Dispersed Fluorescence Spectroscopy of the B 2 E

Dispersed Fluorescence Spectroscopy of ~ ~ the B 2 E’ – X 2 A 2’ transition of NO 3 Masaru Fukushima and Takashi Ishiwata Graduate School of Information Sciences, Hiroshima City University, Hiroshima 731 -3194, Japan
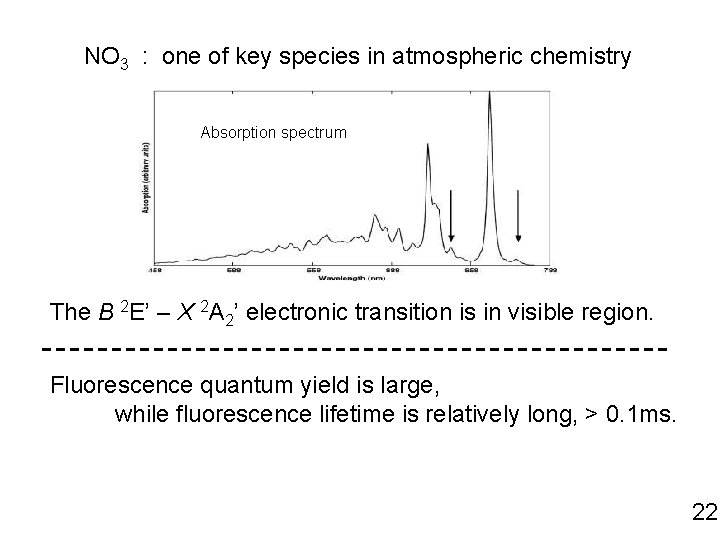
NO 3 : one of key species in atmospheric chemistry Absorption spectrum The B 2 E’ – X 2 A 2’ electronic transition is in visible region. Fluorescence quantum yield is large, while fluorescence lifetime is relatively long, > 0. 1 ms. 22

One of the major points on NO 3 Assignment I ( traditional ) 1, 492 cm-1 vib. band : n 3 fundamental Assignment II ( proposed by John Stanton at 2007 ) 1, 492 cm-1 vib. band : n 3 + n 4 combination This work Dispersed fluorescence spectra from single vibronic levels (1) Vibronic band to n 3 with ~1, 000 cm-1 has not been observed. (2) It is unusual that the n 3 band has negligible intensity while n 3 + n 4 has remarkable intensity. (3) SVL DF spectra show that the 1, 492 cm-1 level has no contribution of the n 4 mode. SVL DF spectroscopy holds Assignment I. 21
