discrepancies Indications for ABO grouping ABO grouping is

discrepancies

Indications for ABO grouping: ABO grouping is required for all of the following individuals: Blood Donors-since it can be life threatening to give the wrong ABO group to the patient. Transfusion recipients-since we need to know the donor blood is ABO compatible. Transplant Candidates and Donors-ABO antigens are found in other tissues as well. Therefore the transplant candidates and donors must be compatible.

Indications for ABO grouping: Newborns (sometimes) If the baby is demonstrating symptoms of Hemolytic Disease of the Newborn, the ABO group needs to be determined along with Rh and others. Paternity testing Since the inheritance of the ABO Blood Group System is very specific, this serves as one of the first methods to determine the likelihood that the accused father is the father or not. Prenatal Patients-To determine whether the mothers may have babies who are suffering from ABO-HDN. It is also beneficial to know the ABO group should she start hemorrhaging.

O B A f o s t c e g p n s i p A u l a o r c i t G c a r P

Practical Aspects of ABO Grouping Patient Identification › The patient MUST be positively identified and preferably banded. Some institutions use specific Blood Bank arm bands. › Ask patient to state his/her name. › Responsible party should identify patient if he/she cannot. › Verify information by comparing to ID band. › Resolve any differences before proceeding with the blood draw.

Practical Aspects of ABO Grouping Labeling of Sample › The information on sample MUST match information on ID band, which would also needs to be consistent with the order. › Information on samples MUST include the following: Name (last, first, middle initial) and no nicknames. Unique identification number such as medical record number or possibly social security number. Date and time sample drawn along with the signature or unique identifier of phlebotomist (on sample or on orders) Gender and birth date desirable but not mandatory. The date of birth provides another unique identifier along with the medical record number and full name of the patient.

Practical Aspects of ABO Grouping Mislabeled Samples › Do NOT accept any sample not properly labeled. › The following are what would warrant an improperly labeled specimen: Missing information Incorrect information Information on sample not matching information on orders › Improperly labeled samples must be discarded if the problem cannot be resolved. › In the case of an emergency blood draw on a patient who is unidentified at that time, the blood specimen must also discarded when both name and medical record number have changed

Practical Aspects of ABO Grouping Technical guidelines › ABO grouping tests should be done at room temperature (2 Oo 24 o. C) or lower; testing in hot environment weakens the reaction. › Routine ABO grouping must include both cell and serum testing as each serves as a check on the other. › Antisera used in the ABO grouping must be used as per the manufacturer’s instructions. › Adequate controls should be put with each batch of tests for quality control of the reagents, alternately once a day the reagents must be checked with appropriate cells. › Before use all the reagents must be checked grossly to rule out any turbidity or contamination.

Practical Aspects of ABO Grouping › Tubes, slides, microplates or gel cards should be labeled properly. › One should not rely on the colour of the dye to identify the antiserum. › The glassware used should be dry and clean. › Serum should be added before adding cells and each tube should be examined after serum has been added to ensure that none has been missed. › Results should be recorded immediately after observation. › Concave mirror (agglutination viewer) or microscope may be used to examine reactions that appear negative by the naked eye.

Practical Aspects of ABO Grouping Blood sample for ABO grouping › Properly labelled samples of clotted blood collected in screwcapped plain test-tube are most suitable for ABO grouping. Ths amples should be stored at 4°C and preferrably be tested within 48 hours. › Haemolysed samples are not suitable for testing. › The blood sample may be centrifuged at 1000 -3000 rpm for 3 minutes for adequate serum separation. › Cells from the test sample should be washed in 0. 9% nromal saline and a 2 -5% cell suspension should be prepared.

Most samples for blood banking are drawn into a red top tube - serum is preferred. No clot activation tube should be used since the patient's red cells may also need to used and no other chemicals should be present Antigens on cells are stable longer (months) in a clot tube.

Plasma or serum ? For antibody detection, plasma has theoretical disadvantage, compared with serum, of: › Containing anticoagulants that inhibit complement activation and therefore possibly interfere with the detection of some complement-activating antibodies. › False positive may form in case of plasma due to the formation of small fibrin clots. A few tests require an EDTA sample if complement is not to be activated. Serum must be tested while fresh to ensure good complement activity.

BLOOD BANKING REAGENTS The techniques used in Blood Bank involves › Specific antigens carried on red blood cells › Specific antibodies for certain red cell antigens.

Sources of Antigen Testing In almost all blood bank techniques we have red cells with antigens present. These red cells may either reagent red cells with known antigens, patient red cells, or donor red cells. The reagent red cells are commercially prepared and have all the red cell antigens identified. When we use red cells where the antigens have already been determined, we can identify the possible antibodies present.

