Disrupt CAD Study Design Stable angina unstable angina

Disrupt CAD Study Design Stable angina, unstable angina or silent ischemia Moderate and severely calcified, de novo coronary lesions RVD 2. 5 – 4. 0 mm, stenosis ≥ 50%, Lesion length ≤ 32 mm 60 patients enrolled 31 subject OCT sub-study 30 day & 6 months follow-up Core Angiographic & OCT Labs (Yale University & CRF) • • • Objective: To assess the safety and performance of the Lithoplasty® System Primary Safety Endpoint: 30 -day MACE (Cardiac death, MI or TVR) Primary Performance Endpoint: Clinical success (residual stenosis <50%) post-PCI with no evidence of in-hospital MACE.
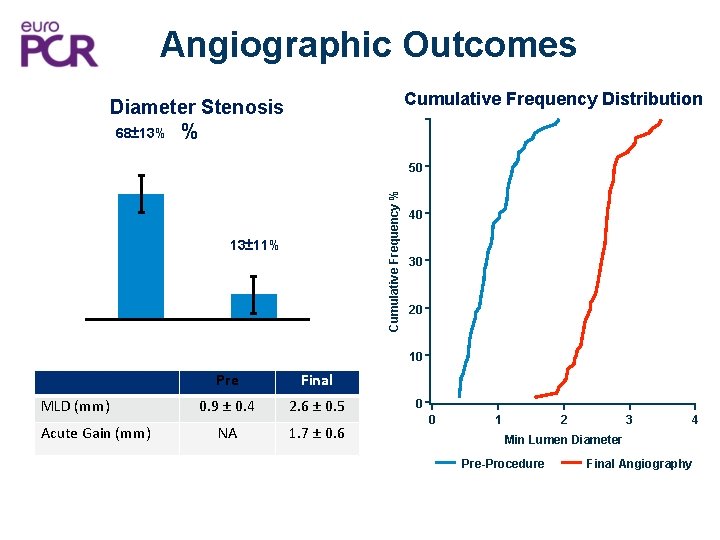
Angiographic Outcomes Cumulative Frequency Distribution Diameter Stenosis 68± 13% % Cumulative Frequency % 50 13± 11% 40 30 20 10 MLD (mm) Acute Gain (mm) Pre Final 0. 9 ± 0. 4 2. 6 ± 0. 5 NA 1. 7 ± 0. 6 0 0 2 1 3 4 Min Lumen Diameter Pre-Procedure Final Angiography
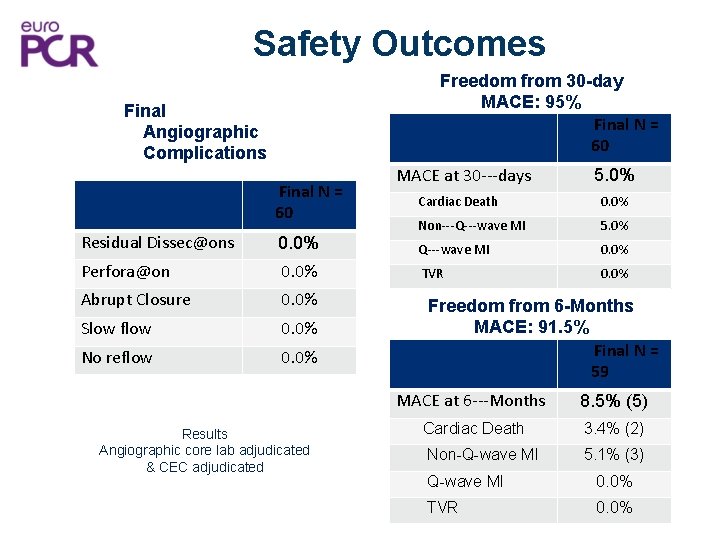
Safety Outcomes Freedom from 30 -day MACE: 95% Final N = 60 Final Angiographic Complications Final N = 60 Residual Dissec@ons 0. 0% Perfora@on 0. 0% Abrupt Closure 0. 0% Slow flow 0. 0% No reflow 0. 0% Results Angiographic core lab adjudicated & CEC adjudicated MACE at 30‐‐‐days 5. 0% Cardiac Death 0. 0% Non‐‐‐Q‐‐‐wave MI 5. 0% Q‐‐‐wave MI 0. 0% TVR 0. 0% Freedom from 6 -Months MACE: 91. 5% Final N = 59 MACE at 6‐‐‐Months 8. 5% (5) Cardiac Death 3. 4% (2) Non-Q-wave MI 5. 1% (3) Q-wave MI 0. 0% TVR 0. 0%

Conclusions • Excellent Lithoplasty balloon performance with 100% stent delivery. • High acute gain and low residual stenosis by core lab. • Low CEC adjudicated 30 -day MACE rate of 5. 0% with minimal vascular complications. • OCT analysis demonstrated heavy calcium burden with substantial luminal acute gain in all groups. • Circumferential calcium fracture confirmed mechanim for lithoplasty validated by OCT analysis. The Shockwave Medical Coronary Lithoplasty System is investigational and is not available for sale.
- Slides: 4