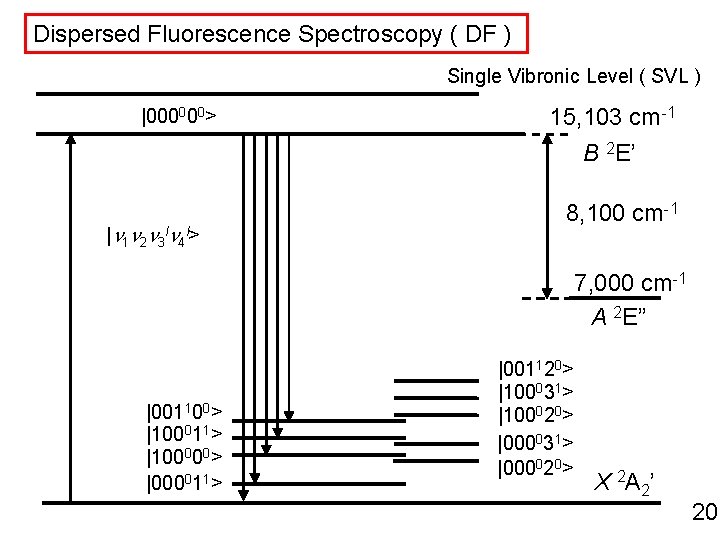
Dispersed Fluorescence Spectroscopy ( DF ) Single Vibronic Level ( SVL ) |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> |001120> |100031> |100020> |000031> |000020> X 2 A 2’ 20
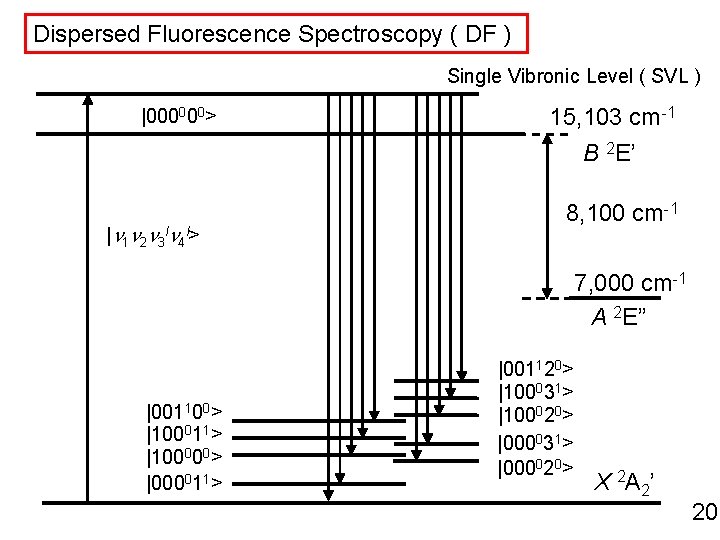
Dispersed Fluorescence Spectroscopy ( DF ) Single Vibronic Level ( SVL ) |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> |001120> |100031> |100020> |000031> |000020> X 2 A 2’ 20
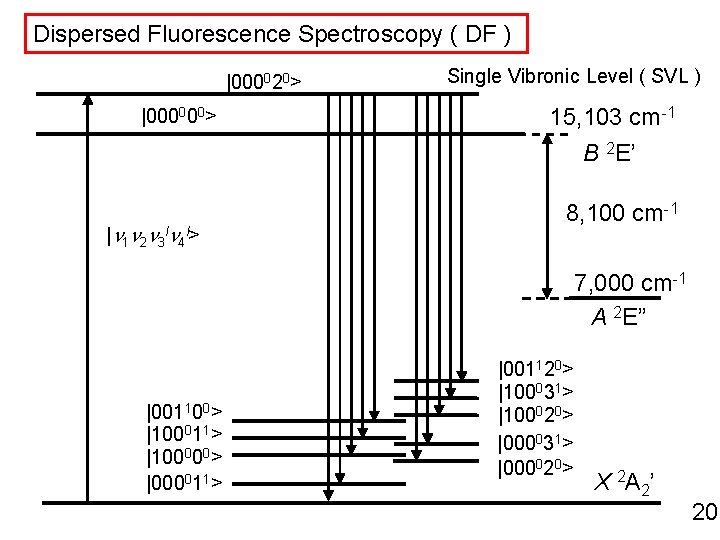
Dispersed Fluorescence Spectroscopy ( DF ) |000020> |000000> Single Vibronic Level ( SVL ) 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> |001120> |100031> |100020> |000031> |000020> X 2 A 2’ 20
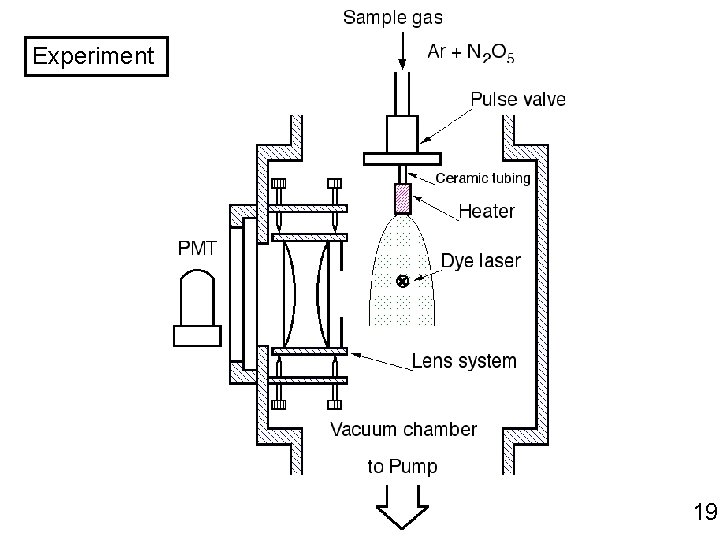
Experiment 19
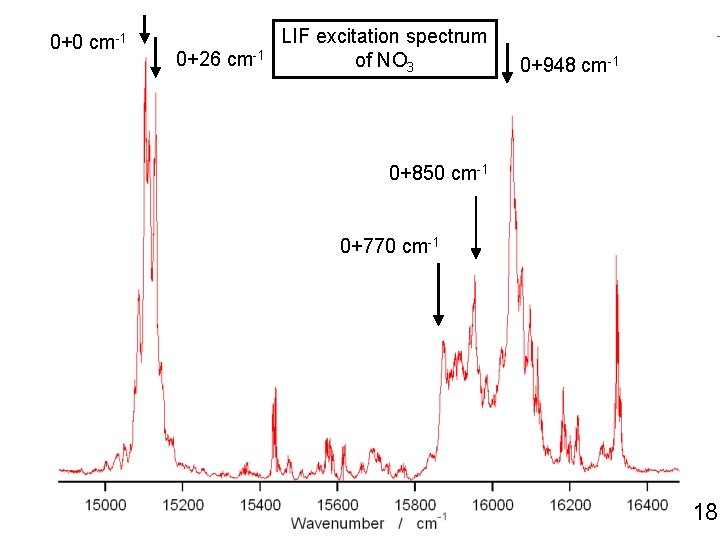
0+0 cm-1 0+26 cm-1 LIF excitation spectrum of NO 3 0+948 cm-1 0+850 cm-1 0+770 cm-1 18
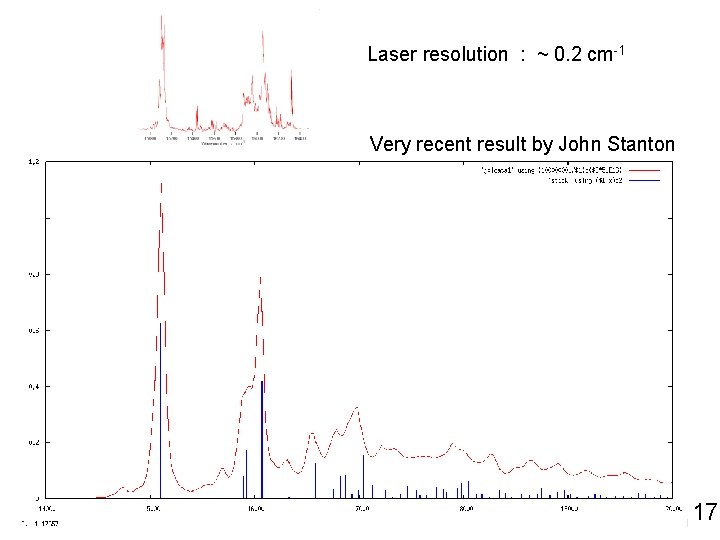
Laser resolution : ~ 0. 2 cm-1 Very recent result by John Stanton 17