The reagent cells used for blood banking include the following: › A 1 and B cells for confirmation of the ABO type in all patients and donors other than newborn babies › Antibody identification cell panel are O cells with the specific antigens known. Usually there are between 8 and 12 different cells in a cell panel. The pattern of positive and negative reactions help identify the antibody.

ABO grouping sera It is essential to use reliable grouping reagents to obtain correct results. Commercially prepared polyclonal (human source) and monoclonal (tissue culture) antisera are available Adequate quality control must be done before purchasing commercial antisera. Each batch of antisera must be checked against A 1, A 2, (if available, otherwise A group) B and 0 cells before use. The potency of any antisera deteriorates rapidly if kept for too long at ambient temperature, grouping sera should therefore, be kept at 4°C or as directed by the manufacturer when not in use. Frozen antisera must be completely thawed before use and no refreezing should be done.

Slide or Tile Testing This technique may be used for emergency ABO grouping tests or for preliminary grouping particularly in an outdoor camp, however it should always be supplemented with a cell and serum grouping using any one of the other above mentioned techniques. Slide or tile testing is not recommended for routine use because it is not reliable for › weakly reactive antigens on cells › serum grouping with low titre anti-A or anti-B Disadvantages › Less sensitive than the tube test › Drying up of the reaction mixture can cause aggregation of cells, giving false positive results. › Weaker reactions are difficult to interpret.

Tube Testing Test tubes either of glass or plastic may be used, of l. Ox 75 mm size. The tube technique is more sensitive than slide technique for ABO grouping. Advantages of tube testing › It allows for fairly long incubation without drying up of the tubes’ contents. › It allows to distinguish hemolysis. › Centrigugation involved enhances the reaction allowing weaker antigens and antibodies to be detected. › Simplicity of reading and grading of results. › Clean and more hygienic. › Requires smaller volume of reagents.
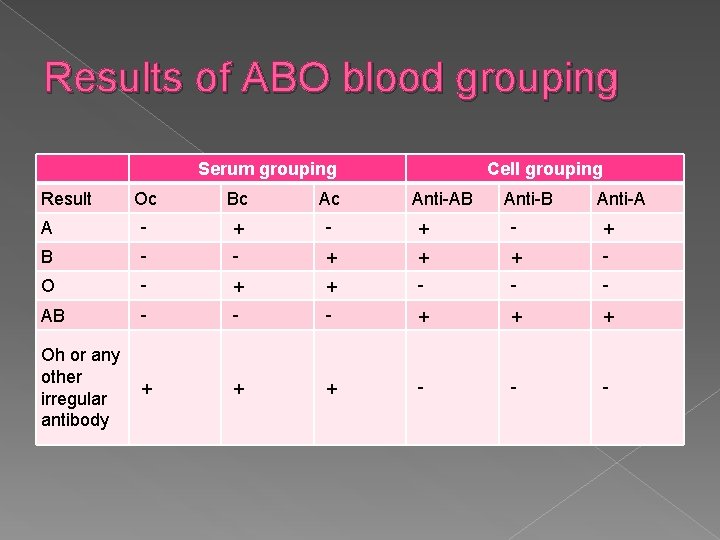
Results of ABO blood grouping Result Serum grouping Cell grouping Oc Bc A - + - + B - - + + + - O - + + - - - AB - - - + + + - - - Oh or any other + irregular antibody Anti-AB Anti-A

e r c a p s i d A O B c n s ie

Discrepancies A discrepancy occurs when the red cell testing does NOT match the serum testing results In other words, the forward does NOT match the reverse
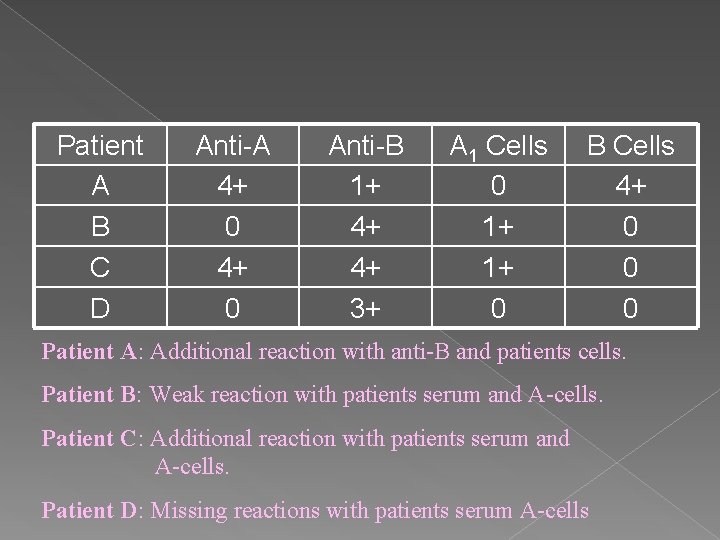
Patient A B C D Anti-A 4+ 0 Anti-B 1+ 4+ 4+ 3+ A 1 Cells 0 1+ 1+ 0 B Cells 4+ 0 0 0 Patient A: Additional reaction with anti-B and patients cells. Patient B: Weak reaction with patients serum and A-cells. Patient C: Additional reaction with patients serum and A-cells. Patient D: Missing reactions with patients serum A-cells

