DISSOLVED OXYGEN 101 Understanding the variables that affect

DISSOLVED OXYGEN 101 Understanding the variables that affect it, setting criteria for healthy water bodies and what to watch for in Lakes and Ponds

DISSOLVED OXYGEN 101 Understanding the variables that affect it, setting criteria for healthy water bodies and what to watch for in Lakes and Ponds Dissolved Oxygen is one of the most, if not the single most, important water quality variable affecting aquatic life. Fish kills are usually the result of low oxygen conditions in water bodies.

PRESENTATION TOPICS • Water – Its special properties • How fish take up oxygen • Solubility of Gases in Water • Air in contact with water • Solubility of Oxygen vs Temperature • Solubility of Oxygen vs Altitude • Dissolved Oxygen Criteria for a healthy fish population • Lakes and Ponds

WATER’S UNIQUE PROPERTIES Water has some unique and special properties, without which life on earth would not be possible.

WATER’S UNIQUE PROPERTIES Water has some unique and special properties, without which life on earth would not be possible. Ability to dissolve gasses: Water, the “universal solvent”. It dissolves more substances, including gasses, than any other liquid.

WATER’S UNIQUE PROPERTIES Water has some unique and special properties, without which life on earth would not be possible. Ability to dissolve gasses: Water, the “universal solvent”. It dissolves more substances, including gasses, than any other liquid. Ice Floats: At 4 ºC water molecules rearrange themselves causing the water to expand. So instead of becoming denser upon further cooling it becomes lighter. It is because of this unusual property that ice floats and lakes don’t freeze solid from top to bottom.

WATER’S UNIQUE PROPERTIES Water has some unique and special properties, without which life on earth would not be possible. Ability to dissolve gasses: Water, the “universal solvent”. It dissolves more substances, including gasses, than any other liquid. Ice Floats: At 4 ºC water molecules rearrange themselves causing the water to expand. So instead of becoming denser upon further cooling it becomes lighter. It is because of this unusual property that ice floats and lakes don’t freeze solid from top to bottom. At 4 ºC water is at its densest. This is why the temperature of water at the bottom of cold water lakes in the winter is around 4 ºC. The densest water will settle to the bottom - if it gets any colder or warmer, it will expand rise.

WATER – A POLAR MOLECULE It is the physical attributes of the water molecule that give it these special properties. Water molecules have a polar arrangement with one side of the molecule (oxygen) having a slight negative electrical charge and the other side (hydrogen) having a slight positive charge. Because of this they become attracted to many different types of molecules which make water a good polar solvent. Water molecules are also attracted to each other, which results in its high surface tension. At 4 ºC these polar molecules adjust themselves forcing the negatively charged oxygen atoms further apart and causing the water to expand become less dense.