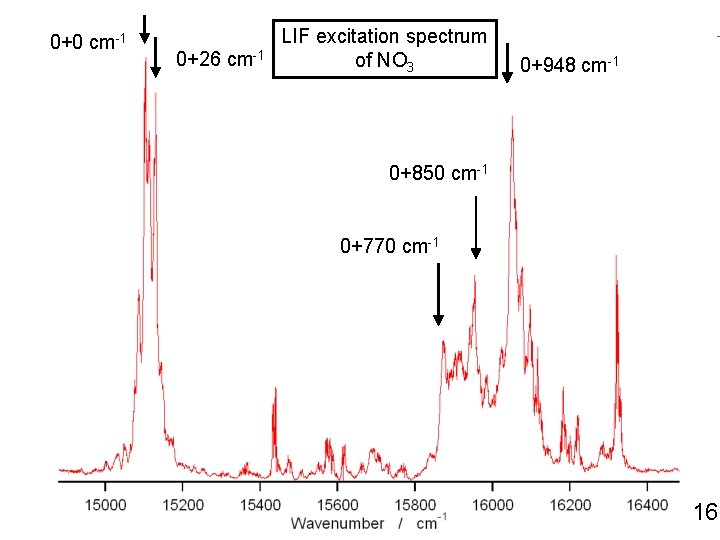
0+0 cm-1 0+26 cm-1 LIF excitation spectrum of NO 3 0+948 cm-1 0+850 cm-1 0+770 cm-1 16
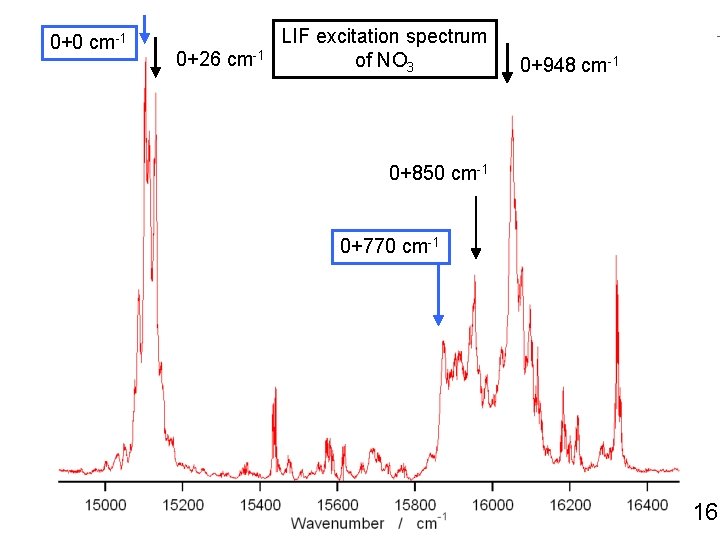
0+0 cm-1 0+26 cm-1 LIF excitation spectrum of NO 3 0+948 cm-1 0+850 cm-1 0+770 cm-1 16
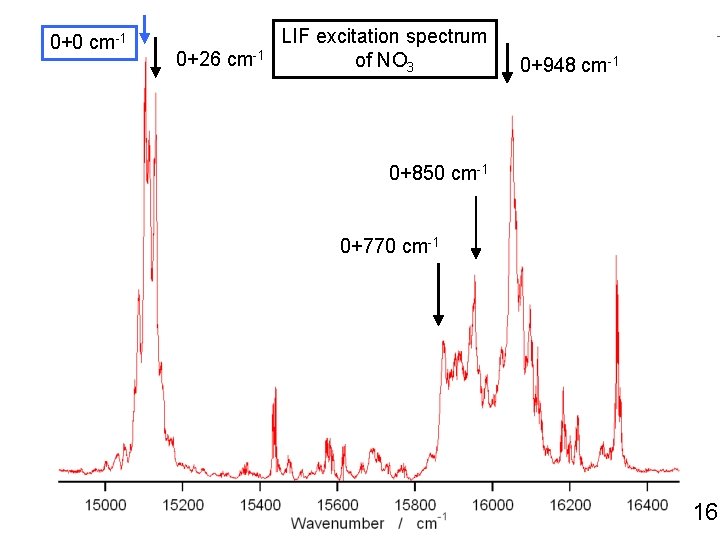
0+0 cm-1 0+26 cm-1 LIF excitation spectrum of NO 3 0+948 cm-1 0+850 cm-1 0+770 cm-1 16
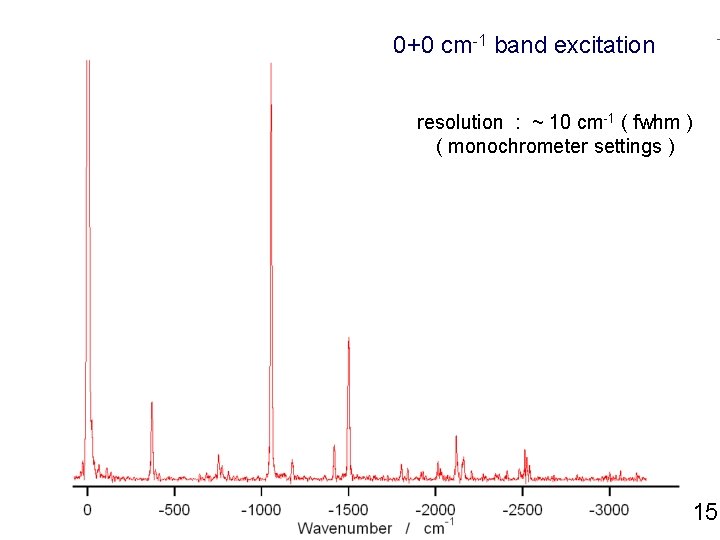
0+0 cm-1 band excitation resolution : ~ 10 cm-1 ( fwhm ) ( monochrometer settings ) 15
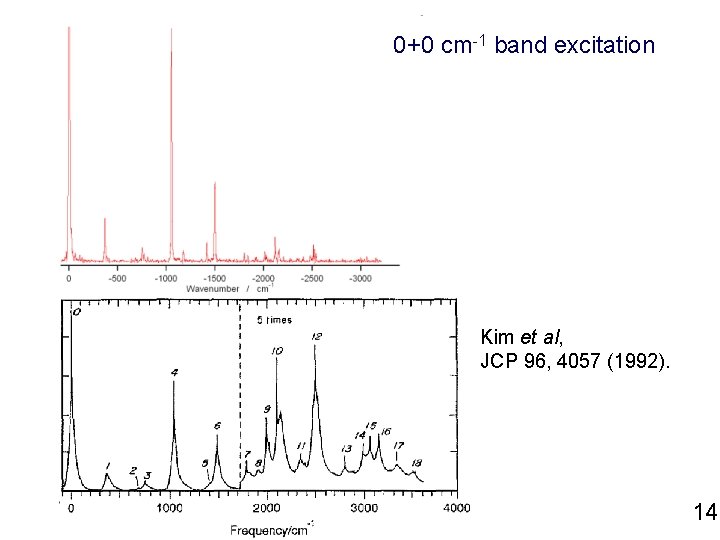
0+0 cm-1 band excitation Kim et al, JCP 96, 4057 (1992). 14
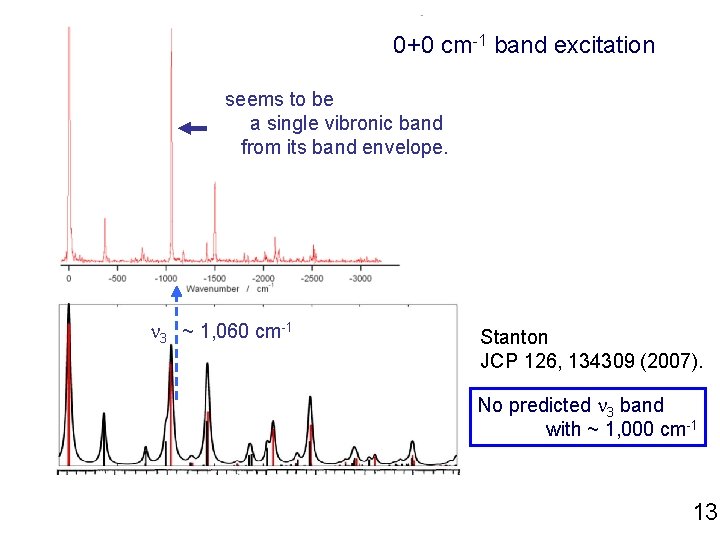
0+0 cm-1 band excitation seems to be a single vibronic band from its band envelope. n 3 ~ 1, 060 cm-1 Stanton JCP 126, 134309 (2007). No predicted n 3 band with ~ 1, 000 cm-1 13
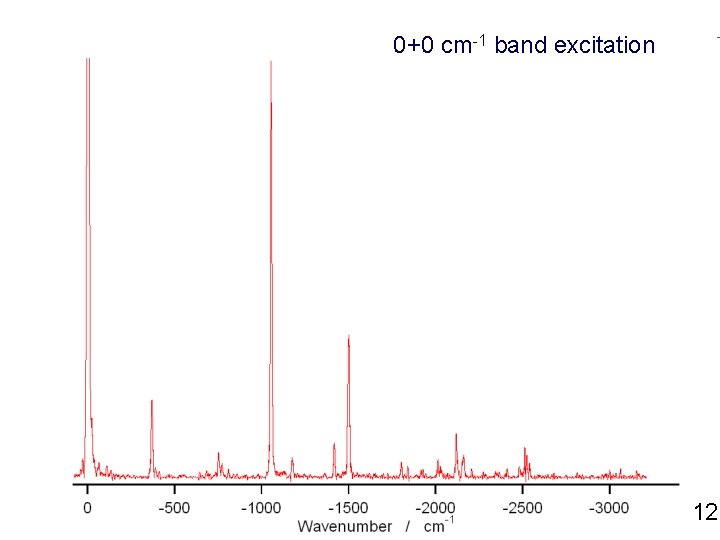
0+0 cm-1 band excitation 12
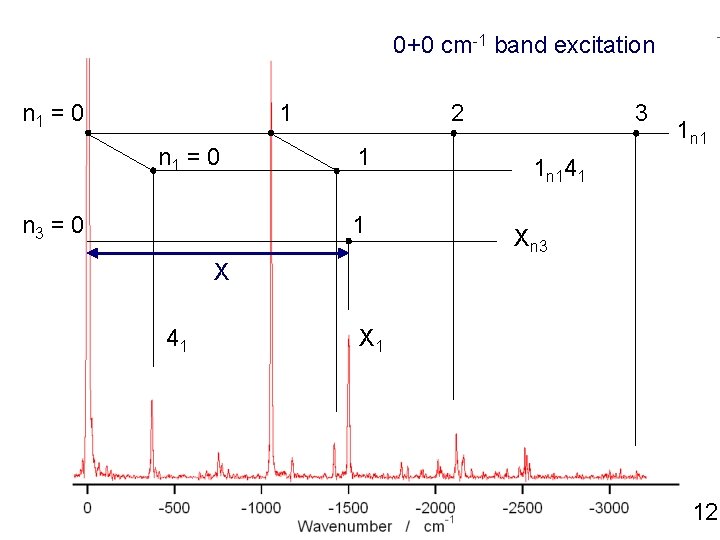
0+0 cm-1 band excitation n 1 = 0 1 n 1 = 0 n 3 = 0 2 1 1 3 1 n 141 Xn 3 X 41 X 1 12