Why ? Most of the time, the problem is technical › Either repeat test on same sample, request a new sample, or wash cells Other times, there is a real discrepancy due to problems with the patient’s red cells or serum If a real discrepancy is encountered, the results must be recorded However, the interpretation is delayed until the discrepancy is RESOLVED

Importance It is important to recognize discrepant results and how to (basically) resolve them Remember, the ABO system is the most important blood group system in relation to transfusions Misinterpreting ABO discrepancies could be life threatening to patients

s r o r r l E a c i n h c e T

Technical Errors Clerical errors › Mislabeled tubes › Patient misidentification › Inaccurate interpretations recorded › Transcription error › Computer entry error › Mix up in samples

Reagent or equipment problems 1. Contaminated sample or reagent Sample contamination Microbial growth in tube Reagent contamination Bacterial growth causes cloudy or discolored appearance…do not use if you see this! Reagents contaminated with other reagents (don’t touch side of tube when dispensing) Saline should be changed regularly Incorrect storage temperatures 2. hemolyzed reagents 3. Using expired reagent

4. Using an uncalibrated centrifuge › Routine maintenance should be performed on a regular basis (daily, weekly, etc) › Keep instruments like centrifuges, thermometers, and timers calibrated Uncalibrated serofuges can cause false results

Procedural errors › › › › Reagents not added Manufacturer’s directions not followed RBC suspensions incorrect concentration Cell buttons not resuspended before grading agglutination Warming during centrifugation Rough dislodgment of the button while reading the result Inaccurate speed and time of centrifuge Too many cells in your cell suspension can lead to decreased or negative reactions since there are too many cells for the number of antibodies present in the reagents.

Procedural errors Failure to detect weak results can occur if you are not watching the reactions while you are shaking them out or if you shake too hard. Failure to detect hemolysis can be a definite problem. Remember a positive reaction can be hemolysis as well as agglutination since the antigen -antibody reaction can bind complement. When complement is bound it can lead to hemolysis that is also an indication of a positive reaction. Dirty glassware can cause the cells to artificially clump.

Clotting deficiencies Serum that does not clot may be due to: › Low platelet counts › Anticoagulant therapy (Heparin, Aspirin, etc) › Factor deficiencies Serum that does not clot completely before testing is prone to developing fibrin clots that may mimic agglutination Thrombin can be added to serum to activate clot formation OR, tubes containing EDTA can be used

Hemolysis Detected in serum after centrifugation (red) Important if not documented Can result from: › Complement binding Anti-A, anti-B, anti-H, and anti-Lea › Bacterial contamination Red supernatant

ABO Discrepancies Problems with RBCs Weak-reacting/Missing antigens Extra antigens Mixed field reactions Problems with SERUM Weak-reacting/Missing antibodies Extra antibodies

e r c s i d R l a e a p c n s ie

ABO Discrepancies must be resolved In RECIPIENTS the discrepancies must be resolved before any blood component is transfused. › If not resolved before blood is needed, transfuse Group O (O NEGATIVE if there is a discrepancy in the Rh type also). In DONORS the discrepancies must be resolved before any blood is labeled with a blood type.

Categories of ABO Discrepancies A. B. C. D. Cell Typing – Additional Reactions Cell Typing – Missing Reactions Serum Typing- Additional Reactions Serum Typing- Missing Reactions
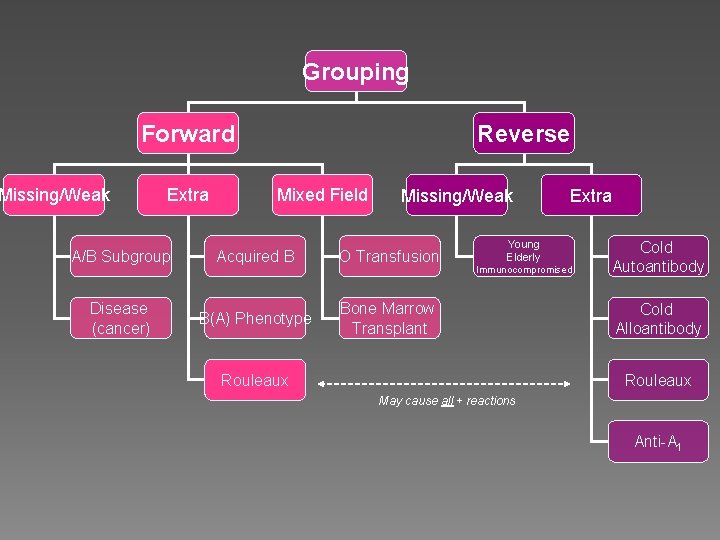
Grouping Forward Missing/Weak Extra Reverse Mixed Field Missing/Weak A/B Subgroup Acquired B O Transfusion Disease (cancer) B(A) Phenotype Bone Marrow Transplant Extra Young Elderly Immunocompromised Rouleaux Cold Autoantibody Cold Alloantibody Rouleaux May cause all + reactions Anti-A 1

