Discovery of Radioactivity Lead block Radioactive substance Phosphorescent
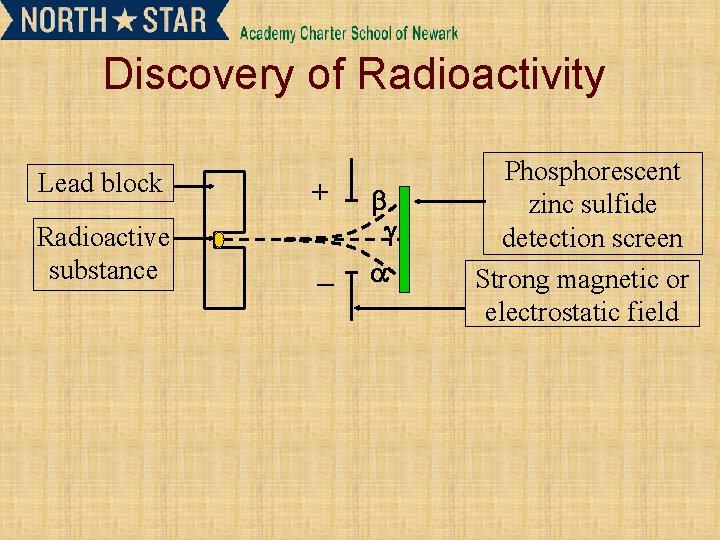
Discovery of Radioactivity Lead block + Radioactive substance – Phosphorescent zinc sulfide detection screen Strong magnetic or electrostatic field

Types of Radioactivity 1) , 2) , 3) 1. Alpha ( ) particles are symbolized as 4 2 He 2. Beta ( ) particles (essentially electrons) are 0 – 1 e 3. Gamma ( ) rays are symbolized as 0 0 • Other symbols: proton = 11 p, neutron = 10 n

Types of Radiation • 1. Alpha Radiation –Helium nuclei are emitted from a U radioactive source –He nuclei are called alpha particles –Contain two protons and two neutrons –Overall +2 charge –Decay Equation: Th He

Types of Radiation • 2. Beta Radiation –Fast-moving electrons created by the break-down of a neutron in an atom –Decay equation: e- C N

Types of Radiation • 3. Gamma Radiation –High-energy electromagnetic radiation given off by a radioisotope

Penetrating power of radioactive emissions. Penetrating power is inversely related to the mass of the emission.

Types of Radiation • 4. positron emission –Positron : • has same mass as an electron but the opposite charge • p + n o + e +

Types of Radiation 5. Electron capture • Capture an inner electron • p+ + e- no + x-ray photon

Now you try some! • Try to write the equation for the alpha decay of thorium-230. • Try to write the equation for the beta decay of Ac-227.

Now you try some! • Try to write the equation for the positron decay of K-38.

Types of Radioactivity • Terms and definitions • “alpha decay” means an particle is given off. • Other: beta decay, • fusion (meaning to bring together), fission (meaning to break apart) • Transmutation when one element changes into another • Artificial. When the radioactivity is induced by bombardment of the nucleus • Half life time it takes for ½ of the original substance to change into something new
Transmutation
Artificial
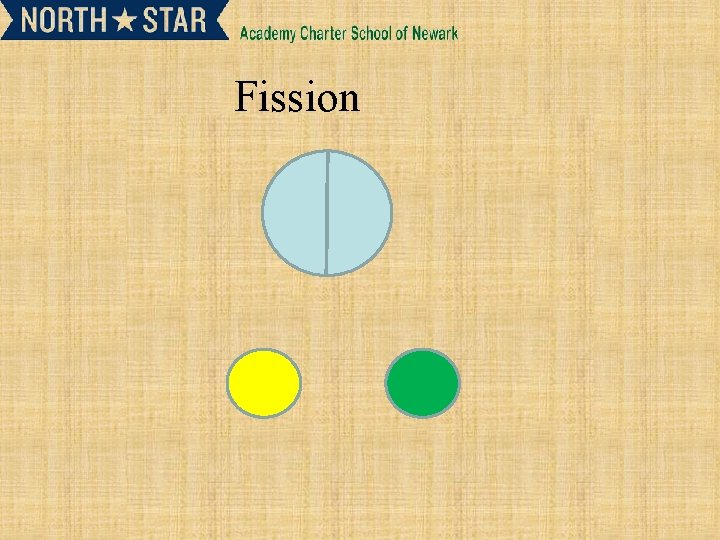
Fission

Fusion

Nuclear equations Q. Write the beta decay for C-14 Q. Write the alpha decay for 209 Po 14 6 C 209 84 0 -1 e Po + 14 7 N 4 205 + He 2 82 Pb Q. Complete this fission reaction 235 1 1 139 + U 0 n 3 0 n + 56 Ba 92 + 94 36 Kr

a) 226 88 b) 84 35 c) 242 94 Ra 4 222 + He 2 86 Rn Br 0 -1 e Pu 4 238 + He 2 92 U 1 14 1 p + 7 N 18 1 e) 8 O + 0 n d) + 84 36 Kr 4 11 2 He + 6 C 0 19 -1 e + 9 F
- Slides: 17