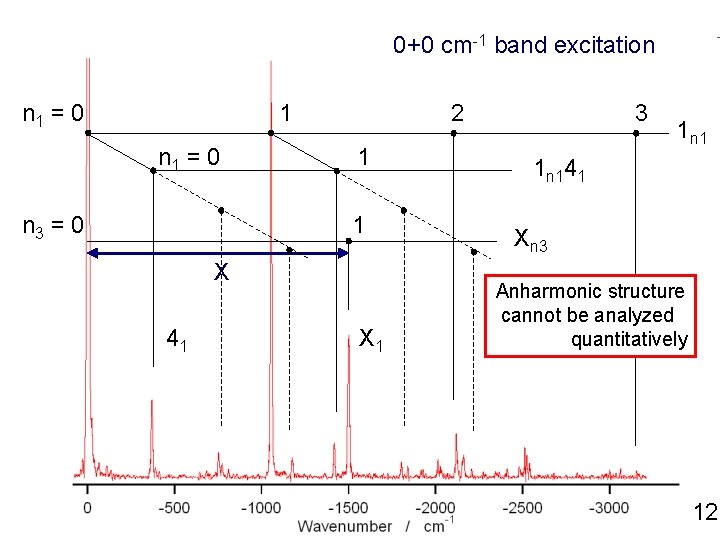
0+0 cm-1 band excitation n 1 = 0 1 n 1 = 0 n 3 = 0 2 1 1 X 41 X 1 3 1 n 141 Xn 3 Anharmonic structure cannot be analyzed quantitatively 12
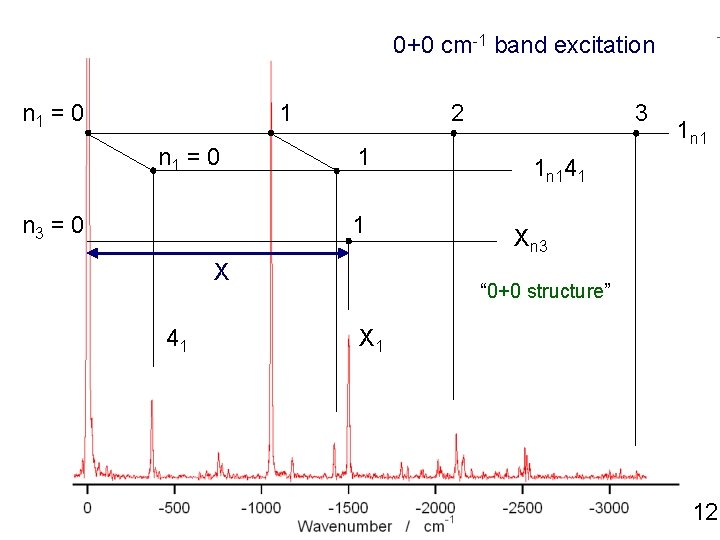
0+0 cm-1 band excitation n 1 = 0 1 n 1 = 0 n 3 = 0 2 1 1 X 41 3 1 n 141 Xn 3 “ 0+0 structure” X 1 12
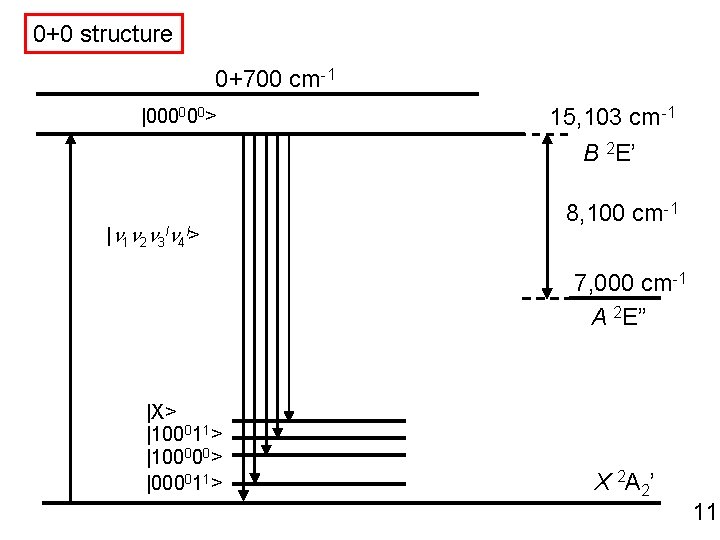
0+0 structure 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |X> |100011> |100000> |000011> X 2 A 2’ 11
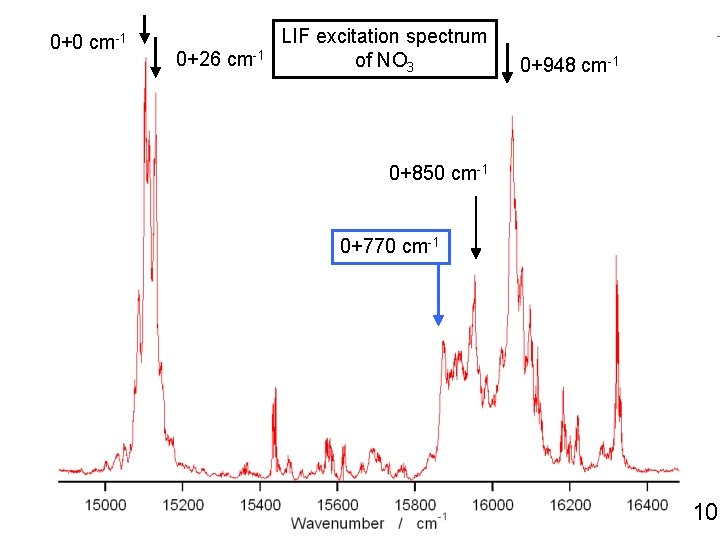
0+0 cm-1 0+26 cm-1 LIF excitation spectrum of NO 3 0+948 cm-1 0+850 cm-1 0+770 cm-1 10
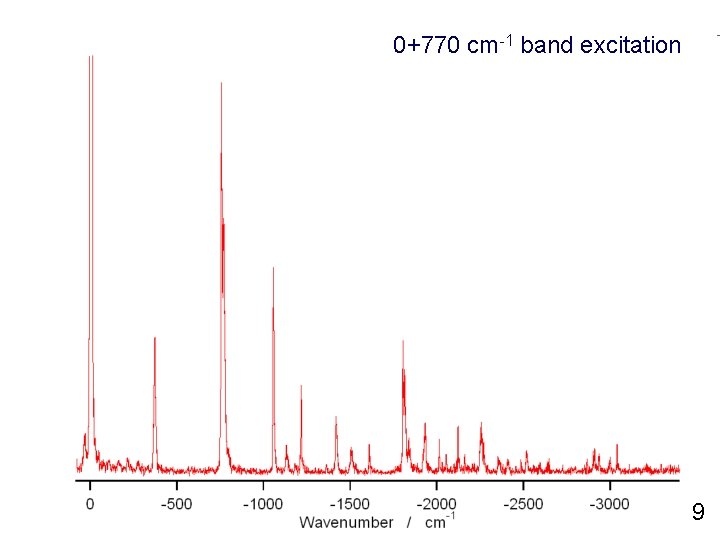
0+770 cm-1 band excitation 9

0+770 cm-1 band excitation Kim et al, JCP 96, 4057 (1992). 8
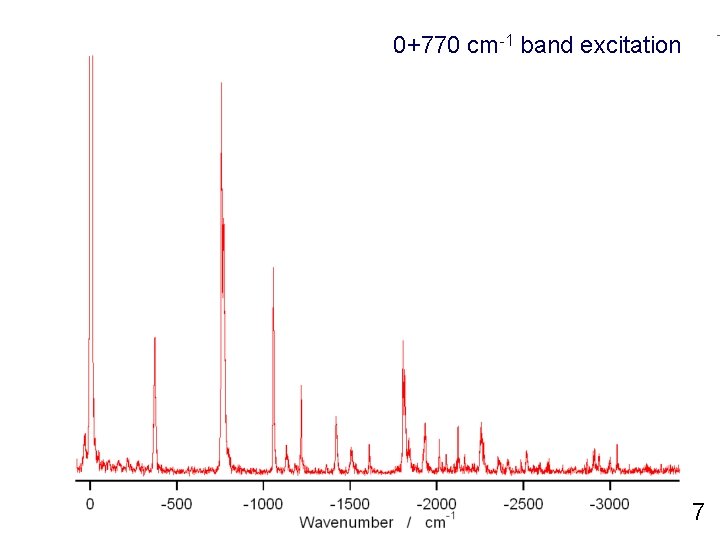
0+770 cm-1 band excitation 7
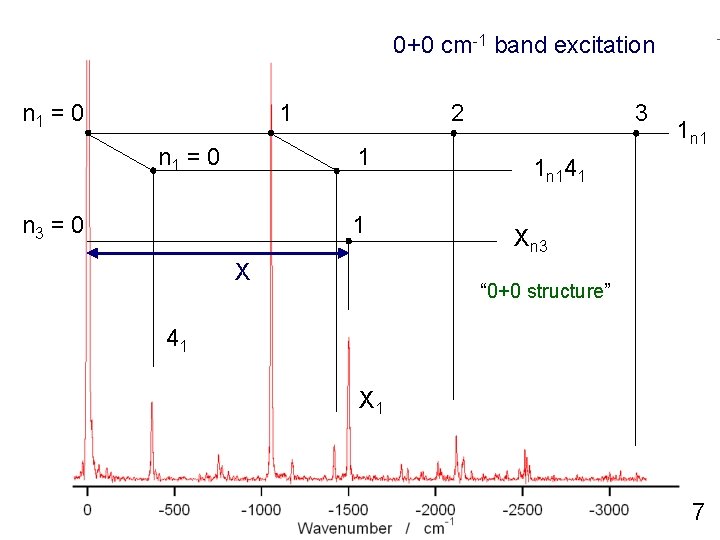
0+0 cm-1 band excitation n 1 = 0 1 n 1 = 0 2 1 n 3 = 0 1 X 3 1 n 141 Xn 3 “ 0+0 structure” 41 X 1 7
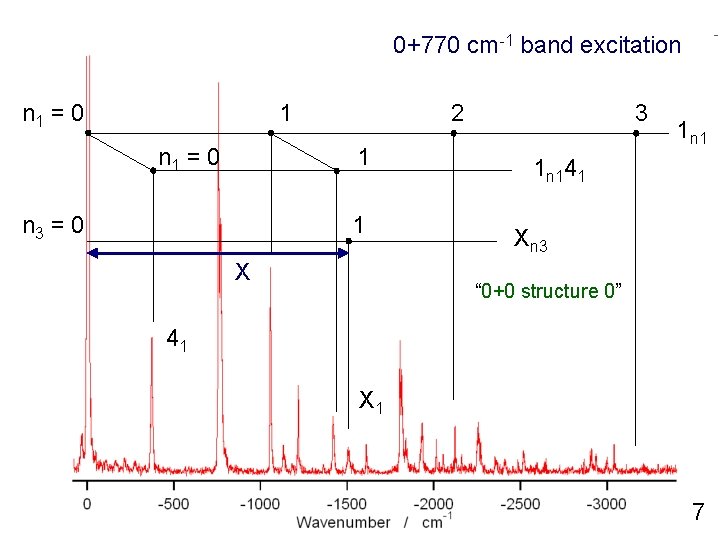
0+770 cm-1 band excitation n 1 = 0 1 n 1 = 0 2 1 n 3 = 0 1 X 3 1 n 141 Xn 3 “ 0+0 structure 0” 41 X 1 7