Cell Typing Additional Reactions
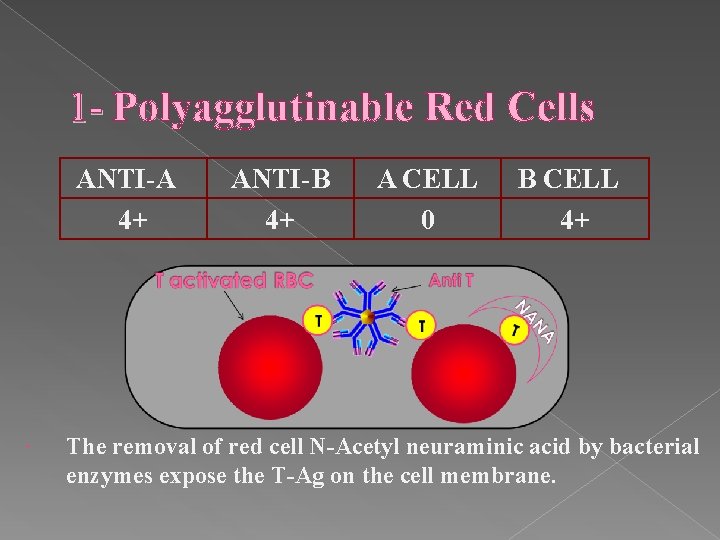
1 - Polyagglutinable Red Cells ANTI-A 4+ ANTI-B 4+ A CELL 0 B CELL 4+ The removal of red cell N-Acetyl neuraminic acid by bacterial enzymes expose the T-Ag on the cell membrane.

• Antibodies to T-antigens are naturally present in most human sera. • This Ab can also be found as a contaminant in some ABO typing reagents. • This cause unexpected agglutination of T Ags on red cells.

2 - Acquired Antigens ANTI-A 4+ • • ANTI-B 2+ A CELL 0 B CELL 4+ Microbial deacetylating enzymes such as E. coli cleave off the N-Acetyl of the Group A 1 N-acetylgalactosamine The remaining galactosamine becomes similar to the Group B galactose Anti-B react with this B-like Ag causing agglutination A-like Ag can also be acquired

Acquired B Bacteria (E. coli) have a deacetylating enzyme that effects the A sugar…. Group A individual N-acetyl galactosamine Bacterial enzyme removes acetyl group Acquired B Phenotype Galactosamine now resembles D -galactose (found in Group B)

Resolving Acquired B Check patient diagnosis: Infection? Some manufacturers produce anti-B reagent that does not react with acquired B Test patients serum with their own RBCs › The patients own anti-B will not react with the acquired B antigen on their red cell (autologous testing)

B(A) phenotype Similar to acquired B Patient is Group B with an apparent extra A antigen The B gene transfers small amounts of the A sugar to the H antigen Sometimes certain anti-A reagents will detect these trace amount of A antigen Resolution: test with another anti-A reagent from another manufacturer

3 - Non-specific Agglutination Wharton’s Jelly – gelatinous substance derived from connective tissue that is found in cord blood and may cause false agglutination Wharton’s Jelly Coats newborn cord cells and the child's type may appear AB. We do not do a reverse on newborn blood since they have not made any anti-A or anti-B yet. Wharton’s jelly

