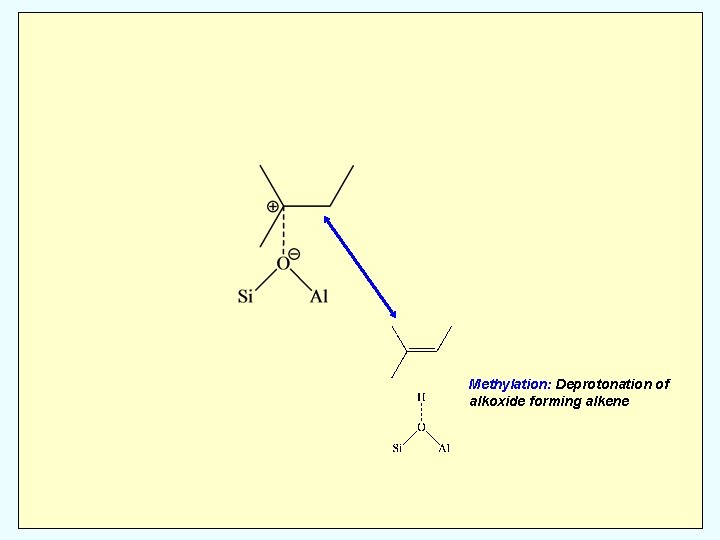
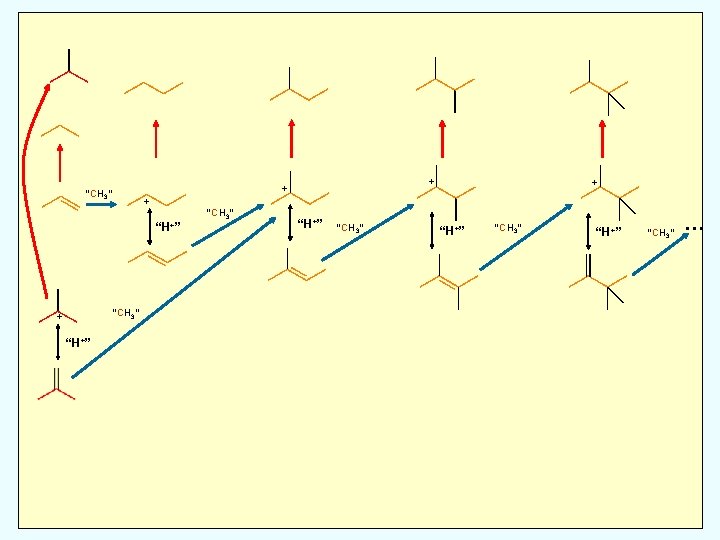
Methylation: Deprotonation of alkoxide forming alkene
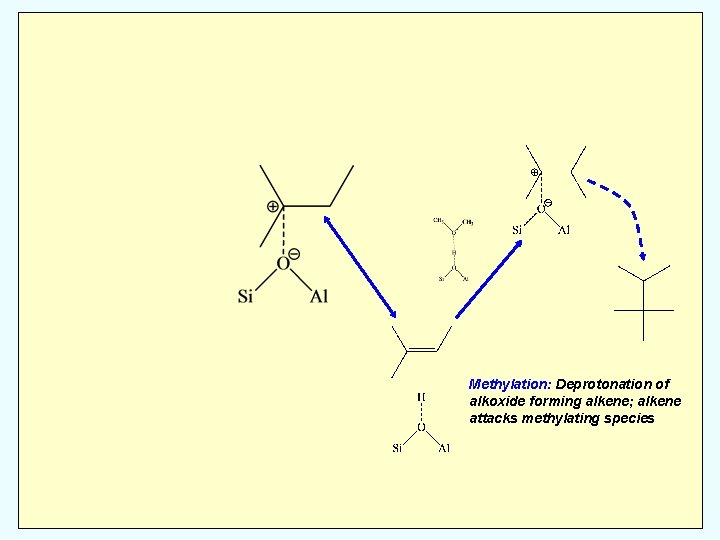
Methylation: Deprotonation of alkoxide forming alkene; alkene attacks methylating species
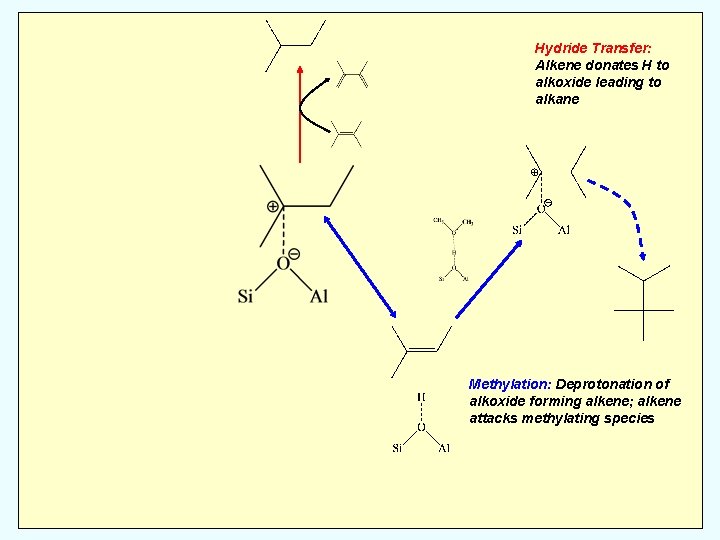
Hydride Transfer: Alkene donates H to alkoxide leading to alkane Methylation: Deprotonation of alkoxide forming alkene; alkene attacks methylating species
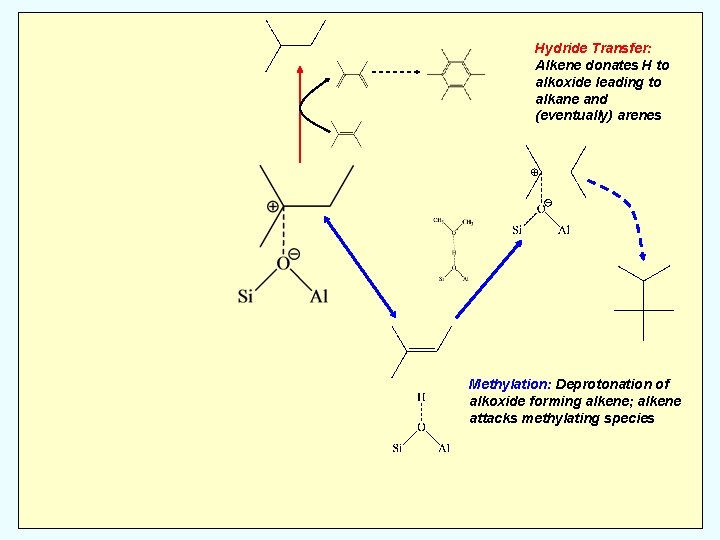
Hydride Transfer: Alkene donates H to alkoxide leading to alkane and (eventually) arenes Methylation: Deprotonation of alkoxide forming alkene; alkene attacks methylating species
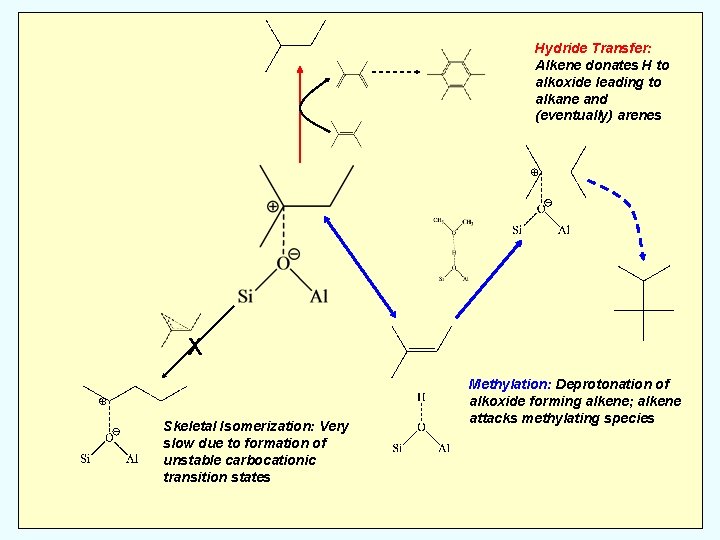