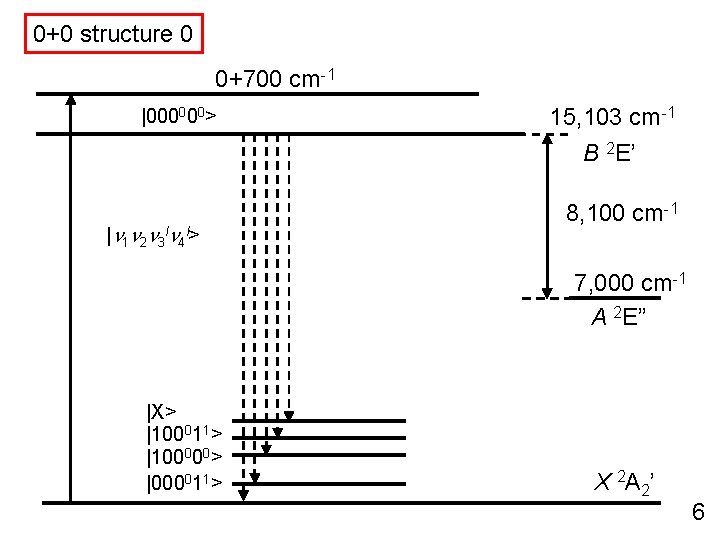
0+0 structure 0 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |X> |100011> |100000> |000011> X 2 A 2’ 6
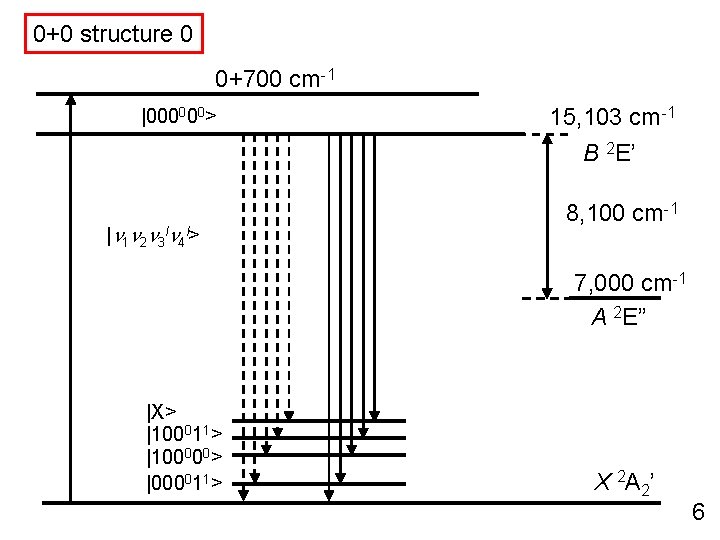
0+0 structure 0 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |X> |100011> |100000> |000011> X 2 A 2’ 6
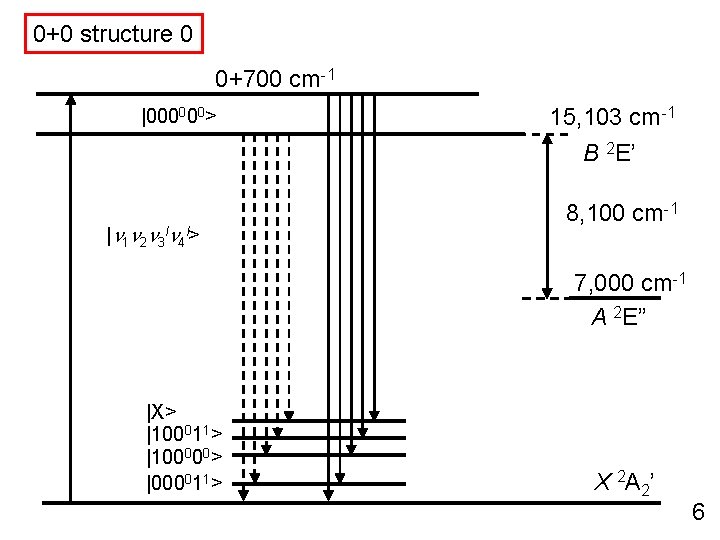
0+0 structure 0 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |X> |100011> |100000> |000011> X 2 A 2’ 6
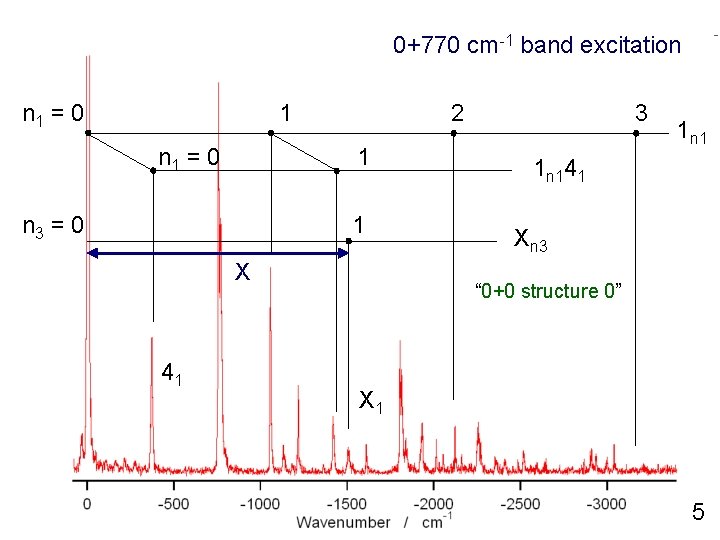
0+770 cm-1 band excitation n 1 = 0 1 n 1 = 0 2 1 n 3 = 0 1 X 41 3 1 n 141 Xn 3 “ 0+0 structure 0” X 1 5
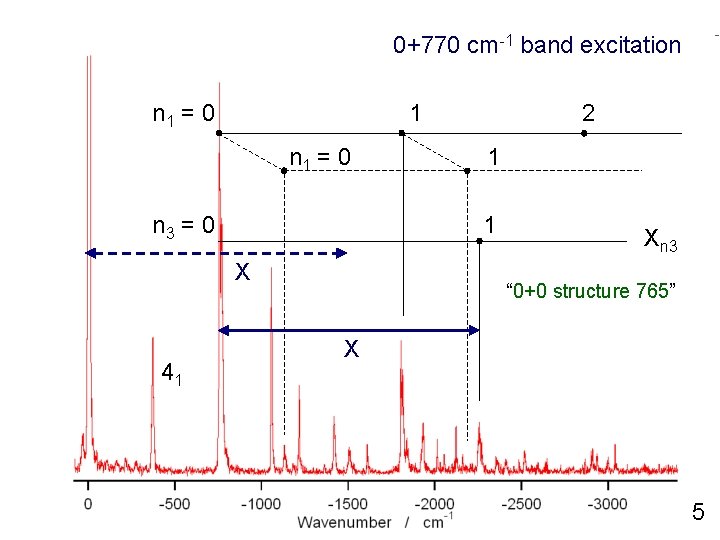
0+770 cm-1 band excitation n 1 = 0 1 n 1 = 0 n 3 = 0 1 1 X 41 2 Xn 3 “ 0+0 structure 765” X 5
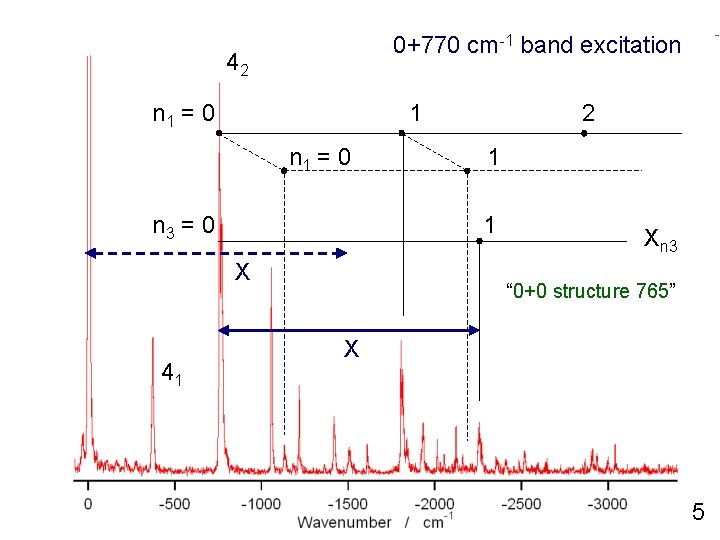
0+770 cm-1 band excitation 42 n 1 = 0 1 n 1 = 0 n 3 = 0 1 1 X 41 2 Xn 3 “ 0+0 structure 765” X 5
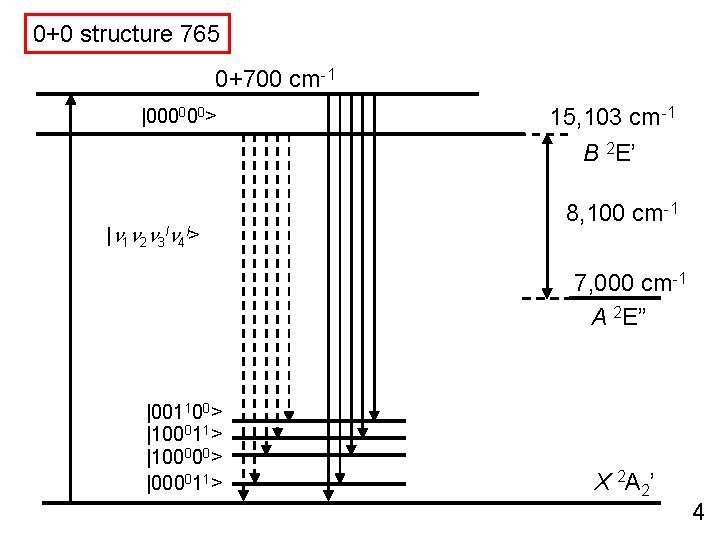
0+0 structure 765 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> X 2 A 2’ 4