If the baby types as an AB recheck by washing cells several times and re-testing since you need to make sure you have removed the Wharton's Jelly and the baby is truly an AB. Better ALWAYS wash cord blood at least 4 to 5 before determining the type of the baby, or request new sample from heel
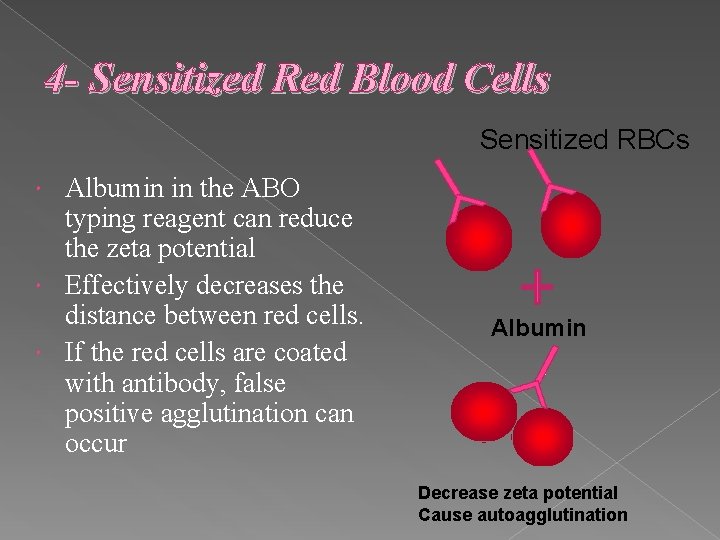
4 - Sensitized Red Blood Cells Sensitized RBCs Albumin in the ABO typing reagent can reduce the zeta potential Effectively decreases the distance between red cells. If the red cells are coated with antibody, false positive agglutination can occur Albumin Decrease zeta potential Cause autoagglutination

Cell Typing Missing Reactions

1 - Subgroups ANTI-A ANTI-B A CELL B CELL 0 0 0 4+ Weak variants of both A & B Carry poorly expressed Ags May not produce expected reactions with anti. A & anti-B They are categorized according to the strength and pattern of reaction with anti-A, anti-H & anti-A, B

2 - Altered Antigen Expression ANTI-A ANTI-B A CELL B CELL 0 0 0 4+ • Weak Ags may be found on RBCs of some people with diseases (Leukemia)
3 - Excessive amounts of group specific substances Patients with certain types of cancer Large amounts of soluble A or B Ags Inhibit anti-A or anti-B typing reagent • Can be resolved by washing RBCs prior to ABO typing
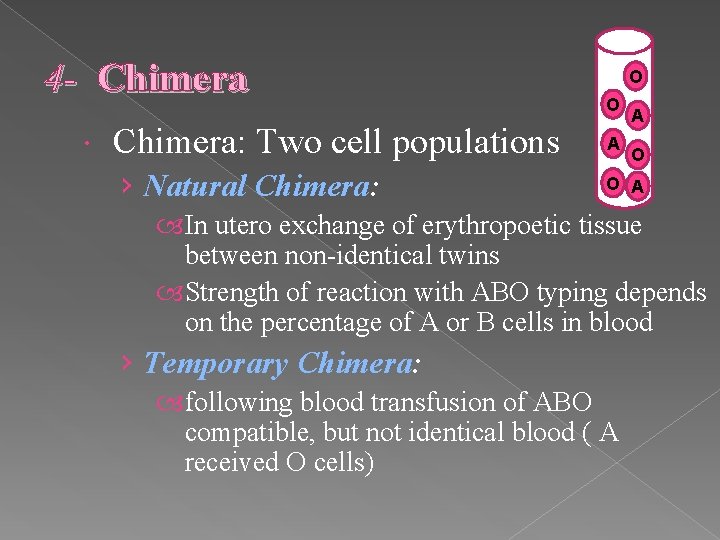
4 - Chimera: Two cell populations › Natural Chimera: O O A A O O A In utero exchange of erythropoetic tissue between non-identical twins Strength of reaction with ABO typing depends on the percentage of A or B cells in blood › Temporary Chimera: following blood transfusion of ABO compatible, but not identical blood ( A received O cells)

Chimera will produce Mixed Field Agglutination Agglutinates are seen with a background of unagglutinated cells Mixed Field Agglutination is also seen in A 3 phenotype (sometimes B 3) Anti-A Anti-B 0 2+ mf A 1 Cells B Cells 4+ 0
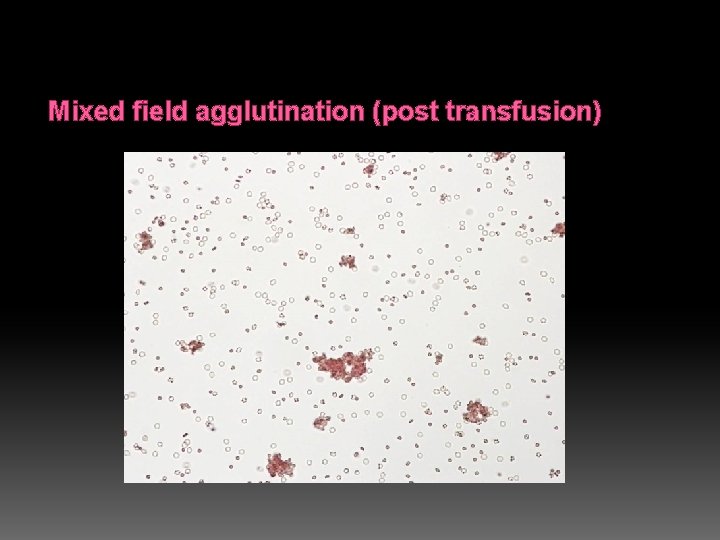
Mixed field agglutination (post transfusion)