HOW DISSOLVED OXYGEN IS MEASURED Dissolved Oxygen is usually measured as a concentration, mg/l or ppm But knowing Dissolved Oxygen concentration in the absence of other information is not very helpful. Dissolved Oxygen can be expressed in three ways: • As a Concentration: mg/l (or ppm) • As Pressure: mm. Hg • As % Saturation: This is a relative value expressing either concentration or pressure as a percentage of their respective saturation values. In determining which of these expressions is most appropriate for setting dissolved oxygen criteria for fish health, one needs to understand the mechanism by which fish utilize oxygen.
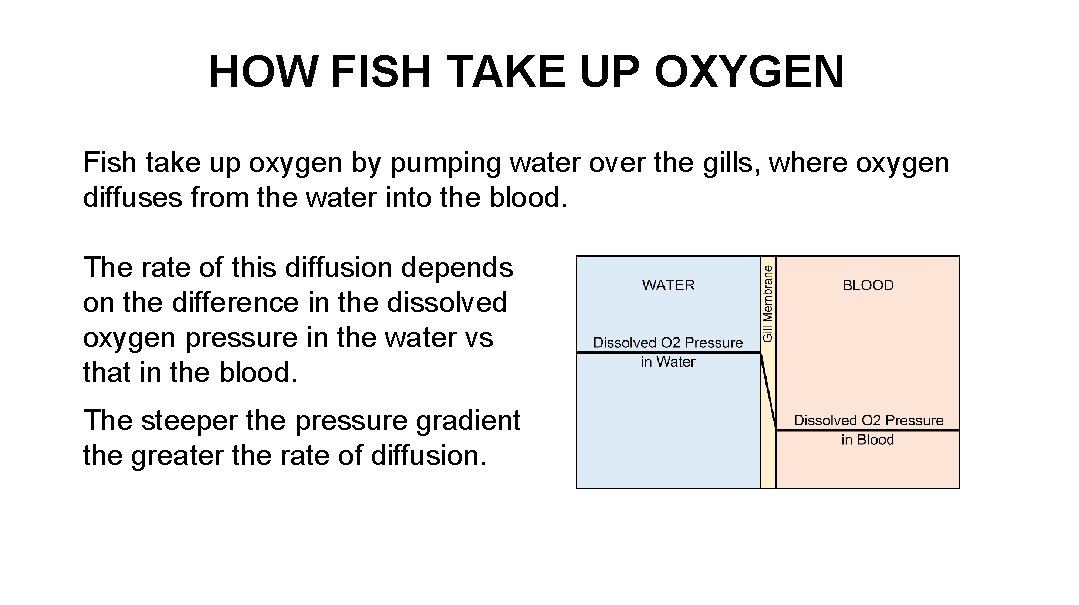
HOW FISH TAKE UP OXYGEN Fish take up oxygen by pumping water over the gills, where oxygen diffuses from the water into the blood. The rate of this diffusion depends on the difference in the dissolved oxygen pressure in the water vs that in the blood. The steeper the pressure gradient the greater the rate of diffusion.

HOW FISH TAKE UP OXYGEN Temperature, as will be explained, affects dissolved oxygen. Warm water holds less dissolved oxygen than cold water. Thus a fish in warm water must pump more water across its gills than in cold water in order to deliver the same amount of oxygen. This is necessary even though the pressure gradient between water and blood is the same. So both dissolved oxygen concentration and dissolved oxygen pressure play a roll.

SOLUBILITY OF GASES IN WATER: Solubility of gases in water is affected by three things: • PRESSURE (e. g. Altitude) • TEMPERATURE • SALINITY

SOLUBILITY OF GASES IN WATER: PRESSURE: The amount of gas that can be dissolved in water is proportional to the pressure of that gas in contact with the water - (Henry’s Law). The higher the pressure the greater the solubility.

SOLUBILITY OF GASES IN WATER: PRESSURE: The amount of gas that can be dissolved in water is proportional to the pressure of that gas in contact with the water - (Henry’s Law). The higher the pressure the greater the solubility. Water becomes saturated with a dissolved gas when the pressure of the dissolved gas, sometimes referred to as “gas tension”, is equal to the pressure of that gas in contact with the water.

SOLUBILITY OF GASES IN WATER: PRESSURE: The amount of gas that can be dissolved in water is proportional to the pressure of that gas in contact with the water - (Henry’s Law). The higher the pressure the greater the solubility. Water becomes saturated with a dissolved gas when the pressure of the dissolved gas, sometimes referred to as “gas tension”, is equal to the pressure of that gas in contact with the water. Gases moves back and forth across the gas liquid interface until pressure equilibrium is reached. If water is supersaturated with a gas, then the gas will want to escape, like when you pop the cap off a bottle of pop.

SOLUBILITY OF GASES IN WATER: PRESSURE vs CONCENTRATION: The actual concentration of the dissolved gas, e. g. mg/l or ppm depends on the solubility of the gas: Gases have different solubility’s: E. g. Oxygen is 2. 3 times more soluble than nitrogen. Carbon Dioxide is 40 times more soluble than oxygen. Concentration of Dissolved Gas = ß x Pressure of Gas in when saturated contact with water Where ß = Solubility Coefficient of the Gas

SOLUBILITY OF GASES IN WATER: Temperature and Salinity affect the solubility of a gas, i. e. the Solubility Coefficient changes with temperature and salinity. TEMPERATURE: Cold water can absorb more gas than hot water. The higher the water temperature the lower the solubility. SALINITY: Salinity affects solubility. The higher the salinity the lower the solubility. For example, oxygen solubility in Sea Water (35 ppt), is approximately 80% that of Fresh Water.