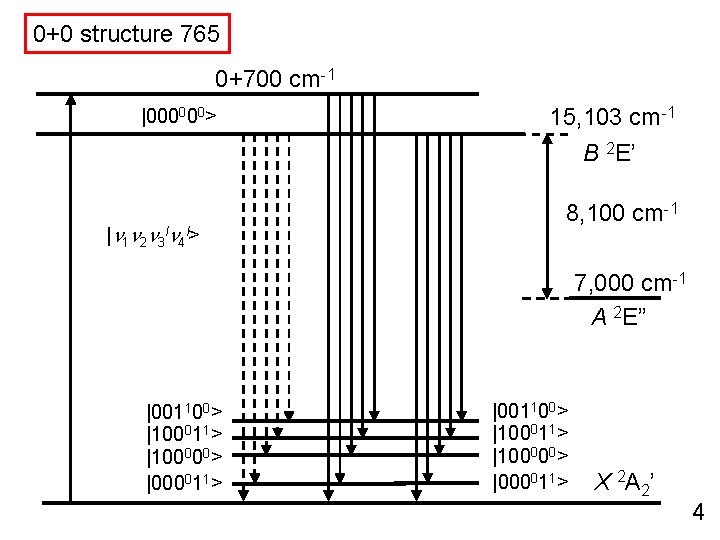
0+0 structure 765 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> X 2 A 2’ 4
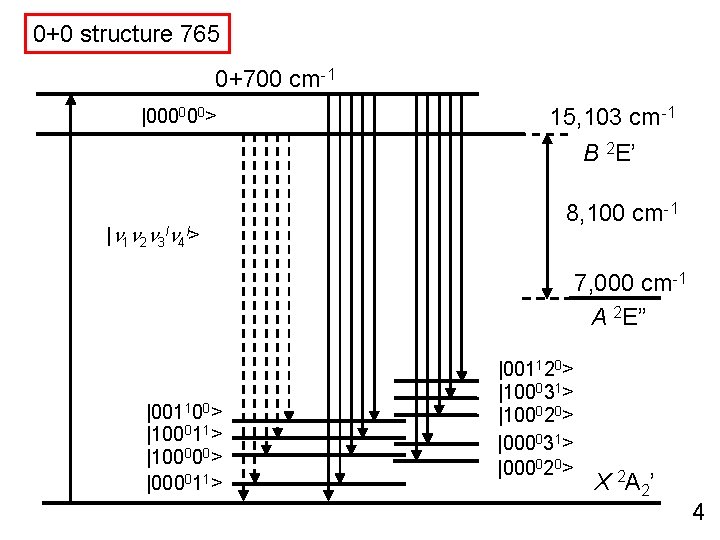
0+0 structure 765 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> |001120> |100031> |100020> |000031> |000020> X 2 A 2’ 4
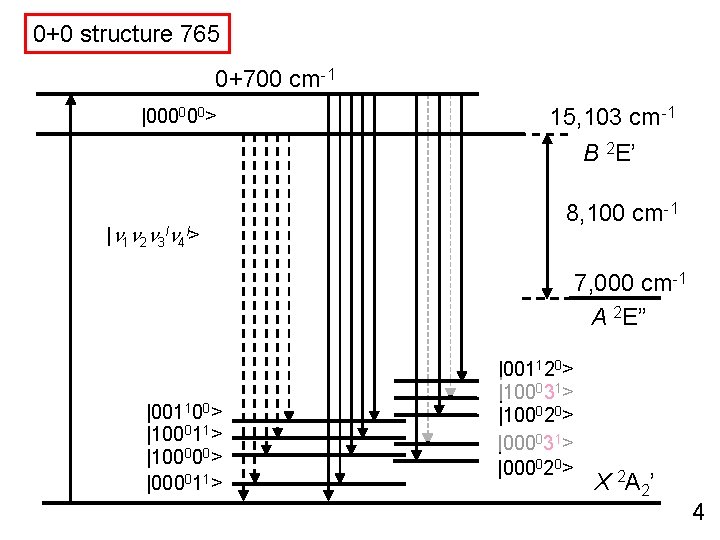
0+0 structure 765 0+700 cm-1 |000000> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> |001120> |100031> |100020> |000031> |000020> X 2 A 2’ 4
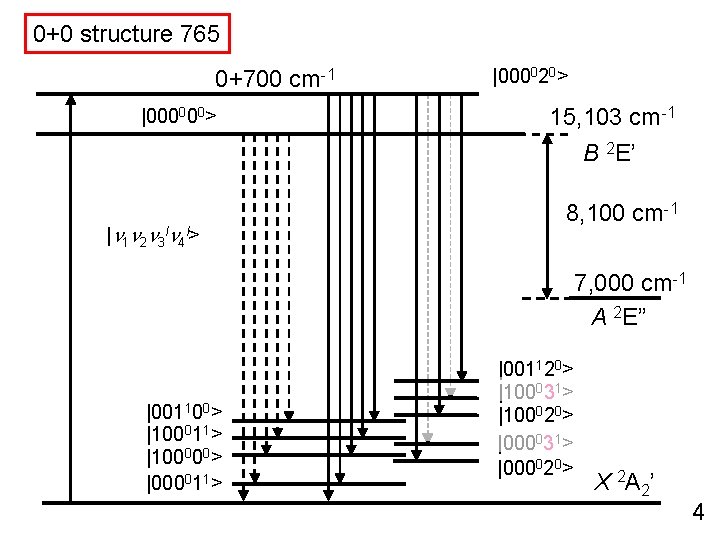
0+0 structure 765 0+700 cm-1 |000000> |000020> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> |001120> |100031> |100020> |000031> |000020> X 2 A 2’ 4
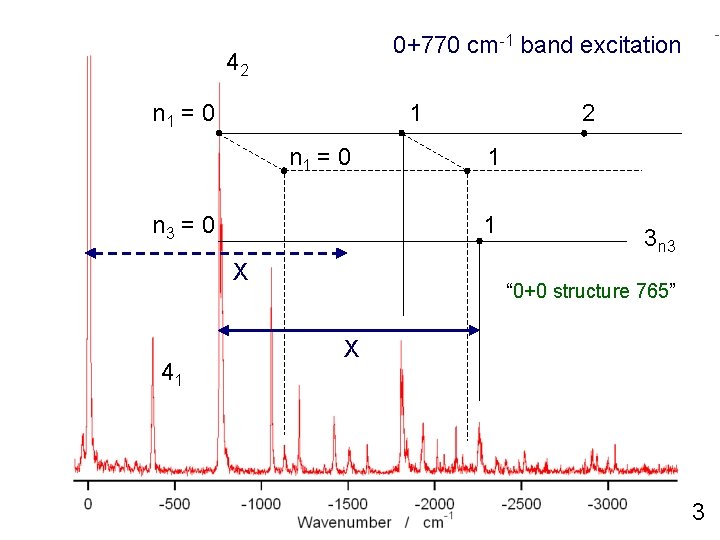
0+770 cm-1 band excitation 42 n 1 = 0 1 n 1 = 0 n 3 = 0 1 1 X 41 2 3 n 3 “ 0+0 structure 765” X 3
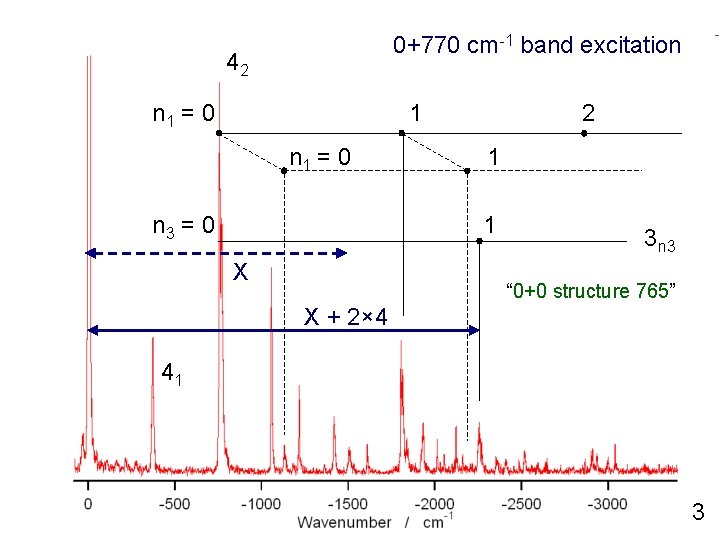
0+770 cm-1 band excitation 42 n 1 = 0 1 n 1 = 0 n 3 = 0 2 1 1 X 3 n 3 “ 0+0 structure 765” X + 2× 4 41 3
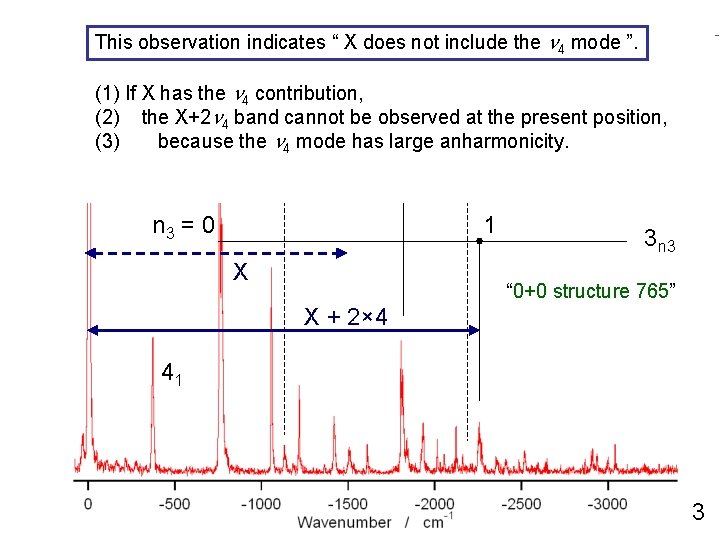