Serum Typing Additional Reactions

1 - Alloantibodies Abs other than anti-A or anti-B Can agglutinate A or B cells if express specific Ag Abs commonly cause this discrepancy anti-M, N, -S, -Lea, Leb, -P 1, A 1 Can be identified by testing serum with a panel of O cells that have been phenotyped for these Ags

2 - Autoantibodies Ig. M autoantibodies can cause false-positive results in cell & serum grouping Problem can be solved by washing cells with warm saline prior to testing In serum typing, autoabsorption can be performed Or serum typing at 37 o. C

3 - Rouleaux formation ANTI-A ANTI-B A CELL B CELL 4+ 0 2+ 4+ Rouleaux may also give false positive cell typing if strong enough Looks like agglutination macroscopically This phenomenon is due to alteration in serum protein concentration caused by: › elevated levels of gammaglobulins › elevated levels of fibrinogen › Also seen with plasma expanders (dextran) Cell grouping- can be avoided by washing RBCs Serum grouping- addition of saline

Reverse Grouping Extra Antibodies `

1 - Cold antibodies Sometimes a patient will develop cold-reacting allo- or auto-antibodies that appear as “extra” antibodies on reverse typing Alloantibodies are made against foreign red cells Autoantibodies are made against ones own red cells. Cold reacting antibodies cause agglutination with red cells at room temperature and below. The autocontrol will be positive. › Resolution: warming tube to 37° and washing red cells can disperse agglutination; breaking the Ig. M bonds with 2 ME will also disperse cells

2 - Rouleaux Can cause both extra antigens and extra antibodies “stack of coins” appearance May falsely appear as agglutination due to the increase of serum proteins (globulins) Associated with: › Multiple meloma › Macroglobulinemia › Hydroxyethyl starch (HES), dextran, etc

Resolving Rouleaux Remove proteins! If the forward grouping is affected, wash cells to remove protein and repeat test If the reverse grouping is affected, perform saline replacement technique (more common) › Cells (reagent) and serum (patient) centrifuged to allow antigen and antibody to react (if present) › Serum is removed and replaced by an equal volume of saline (saline disperses cells)* › Tube is mixed, centrifuged, and reexamined for agglutination (macro and micro)

3 - Anti-A 1 Sometimes A 2 (or A 2 B) individuals will develop an anti-A 1 antibody A 2 (or A 2 B) individuals have less antigen sites than A 1 individuals The antibody is a naturally occurring Ig. M Reacts with A 1 Cells, but not A 2 Cells + A 1 cells Anti-A 1 from patient + A 2 cells AGGLUTINATION NO AGGLUTINATION

Resolving anti-A 1 discrepancy 2 steps: › Typing patient RBCs with Anti-A 1 lectin › Repeat reverse grouping with A 2 Cells instead of A 1 Cells › Both results should yield NO agglutination Anti-A Anti-B 4+ 0 A 1 Cells 2+ B Cells 4+

Serum Typing Missing Reactions

1 - Newborns Infants develop ABO Abs by 3 -6 months of age Serum typing before this time: › Weak reaction › Negative reaction

2 - Patients with Hypogamma-globulinemia Have low levels of immunoglobulins Anti-A & anti-B may not be detected
3 - Chimera Twins that have chimeric blood group can lack A & B Abs Chimera with 98% O cells & 2% B cells › Group as O › Serum contain only anti-A

4 - Reagent Problems Deterioration of Ags on A or B cells used for serum typing › Weak › Negative reaction

Resolving Weak or Missing antibodies Determine patients age, diagnosis Incubate serum testing for 15 minutes (RT) to enhance antibody reactions If negative, place serum testing at 4°C for 5 minutes with autologous control (a. k. a. Autocontrol, AC) This is called a “mini-cold” panel and should enhance the reactivity of the antibodies

s ie c n a d O s e R B A g n i v l o p e r c is

Resolving ABO Discrepancies Finding the problem › Forward type tests for the antigen (red cell) › Reverse type tests for the antibody (serum) › Identify what the patient types as in both Forward & Reverse Groupings › Is there a weaker than usual reaction? › Is it a missing, weak, or extra reaction Get the patient’s history › › age Recent transplant Recent transfusion Patient medications

