distillation boiling separate physically Matter Mixture differences or
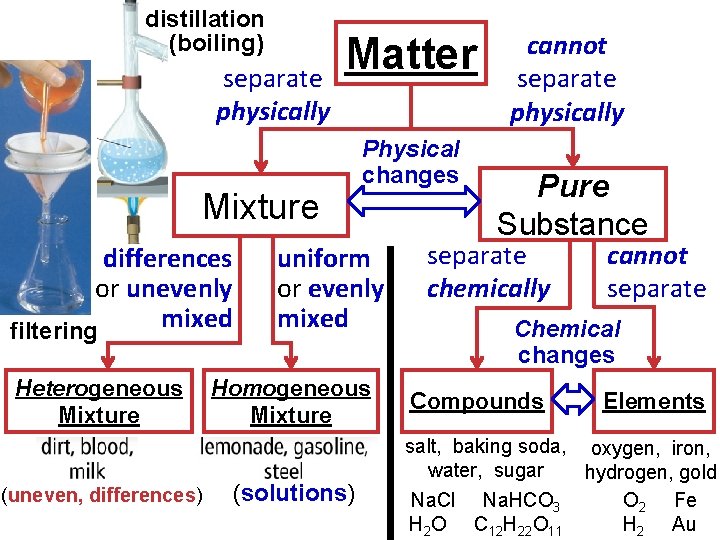
distillation (boiling) separate physically Matter Mixture differences or unevenly mixed filtering Heterogeneous Mixture (uneven, differences) Physical changes uniform or evenly mixed Homogeneous Mixture (solutions) cannot separate physically Pure Substance separate chemically cannot separate Chemical changes Compounds Elements salt, baking soda, oxygen, iron, water, sugar hydrogen, gold Na. Cl Na. HCO 3 O 2 Fe H 2 O C 12 H 22 O 11 H 2 Au

Pure Substances Element: (cannot be separated) (chemically or physically) -only one type of atom -simplest form of matter retaining its properties Examples: Carbon (C) Helium (He) Gold (Au)

Pure Substances Compound: (can be separated… chemically) different elements chemically bonded together Examples: Carbon Dioxide (CO 2) Water (H 2 O) Salt (Na. Cl) Sucrose (C 12 H 22 O 11)

completely new substance with completely new properties Na. Cl (compound) Na (element) Sodium is a highly reactive metal stored in oil. Cl 2 (element) Chlorine kills organisms in pools. It’s also a poisonous green gas.
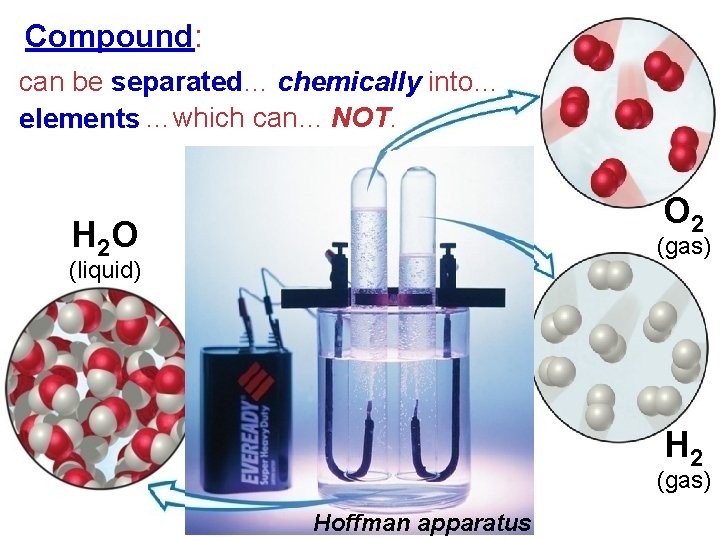
Compound: can be separated… chemically into… elements …which can… NOT. O 2 H 2 O (gas) (liquid) H 2 (gas) Hoffman apparatus
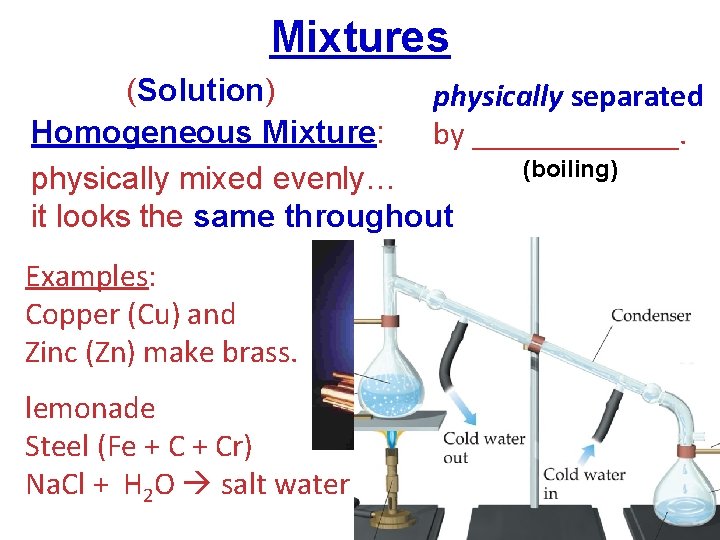
Mixtures (Solution) physically separated distillation Homogeneous Mixture: by _______. (boiling) physically mixed evenly… it looks the same throughout Examples: Copper (Cu) and Zinc (Zn) make brass. lemonade Steel (Fe + Cr) Na. Cl + H 2 O salt water