Hydride Transfer: Alkene donates H to alkoxide leading to alkane and (eventually) arenes X Skeletal Isomerization: Very slow due to formation of unstable carbocationic transition states Methylation: Deprotonation of alkoxide forming alkene; alkene attacks methylating species
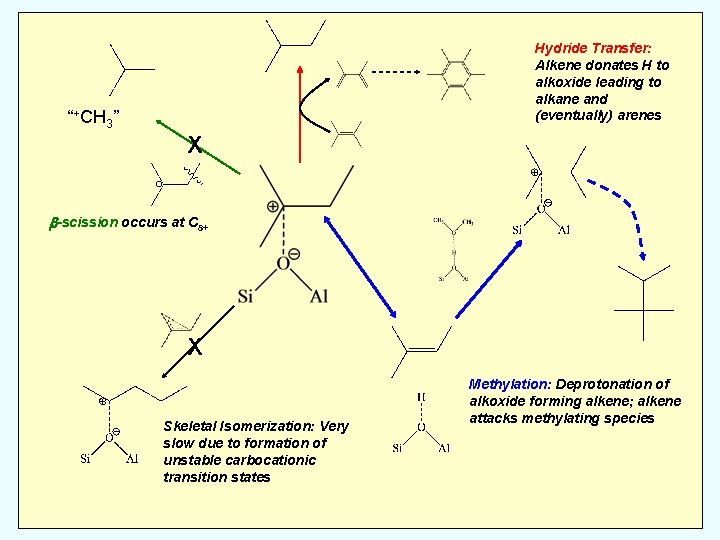
Hydride Transfer: Alkene donates H to alkoxide leading to alkane and (eventually) arenes “+CH 3” X b-scission occurs at C 8+ X Skeletal Isomerization: Very slow due to formation of unstable carbocationic transition states Methylation: Deprotonation of alkoxide forming alkene; alkene attacks methylating species
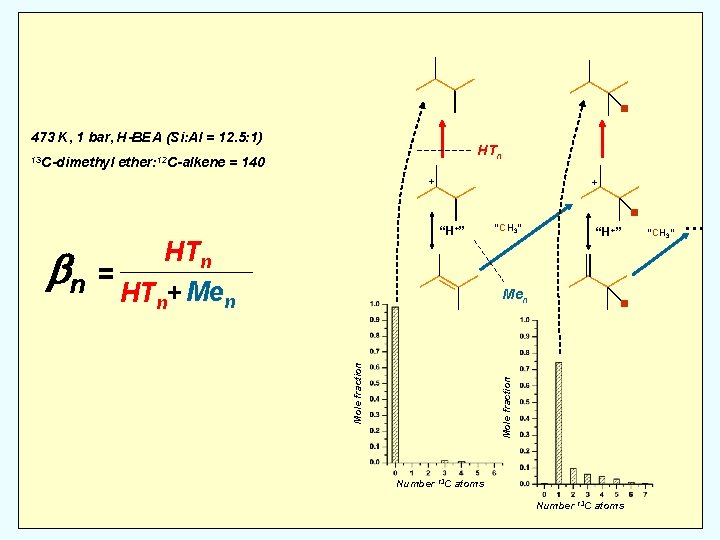
. 473 K, 1 bar, H-BEA (Si: Al = 12. 5: 1) 13 C-dimethyl HTn ether: 12 C-alkene = 140 + + “H+” HTn bn = HT + Me n n “CH 3” . Mole fraction Men “H+” . Number 13 C atoms “CH 3” …
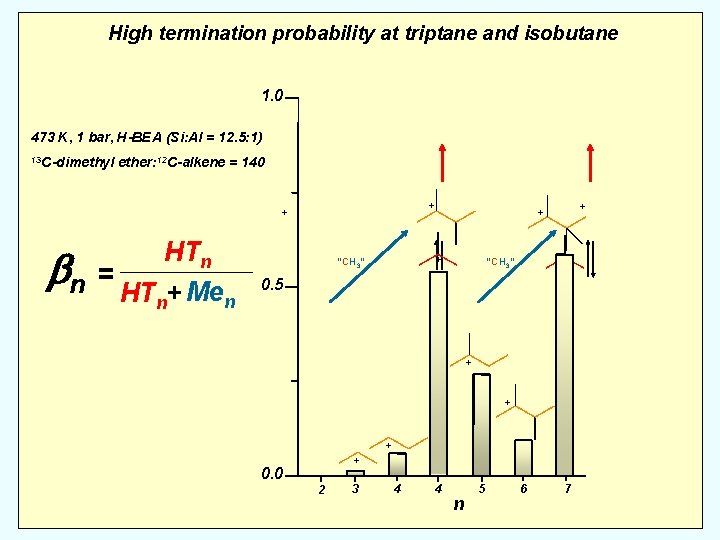