Let’s practice !
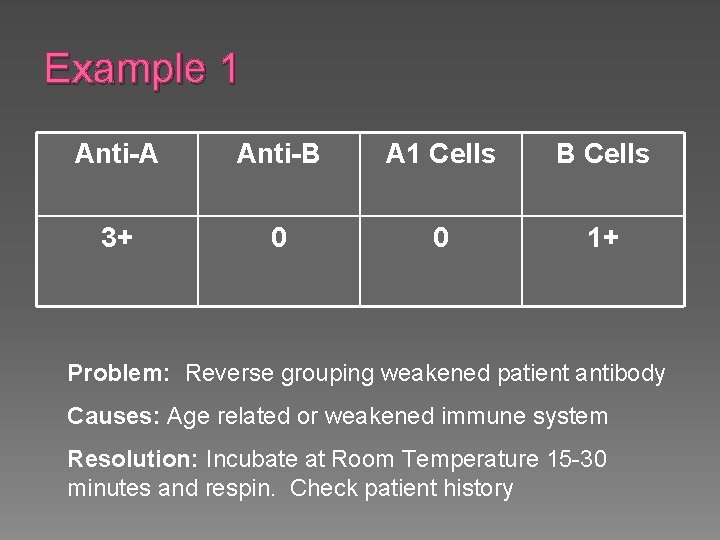
Example 1 Anti-A Anti-B A 1 Cells B Cells 3+ 0 0 1+ Problem: Reverse grouping weakened patient antibody Causes: Age related or weakened immune system Resolution: Incubate at Room Temperature 15 -30 minutes and respin. Check patient history

Example 2 Anti-A Anti-B A 1 Cells B Cells 3+ 1+ 0 4+ Problem: 1+ reaction with anti-B. Appears to have additional antigens Causes: Acquired B antigen Resolution: Patient history – bowel obstruction, carcinoma of colon/rectum(E. coli)
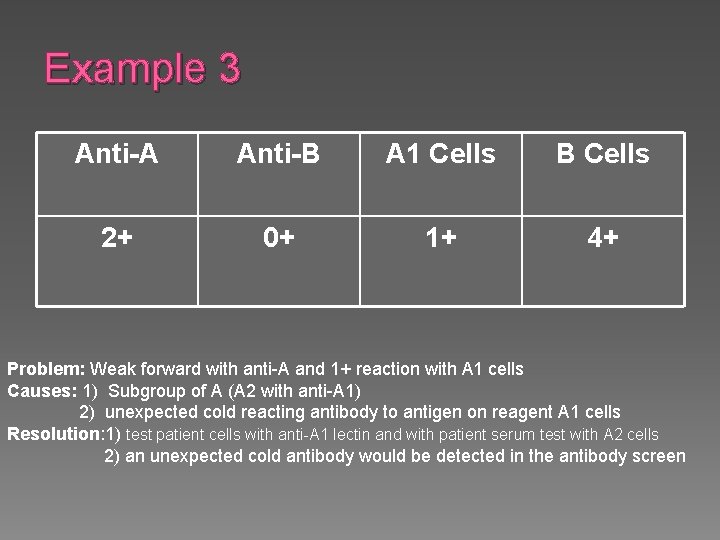
Example 3 Anti-A Anti-B A 1 Cells B Cells 2+ 0+ 1+ 4+ Problem: Weak forward with anti-A and 1+ reaction with A 1 cells Causes: 1) Subgroup of A (A 2 with anti-A 1) 2) unexpected cold reacting antibody to antigen on reagent A 1 cells Resolution: 1) test patient cells with anti-A 1 lectin and with patient serum test with A 2 cells 2) an unexpected cold antibody would be detected in the antibody screen

Example 4 Anti-A Anti-B A 1 Cells B Cells 0 0 0 3+ Problem: missing antigen in forward grouping. Patient appears as group A in reverse grouping Causes: A subgroup Resolution: extend incubation time because this may enhance the reaction. Test with a polyclonal or monoclonal blend of anti-A, B (may contain subgroup antigens)…. .
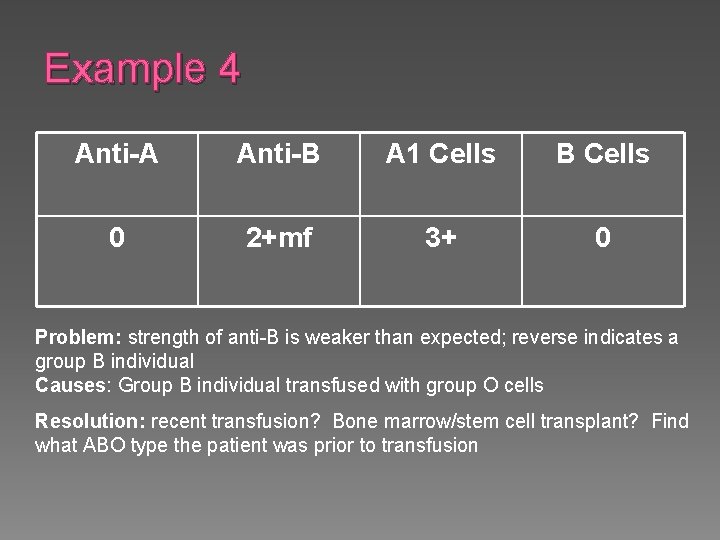
Example 4 Anti-A Anti-B A 1 Cells B Cells 0 2+mf 3+ 0 Problem: strength of anti-B is weaker than expected; reverse indicates a group B individual Causes: Group B individual transfused with group O cells Resolution: recent transfusion? Bone marrow/stem cell transplant? Find what ABO type the patient was prior to transfusion