AIR IN CONTACT WITH WATER: Air is a mixture of gases. COMPOSITION OF AIR: Nitrogen (N 2) Oxygen (O 2)20. 94 % Argon (Ar) 0. 94 % Carbon Dioxide (CO 2) 0. 04 % 78. 08 %

AIR IN CONTACT WITH WATER: Air is a mixture of gases. COMPOSITION OF AIR: Nitrogen (N 2) 78. 08 % Oxygen (O 2) 20. 94 % Argon (Ar) 0. 94 % Carbon Dioxide (CO 2) 0. 04 % In order to determine the solubility of a constituent gas in air, you need to know its Partial Pressure. The partial pressure of a constituent gas in a mixture of gases is equal to its volume fraction multiplied by the total pressure of all the gases, which in the case of air, is the Barometric Pressure. Hence: Barometric Pressure, BP = PN 2 + PO 2 + PAr + PCO 2 So, partial pressure of oxygen, PO 2 = 20. 94/100 x BP NOTE: Water Vapour Pressure has been omitted from above for simplicity. It should be included in actual calculations
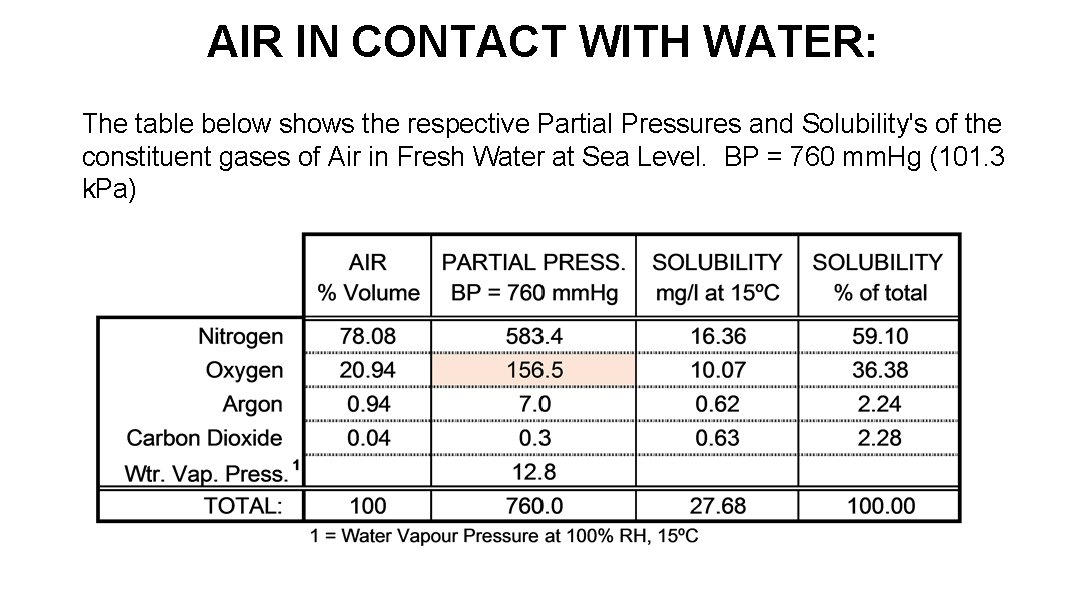
AIR IN CONTACT WITH WATER: The table below shows the respective Partial Pressures and Solubility's of the constituent gases of Air in Fresh Water at Sea Level. BP = 760 mm. Hg (101. 3 k. Pa)
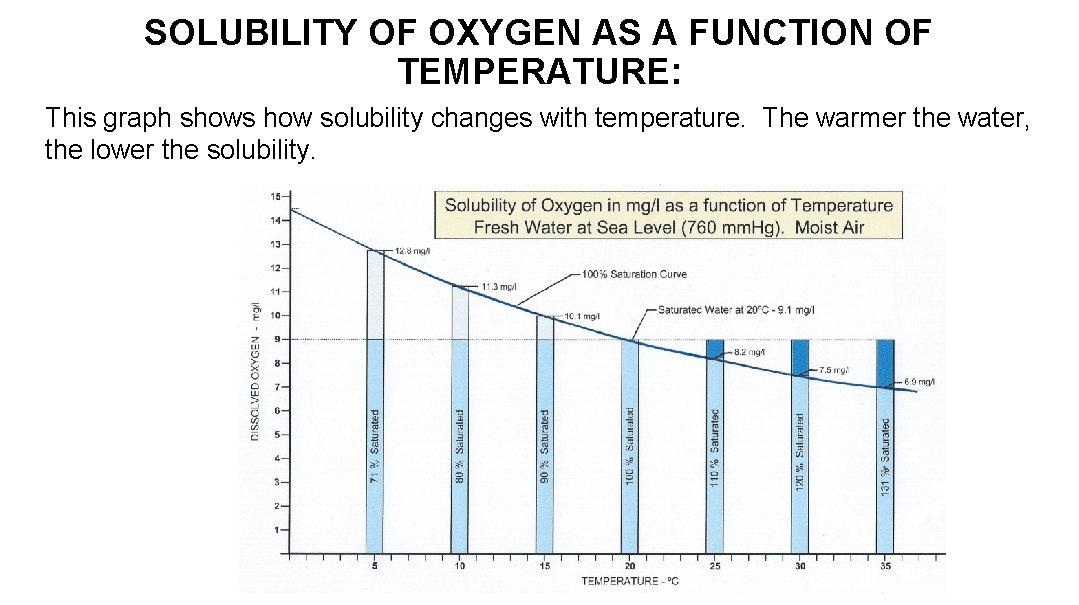
SOLUBILITY OF OXYGEN AS A FUNCTION OF TEMPERATURE: This graph shows how solubility changes with temperature. The warmer the water, the lower the solubility.
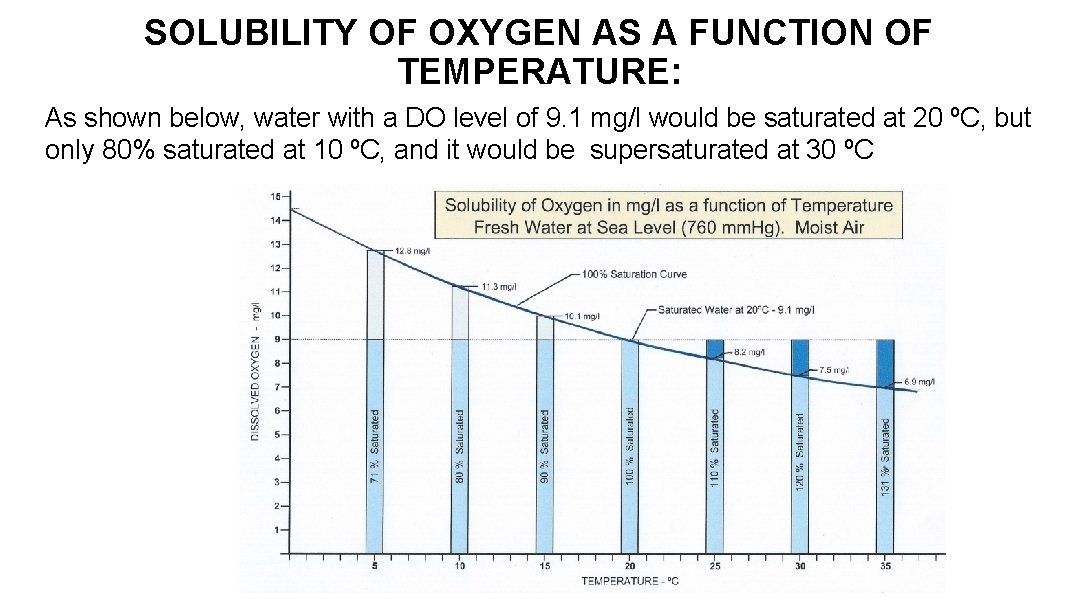
SOLUBILITY OF OXYGEN AS A FUNCTION OF TEMPERATURE: As shown below, water with a DO level of 9. 1 mg/l would be saturated at 20 ºC, but only 80% saturated at 10 ºC, and it would be supersaturated at 30 ºC