High termination probability at triptane and isobutane 1. 0 473 K, 1 bar, H-BEA (Si: Al = 12. 5: 1) 13 C-dimethyl ether: 12 C-alkene = 140 + + HTn bn = HT + Me n n + “CH 3” + + + “CH 3” 0. 5 + + 0. 0 2 3 4 4 n 5 6 7
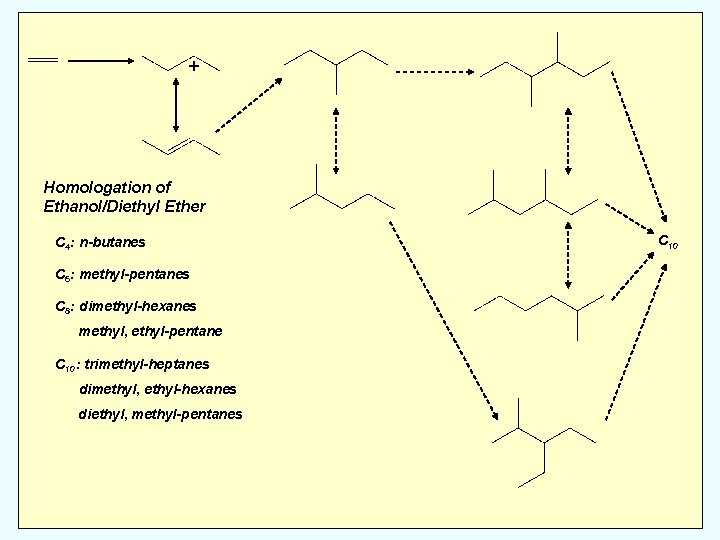
+ Homologation of Ethanol/Diethyl Ether C 4: n-butanes C 6: methyl-pentanes C 8: dimethyl-hexanes methyl, ethyl-pentane C 10: trimethyl-heptanes dimethyl, ethyl-hexanes diethyl, methyl-pentanes C 10
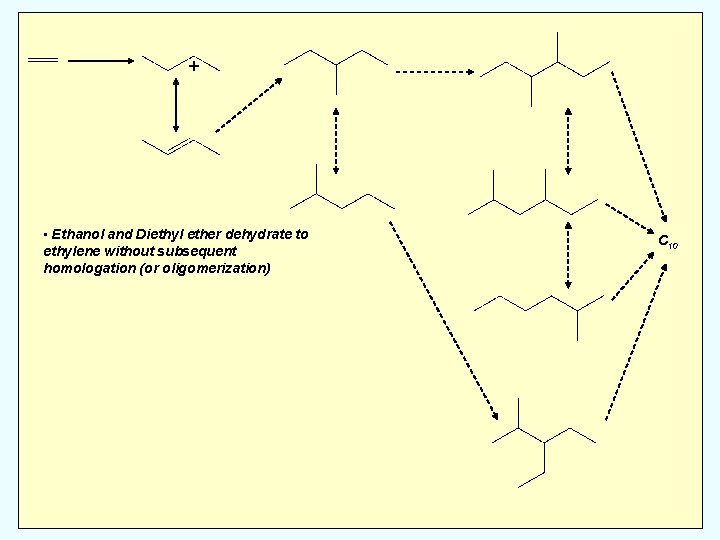
+ • Ethanol and Diethyl ether dehydrate to ethylene without subsequent homologation (or oligomerization) C 10
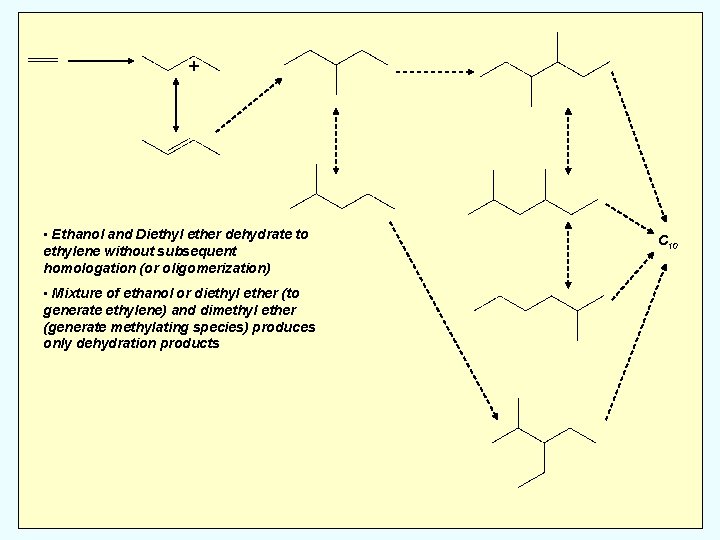
+ • Ethanol and Diethyl ether dehydrate to ethylene without subsequent homologation (or oligomerization) • Mixture of ethanol or diethyl ether (to generate ethylene) and dimethyl ether (generate methylating species) produces only dehydration products C 10