This observation indicates “ X does not include n 4 mode ”. 0+770 cm-1 the band excitation 42 (1) If X has the n 4 contribution, n 1 X+2 = 0 n 4 band cannot be observed 1 (2) the at the present 2 position, (3) because the n 4 mode has large anharmonicity. n 1 = 0 n 3 = 0 1 1 X 3 n 3 “ 0+0 structure 765” X + 2× 4 41 3
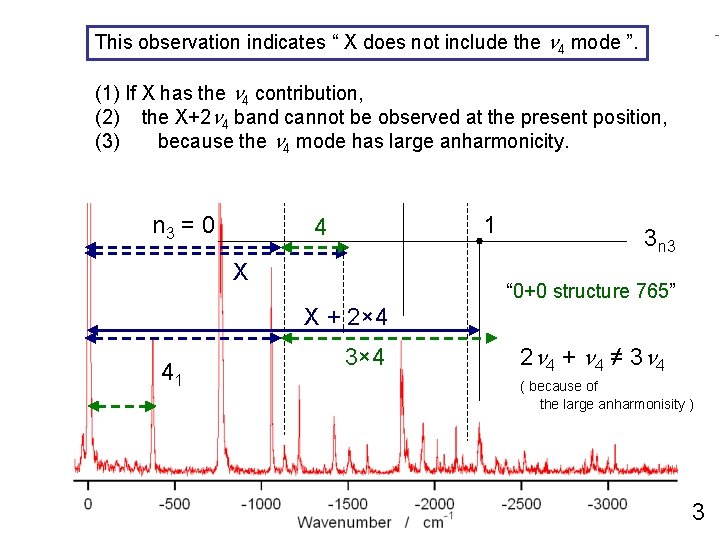
This observation indicates “ X does not include n 4 mode ”. 0+770 cm-1 the band excitation 42 (1) If X has the n 4 contribution, n 1 X+2 = 0 n 4 band cannot be observed 1 (2) the at the present 2 position, (3) because the n 4 mode has large anharmonicity. n 3 = 0 n 1 = 0 1 4 1 X 3 n 3 “ 0+0 structure 765” X + 2× 4 41 3× 4 2 n 4 + n 4 ≠ 3 n 4 ( because of the large anharmonisity ) 3
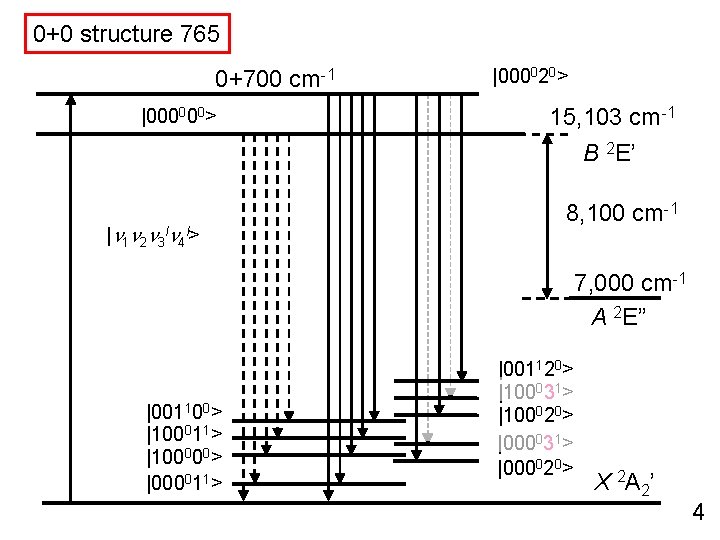
0+0 structure 765 0+700 cm-1 |000000> |000020> 15, 103 cm-1 B 2 E’ |n 1 n 2 n 3 ln 4 l> 8, 100 cm-1 7, 000 cm-1 A 2 E” |001100> |100011> |100000> |000011> |001120> |100031> |100020> |000031> |000020> X 2 A 2’ 4
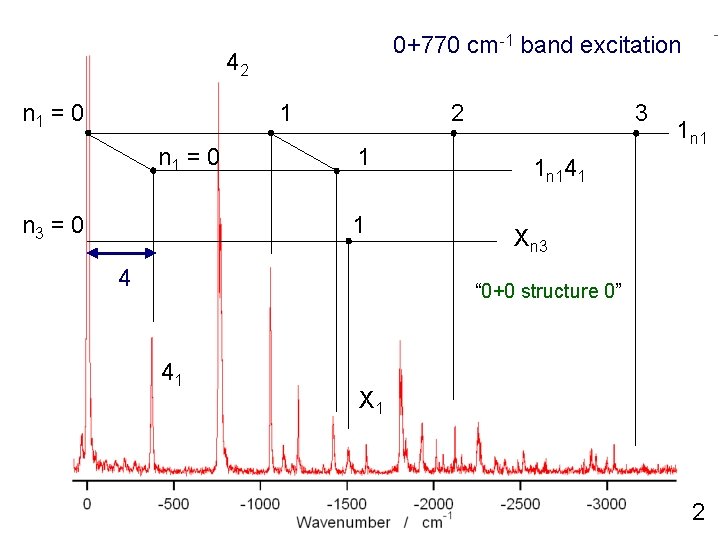