SOLUBILITY OF OXYGEN AS A FUNCTION OF TEMPERATURE: Streams can sometimes become supersaturated in summer due to heating from the sun. Water starts off cold and saturated with oxygen as it cascades down from the mountain. As it flows more gently across flatter ground it picks up heat from the sun becomes supersaturated. Dissolved oxygen concentration increases by approx. 2% per ºC rise in temperature.
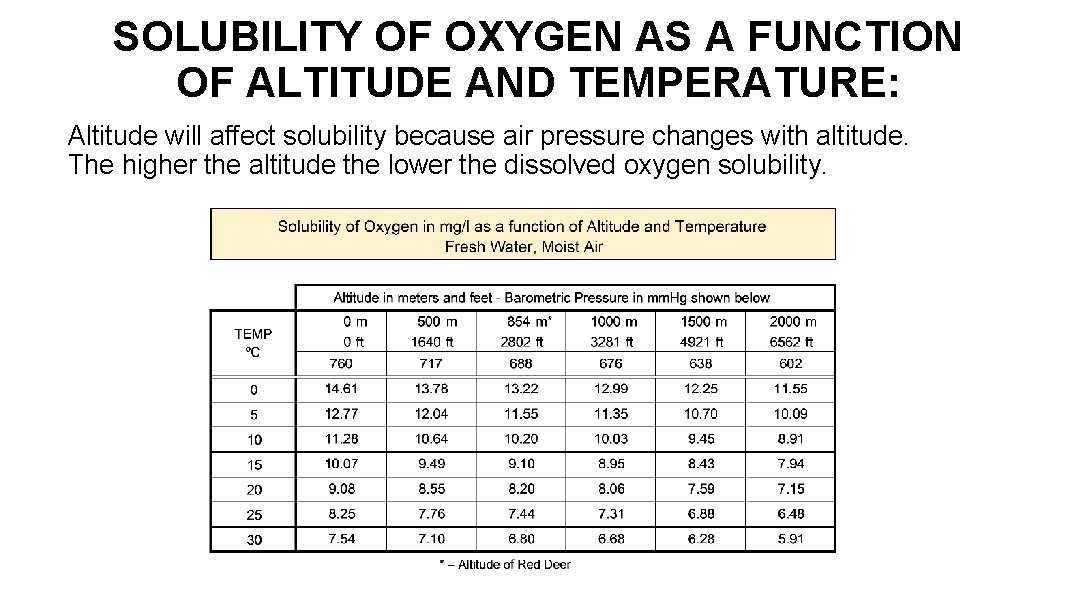
SOLUBILITY OF OXYGEN AS A FUNCTION OF ALTITUDE AND TEMPERATURE: Altitude will affect solubility because air pressure changes with altitude. The higher the altitude the lower the dissolved oxygen solubility.

SOLUBILITY OF OXYGEN AS A FUNCTION OF ALTITUDE AND TEMPERATURE: It is unlikely that the solubility tables you are using will give values for your altitude. Often oxygen solubility values are only given for standard barometric pressure i. e. average Barometric Pressure at sea level. If you know the oxygen solubility at a given barometric pressure, you can correct for the barometric pressure at your location as follows: O 2 Solubility at BP 2 = Solubility at BP 1 x BP 2/BP 1 To determine Barometric Pressure at a given altitude, or elevation, use the formula below: Log 10 BP = 2. 880814 - (H ÷ 19748. 2) Where H = Elevation in meters at your location. NOTE: The above does not take into account water vapour pressure, but it is a close enough approximation. To be more correct, water vapour pressure, if you know what it is, should be deducted from barometric pressure.