+ • Ethanol and Diethyl ether dehydrate to ethylene without subsequent homologation (or oligomerization) • Mixture of ethanol or diethyl ether (to generate ethylene) and dimethyl ether (generate methylating species) produces only dehydration products • Does dimethyl ether methylate ethylene? C 10

High termination probability at triptane and isobutane 1. 0 473 K, 1 bar, H-BEA (Si: Al = 12. 5: 1) 13 C-dimethyl ether: 12 C-alkene = 140 + + HTn bn = HT + Me n n + “CH 3” + + + “CH 3” 0. 5 + + 0. 0 2 3 4 4 n 5 6 7
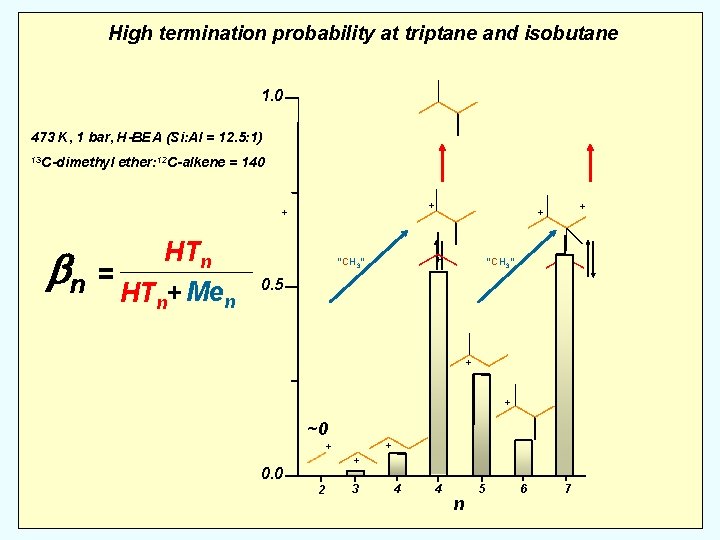
High termination probability at triptane and isobutane 1. 0 473 K, 1 bar, H-BEA (Si: Al = 12. 5: 1) 13 C-dimethyl ether: 12 C-alkene = 140 + + HTn bn = HT + Me n n + “CH 3” + + + “CH 3” 0. 5 + + ~0 + + + 0. 0 2 3 4 4 n 5 6 7
