Distillation Distillation is a process of separating a














- Slides: 19

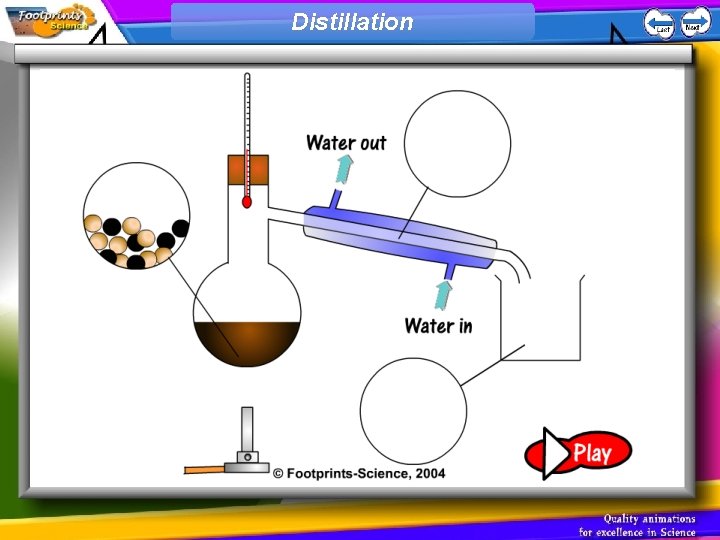
Distillation • Distillation is a process of separating a mixture of liquids by boiling it.

Crude Oil Formation, Composition, Purification and Properties

Formation of Oil 1. Crude oil is found in the Earth’s crust 2. It was formed from the remains of sea creatures that lived millions of years ago 3. It is a fossil fuel

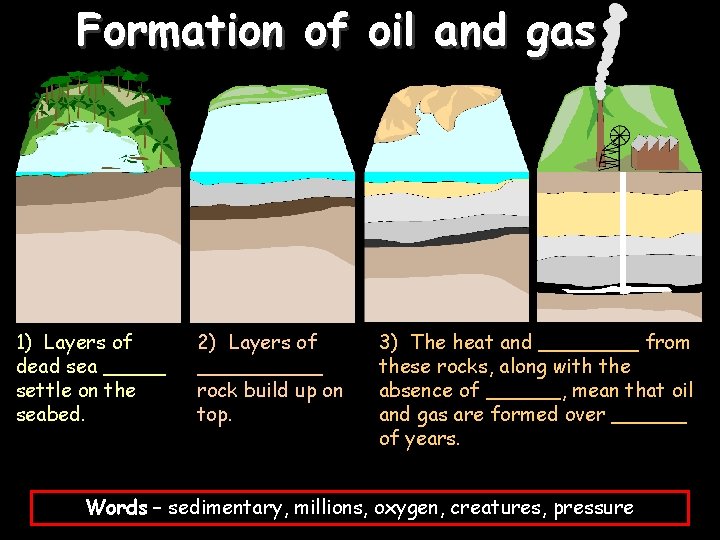
Formation of oil and gas 1) Layers of dead sea _____ settle on the seabed. 2) Layers of _____ rock build up on top. 3) The heat and ____ from these rocks, along with the absence of ______, mean that oil and gas are formed over ______ of years. Words – sedimentary, millions, oxygen, creatures, pressure

Formation of Oil

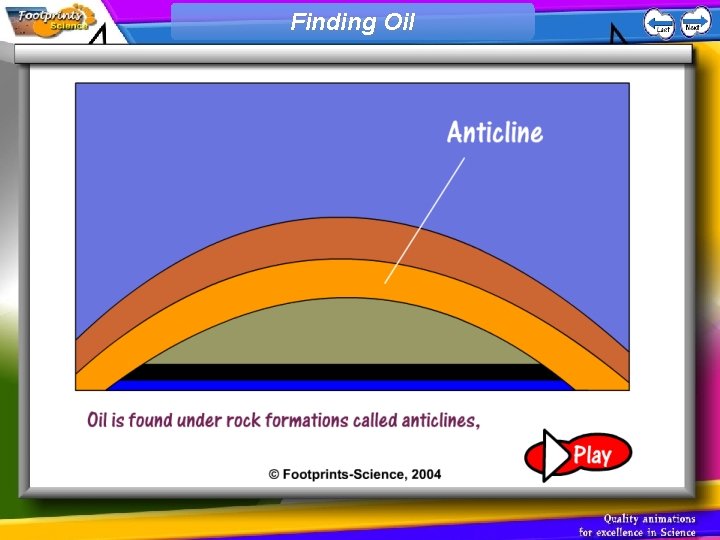
Finding Oil

Crude Oil 1. Crude oil contains a mixture of hydrocarbons 2. Hydrocarbons have different lengths and therefore different boiling points 3. They can be separated by fractional distillation 4. Each fraction is put to a different use depending on the properties of the hydrocarbon

Crude Oil

Distillation

Uses of hydrocarbons Most of the hydrocarbons present in crude oil can be used as fuels because they burn easily and give out a lot of heat energy. As the chain length increases they become less flammable and so purification is aimed to make simple mixtures containing similar molecules with similar behaviour.