0+770 cm-1 band excitation 42 n 1 = 0 1 n 1 = 0 n 3 = 0 2 1 1 4 3 1 n 141 Xn 3 “ 0+0 structure 0” 41 X 1 2
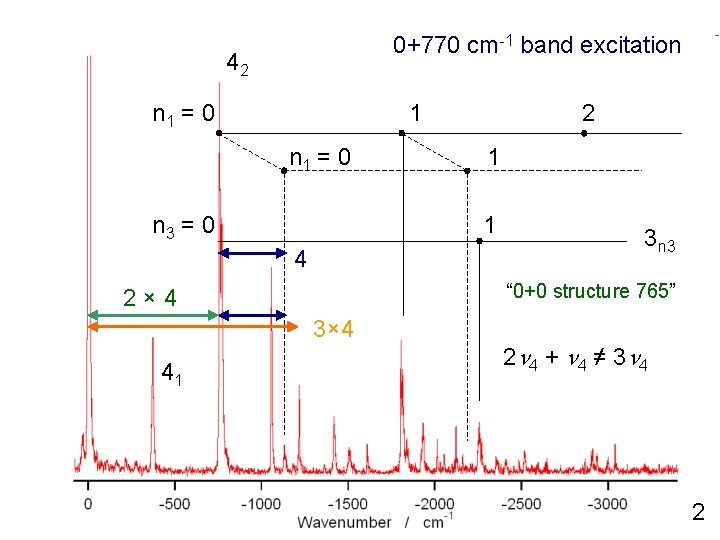
0+770 cm-1 band excitation 42 n 1 = 0 1 n 1 = 0 n 3 = 0 1 1 4 3 n 3 “ 0+0 structure 765” 2× 4 3× 4 41 2 2 n 4 + n 4 ≠ 3 n 4 2
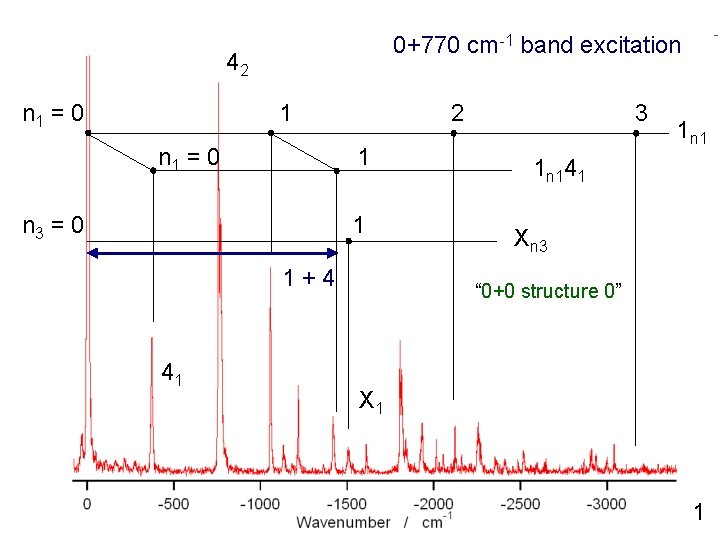
0+770 cm-1 band excitation 42 n 1 = 0 1 n 1 = 0 2 1 n 3 = 0 1 1+4 41 3 1 n 141 Xn 3 “ 0+0 structure 0” X 1 1
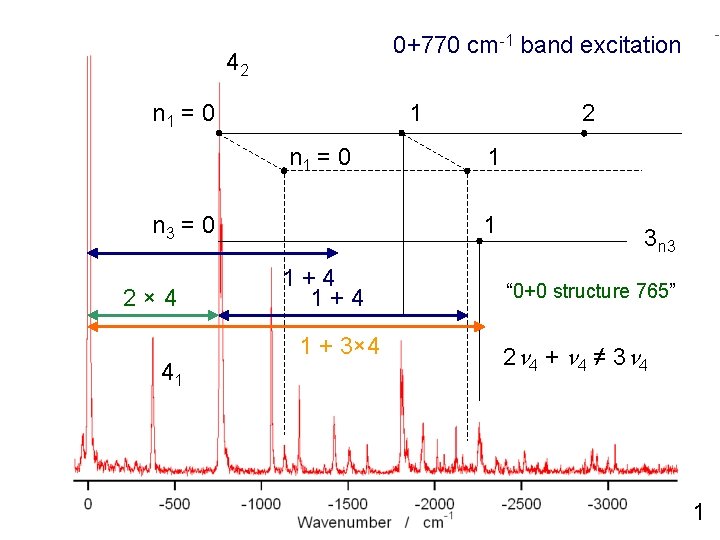
0+770 cm-1 band excitation 42 n 1 = 0 1 n 1 = 0 n 3 = 0 2× 4 41 2 1 1 1+4 1 + 3× 4 3 n 3 “ 0+0 structure 765” 2 n 4 + n 4 ≠ 3 n 4 1

Conclusions (1) Vibronic band to n 3 with ~1, 000 cm-1 has not been observed. (2) It is unusual that the n 3 band has negligible intensity while n 3 + n 4 has remarkable intensity. (3) SVL DF spectra show that the 1, 492 cm-1 level has no contribution of the n 4 mode. SVL DF spectroscopy holds Assignment I. Future works (1) Spectra from the higher bands ( laser dye R 640 ). (2) Isotope shift. Acknowledgement Prof. John Stanton ( for his stick spectrum ) 0
- Slides: 47