DISSOLVED OXYGEN CRITERIA FOR A HEALTHY FISH POPULATION In a paper "Minimal Dissolved Oxygen Requirements for Aquatic Life", John Davis (DFO 1975) established dissolved oxygen criteria based on incipient oxygen response thresholds expressed in terms of Po 2 (mm. Hg) as well as oxygen concentration (mg/l). ● In colder water, oxygen response thresholds are dictated by dissolved O 2 pressure, Po 2 to maintain the necessary pressure gradient. ● In warmer water, where the oxygen concentration is much lower than in colder water with the same Po 2, it is the mg/l value that determines the requirements for respiration.

DISSOLVED OXYGEN CRITERIA FOR A HEALTHY FISH POPULATION From statistical data collected, three levels of protection were derived: Level A This represents more or less ideal conditions and assures a high degree of safety for fish stocks. Level B This is the oxygen value where the average member of a fish population starts to exhibit symptoms of distress. Some degree of risk exists if the O 2 minimum is prolonged beyond a few hours. Level C At this level a large portion of a fish population will be affected and if prolonged beyond a few hours, the effects can be serious.

DISSOLVED OXYGEN CRITERIA FOR A HEALTHY FISH POPULATION By way of example, we will determine which of the two, mm. Hg or mg/l, will dictate dissolved oxygen criteria: Consider Protection Level A, mixed fresh water fish population, BP = 760 mm. Hg (sea level) Fresh Water at 10ºC: PO 2 = 157 mm. Hg DO Conc = 11. 3 mg/l mm. Hg as % sat. = 110/157 = 70% mg/l as % sat = 7. 25/11. 3 = 64% Therefore PO 2 determines DO level. Fresh Water at 20ºC: PO 2 = 157 mm. Hg DO Conc = 9. 1 mg/l mm. Hg as % sat. = 110/157 = 70% mg/l as % sat = 7. 25/9. 1 = 80% Therefore DO Conc. determines DO level.
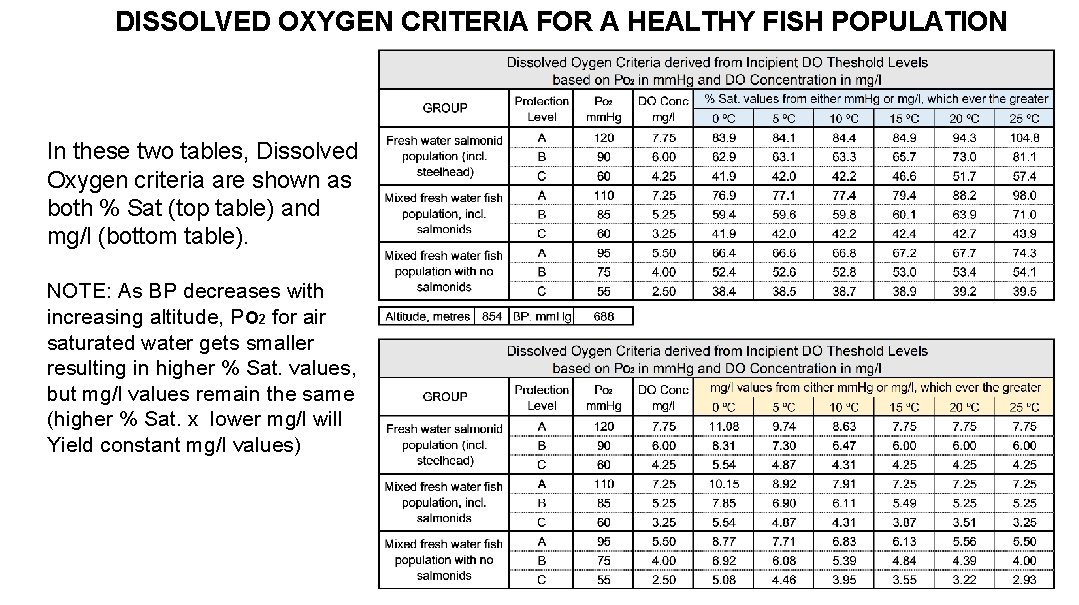
DISSOLVED OXYGEN CRITERIA FOR A HEALTHY FISH POPULATION In these two tables, Dissolved Oxygen criteria are shown as both % Sat (top table) and mg/l (bottom table). NOTE: As BP decreases with increasing altitude, PO 2 for air saturated water gets smaller resulting in higher % Sat. values, but mg/l values remain the same (higher % Sat. x lower mg/l will Yield constant mg/l values)