Mixtures Heterogeneous Mixture: physically mixed unevenly… can see the different parts Examples: Sand Granite Wood 2 Types: Suspensions & Colloids

Separation (Group Discussion) Sand + Iron (Fe) + Salt (Na. Cl) is a(n) heterogeneous mixture ___________ that can be separated by … Physically: -pulling out Fe with magnet Physically: -dissolving Na. Cl in water -filtering sand out -evaporating the water Chemically: -decomposing Na. Cl with electricity into Na and Cl

Quick Quiz! 1. Passing an electric current through a substance produces oxygen and sulfur. This substance cannot be a(n) A. compound. B. mixture. C. element. D. solution.

Quick Quiz. 2. Which of the following is a mixture? A. sodium chloride (Na. Cl) B. carbon dioxide (CO 2) C. sucrose (sugar, C 12 H 22 O 11) D. air (N 2, O 2, CO 2, H 2, Ar, …)

Quick Quiz. + 3. A blue-green solid is decomposed by heating. A colorless gas and a black solid appear. The blue-green solid must have been a(n) A. heterogeneous mixture B. homogeneous mixture C. compound D. element
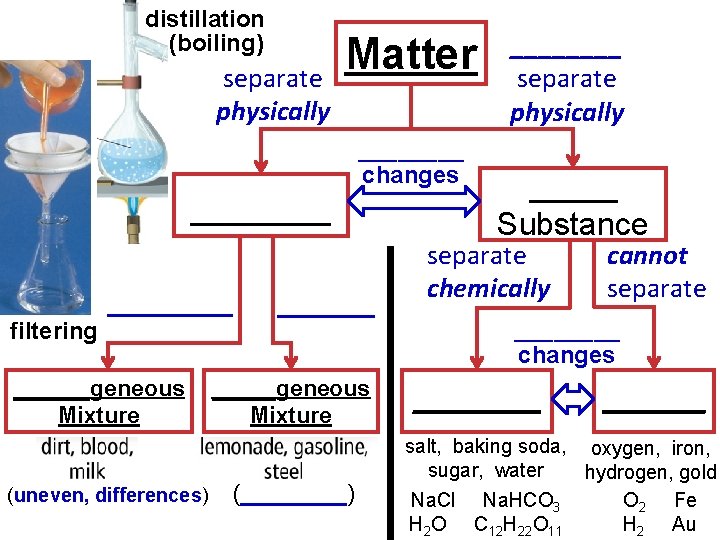
distillation (boiling) separate physically Matter _______ filtering ______geneous Mixture (uneven, differences) ____ changes _______geneous Mixture (____) ____ separate physically _____ Substance separate chemically cannot separate ____ changes _____ salt, baking soda, oxygen, iron, sugar, water hydrogen, gold Na. Cl Na. HCO 3 O 2 Fe H 2 O C 12 H 22 O 11 H 2 Au
- Slides: 12