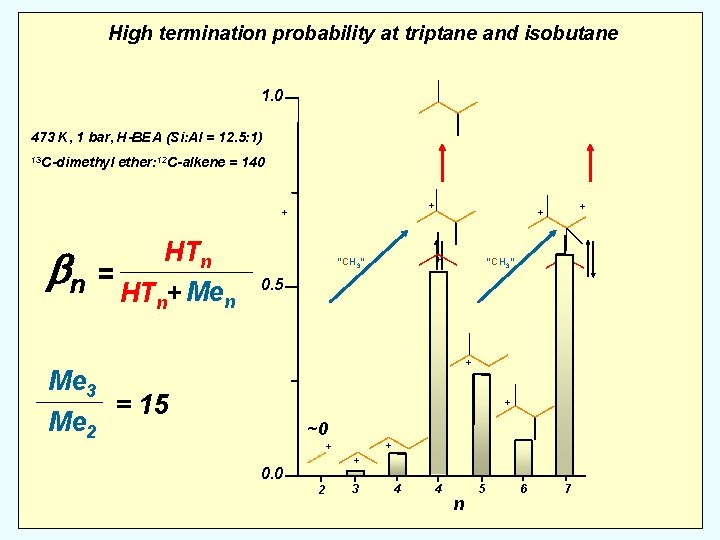
High termination probability at triptane and isobutane 1. 0 473 K, 1 bar, H-BEA (Si: Al = 12. 5: 1) 13 C-dimethyl ether: 12 C-alkene = 140 + + HTn bn = HT + Me n n Me 3 Me 2 + “CH 3” + + + “CH 3” 0. 5 + = 15 + ~0 + + + 0. 0 2 3 4 4 n 5 6 7

vs. Increasing alkene size and changing shape vs. Protonated oxygenate versus methyl species Alkene attacking alkoxide versus surface methylating species


• How does HT/M change with changing alkene size/shape? • Is there a difference in reactivity of surface methyl species and protonated DME molecules? • What is the difference between reaction of alkene with alkoxides and protonated ether molecules? (Is there something ‘special’ about C 1 surface groups? )
- Slides: 21