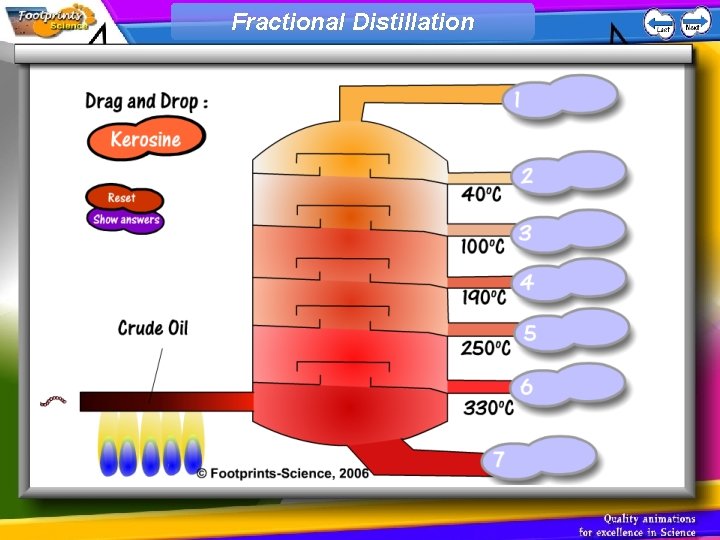
Fractional Distillation

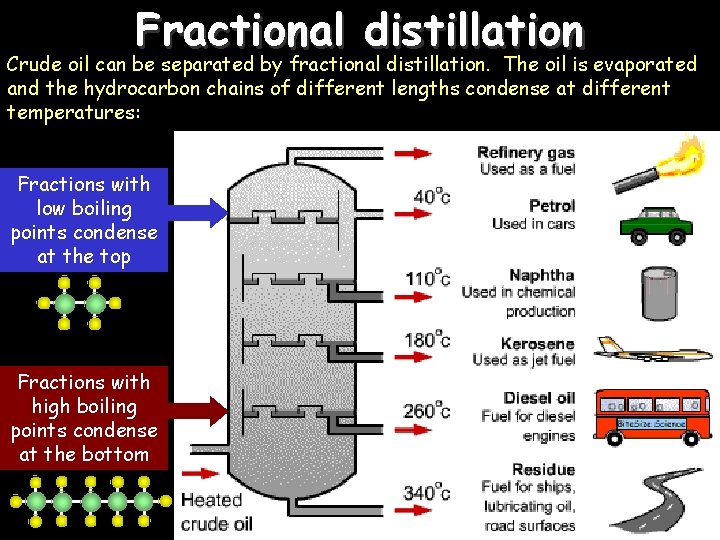
Fractional distillation Crude oil can be separated by fractional distillation. The oil is evaporated and the hydrocarbon chains of different lengths condense at different temperatures: Fractions with low boiling points condense at the top Fractions with high boiling points condense at the bottom

Hydrocarbons 1. A hydrocarbon is a molecule which contains hydrogen and carbon atoms only 2. The longer the carbon chain the; • Higher the boiling point • Less volatile it is • More viscous it is • Less flammable it is

Comparing hydrocarbons

Which is a correct statement about crude oil? A. B. C. D. A mixture of carbohydrates. Formed by the decay of dead sea creatures. Consist of a mixture of very large molecules. Is purified in an oil rig. © Boardworks Ltd 2003

Which is a correct statement about fractional distillation? A. Oil is separated into fractions with the same size molecule. B. Oil is separated into fractions with the same density. C. Oil is separated into fractions with similar size molecules. D. Oil is separated into alkanes and alkenes. © Boardworks Ltd 2003

Which is a correct order for these fractions working down from the top of the column? A. B. C. D. Fuel gas, kerosine, petrol, diesel, bitumen. Fuel gas, diesel, kerosine, petrol, bitumen. Fuel gas, petrol, diesel, kerosine, bitumen. Fuel gas, petrol, kerosine, diesel, bitumen. © Boardworks Ltd 2003

Which of these is a true statement about the changes that occur as hydrocarbon molecules get larger? A. Boiling point decreases. B. Viscosity increases. C. Flammability increases. D. Transparency increases. © Boardworks Ltd 2003

Problems caused by burning hydrocarbons 1. When a hydrocarbon burns carbon dioxide and water are made. 2. E. g. CH 4 + 3 O 2 → CO 2+ 2 H 2 O 3. Carbon dioxide adds to the greenhouse effect. 4. All fuels also contain sulphur impurities from the original sea creatures. When the sulphur burns in an engine sulphur dioxide is made. 5. Sulphur dioxide adds to acid rain