LAKES AND PONDS OXYGENATION OF LAKES AND PONDS: From Natural Aeration: Oxygen from the air gets absorbed at the surface of the water. Turbulence and wave action increases surface area, the greater the surface area the more oxygen is absorbed.

LAKES AND PONDS OXYGENATION OF LAKES AND PONDS: From Natural Aeration: Oxygen from the air gets absorbed at the surface of the water. Turbulence and wave action increases surface area, the greater the surface area the more oxygen is absorbed. From Photosynthesis of: Most oxygen comes from photosynthesis of aquatic plants, Aquatic Plants algae and phytoplankton. This only happens during daylight hours, at night aquatic plants consume oxygen. There is a diurnal fluctuation in oxygen levels: Rising during the day when O 2 from photosynthesis exceeds O 2 demand of aquatic life, then dropping during the night. On bright sunny days water can become super saturated with oxygen. Conversely on dark cloudy days oxygen levels can drop dangerously low at night.

LAKES AND PONDS CAUSES OF OXYGEN DEPLETION: Decomposition of: While healthy algae blooms and aquatic plants will produce enough Organic Matter O 2 to support aquatic life over 24 hour period, dying populations may consume more O 2 than they produce. This plus the decay of other organic matter can lead to dangerously low oxygen levels.

LAKES AND PONDS CAUSES OF OXYGEN DEPLETION: Decomposition of: While healthy algae blooms and aquatic plants will produce enough Organic Matter O 2 to support aquatic life over 24 hour period, dying populations may consume more O 2 than they produce. This plus the decay of other organic matter can lead to dangerously low oxygen levels. Eutrophication: Discharge of phosphates and other nutrients from fertilizers, livestock and sewage induces explosive growth of algae and plants, the decaying of which depletes dissolved oxygen.

LAKES AND PONDS CAUSES OF OXYGEN DEPLETION: Decomposition of: While healthy algae blooms and aquatic plants will produce enough Organic Matter O 2 to support aquatic life over 24 hour period, dying populations may consume more O 2 than they produce. This plus the decay of other organic matter can lead to dangerously low oxygen levels. Eutrophication: Discharge of phosphates and other nutrients from fertilizers, livestock and sewage induces explosive growth of algae and plants, the decaying of which depletes dissolved oxygen. Ice & Snow cover: Prolonged ice and snow cover can be a problem. Not only does the water not have access to air, snow covered ice prevents adequate sunlight penetration for photosynthesis. This together with decaying organic matter may lead to Winter Kill. A preventive measure is to partially clear snow in long strips to allow sunlight to penetrate, or use an aerator(s) to keep an open area of water.

LAKES AND PONDS CAUSES OF OXYGEN DEPLETION: Lake Stratification and Turnover: Epilimnion: Upper warm layer in contact with the air higher in dissolved oxygen. Thermocline: Thin distinct layer in which temperature changes rapidly with depth. Hypolimnion: Cold bottom layer deficient in oxygen due to reduced sunlight and decaying bottom sediment. Seasonal lake turnover results in the oxygen deficient bottom layer coming to the surface and mixing with the surface water, sometimes resulting in the entire lake or pond becoming oxygen deficient.
- Slides: 35