Example 5 Anti-A Anti-B A 1 Cells B Cells 4+ 4+ 0 1+ Problem: : Forward shows AB individual, Reverse shows weaker “extra” reaction with B cells (looks like a group A) Causes: Possible cold allo- or autoantibody (patient may have an antibody to another blood group system; A 1 and B cells may have the antigens to these antibodies) (allo: P, M, N, Lewis) (auto: I or IH) Resolution: screen for antibodies using Screening Cells and an autocontrol (we’ll talk later about Ab screens
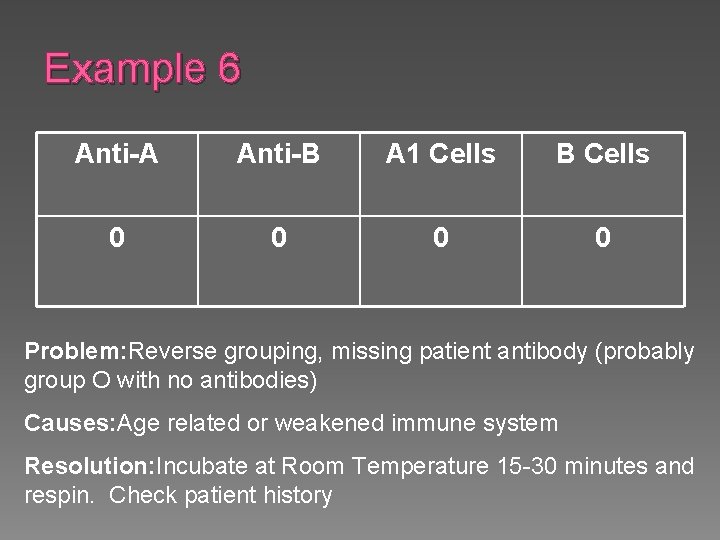
Example 6 Anti-A Anti-B A 1 Cells B Cells 0 0 Problem: Reverse grouping, missing patient antibody (probably group O with no antibodies) Causes: Age related or weakened immune system Resolution: Incubate at Room Temperature 15 -30 minutes and respin. Check patient history
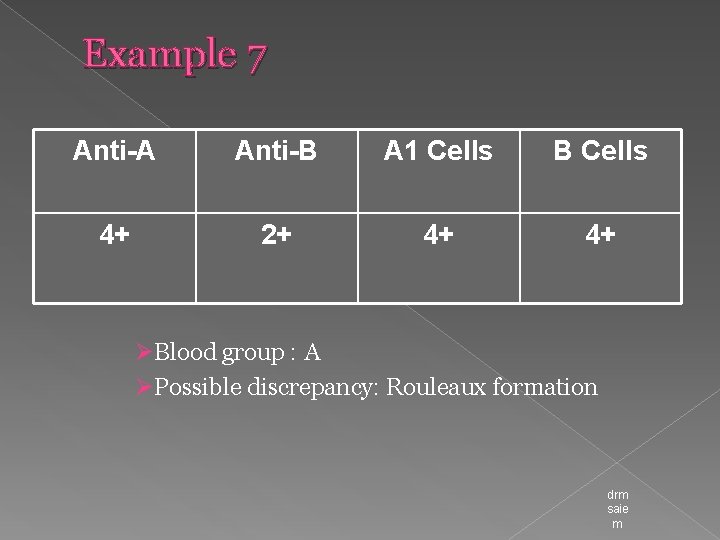
Example 7 Anti-A Anti-B A 1 Cells B Cells 4+ 2+ 4+ 4+ ØBlood group : A ØPossible discrepancy: Rouleaux formation drm saie m

Example 8 Anti-A Anti-B A 1 Cells B Cells 4+ 0 1+ 4+ Blood group: A Ø Possible discrepancy: Missing Ag. Ø drm saie m
- Slides: 82