Displacement reactions AcidBase reactions recombine ions H transfer

Displacement reactions Acid-Base reactions recombine ions H+ transfer Fe 2 O 3 (s) + Al (s) Al 2 O 3 (s) + Fe(l) thermite reaction Oxidation-Reduction reactions (redox) e- transfer Fe 2 O 3 = iron (III )oxide Fe 3+ Fe (l) = Fe 0 26 protons Fe 0 has _____ Fe 3+ has_____ 26 protons Fe 3+ has ____ 23 electrons 26 electrons Fe 0 has ____ Fe 3+ gained 3 electrons reducing the oxidation state gaining e- = reduction 3+ 0

Fe 2 O 3 (s) + Al (s) Al 2 O 3 (s) + Fe(l) Al 2 O 3 aluminum oxide Al (s) = Al 0 has _____ 13 electrons oxidation state = 0 Al 0 loses___ 3 elosing e- = oxidation LEO GER 10 electrons Al 3+has ______ oxidation state = 3+ increase oxidation number 0 3+ Lose Electrons Oxidation Gain Electrons Reduction

Rules for oxidation numbers 1. Oxidation number for elements is zero. N 2, O 2, Na(s), Co(s), He (g) 2. Oxidation number of monatomic ions is the same as their charge Group IA = +1 Al 3+, Zn 2+, Cd 2+, Ag+ Group IIA = +2 3. Oxidation number of oxygen in most compounds is – 2. Exceptions: H 2 O 2, O 2 - (peroxides) – 1 4. Oxidation number of hydrogen is +1 Exceptions: bonded to metals Li. H 5. Fluorine is always – 1. Other halogens are – 1. Exceptions: bonded to O, they are positive

Assign oxidation numbers to all of the elements: Li 2 O Li = +1 O = -2 PF 3 P = +3 F = -1 HNO 3 H = +1 N = +5 Mn. O 4 - Mn = +7 O = -2 Cr 2 O 72 - Cr = +6 O = -2

What is the oxidation state of the highlighted element? P 2 O 5 +5 diphosphorous pentoxide Na. H -1 sodium hydride Sn. Br 4 +4 tin (IV) bromide Ba. O 2 -1 barium peroxide

Redox reactions 0 +1 0 +2 Zn (s) + 2 H+ (aq) H 2 (g) + Zn 2+ Zn 0 +2 increase oxidation state lose electrons oxidized reducing agent H +1 0 decrease oxidation state gain electrons reduced oxidizing agent

0 0 +1 -2 2 H 2(g) + O 2 (g) 2 H 2 O(l) H loses or gains electrons is oxidized or reduced oxidizing or reducing agent O loses or gains electrons is oxidized or reduced oxidizing or reducing agent
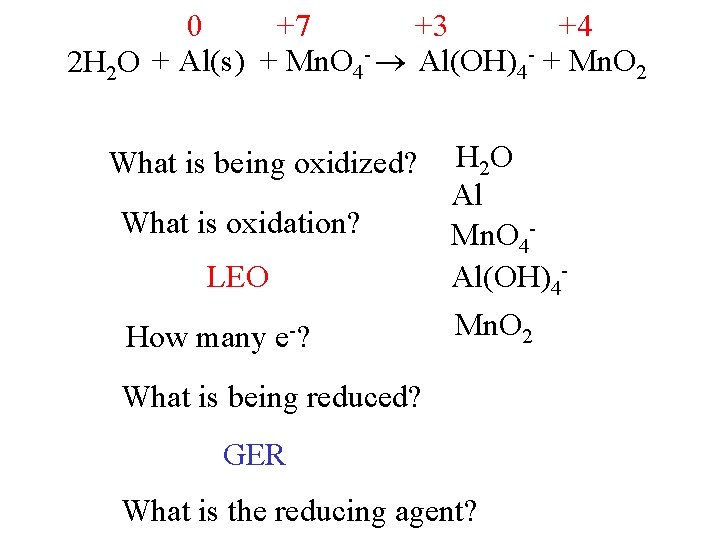
0 +7 +3 +4 2 H 2 O + Al(s) + Mn. O 4 - Al(OH)4 - + Mn. O 2 What is being oxidized? What is oxidation? LEO How many e-? H 2 O Al Mn. O 4 Al(OH)4 Mn. O 2 What is being reduced? GER What is the reducing agent?

What is the oxidation state of S in each of these compounds: H 2 S S = -2 H = +1 S 8 S=0 SCl 2 S = +2 Cl = -1 Na 2 SO 3 S = +4 O = -2 SO 42 - Na = +1 S = +6 O = -2 Which of these compounds can not act as a reducing agent? Which of these compounds can not act as a oxidizing agent?

0 +4 -2 +1 -2 +2 -2 +1 Cd(s) + Ni. O 2 + 2 H 2 O Cd(OH)2 + Ni(OH)2 What is being oxidized? What is being reduced? What is the oxidizing agent? What is the reducing agent? How many electrons are transferred?
- Slides: 10