DisclosureDisclaimer All material is intended for your medical







- Slides: 15

Disclosure/Disclaimer • All material is intended for your medical education purposes only 1

Effects Of Exenatide Versus Sitagliptin On Postprandial Glucose, Insulin and Glucagon Secretion, Gastric Emptying, And Caloric Intake: A Randomized, Cross-Over Study Ralph A. De. Fronzo 1; Ted Okerson 2; Prabhakar Viswanathan 2; Xuesong Guan 2; John H. Holcombe 3; Leigh Mac. Conell 2 1 Division of Diabetes, University of Texas Health Science Center, San Antonio, TX, USA; 2 Amylin Pharmaceuticals, Inc. , San Diego, CA, USA; 3 Eli Lilly and Company, Indianapolis, IN, USA

Introduction • This study provides the first head-to-head comparison of incretin-based therapies developed to treat T 2 DM 1: – The GLP-1 receptor agonist, exenatide – The dipeptidyl peptidase-4 (DPP-4) inhibitor, sitagliptin • Exenatide binds directly to the GLP-1 receptor and is at least equipotent, if not more potent, than GLP‑ 12, 3 in: – Stimulation of glucose-dependent secretion of insulin – Restoration of first-phase and second-phase insulin secretion – Suppression of inappropriately elevated postprandial glucagon secretion – Slowing of gastric emptying – Reduction in food intake – Promotion of β-cell proliferation and islet neogenesis from precursor cells in both in vitro and in vivo models of diabetes 1 3

Introduction • The DPP-4 inhibitor sitagliptin inhibits the proteolytic cleavage of GLP-1 in the circulation by binding to and reducing the activity of the DPP-4 enzyme by approximately 80%, increasing the concentration of endogenous GLP-1 by approximately 2 -fold, thereby: – Reducing plasma glucagon and increasing insulin and C-peptide concentrations, and increasing β -cell mass and function 1 4

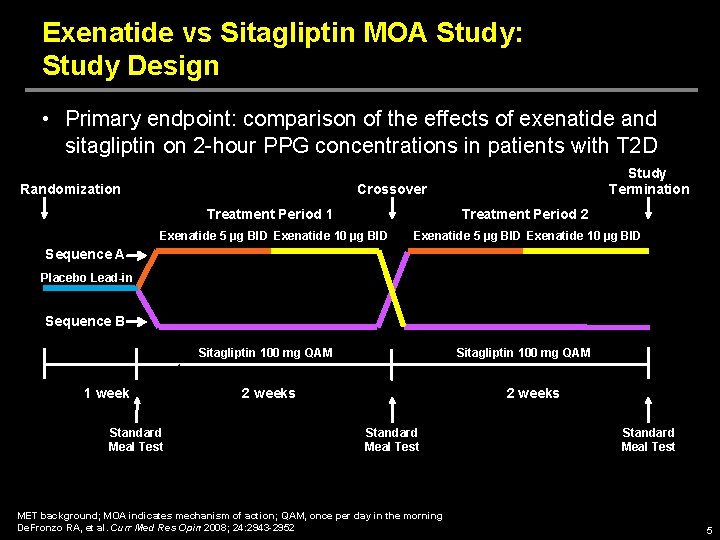
Exenatide vs Sitagliptin MOA Study: Study Design • Primary endpoint: comparison of the effects of exenatide and sitagliptin on 2 -hour PPG concentrations in patients with T 2 D Randomization Study Termination Crossover Treatment Period 1 Treatment Period 2 Exenatide 5 µg BID Exenatide 10 µg BID Sequence A Placebo Lead-in Sequence B Sitagliptin 100 mg QAM 1 week Standard Meal Test Sitagliptin 100 mg QAM 2 weeks Standard Meal Test MET background; MOA indicates mechanism of action; QAM, once per day in the morning De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 Standard Meal Test 5

Demographic and Baseline Characteristics Evaluable Patients (n = 61) Sex, Female/Male (%) 54/46 Age (y) 54 ± 9 Race, Caucasian/Black/Hispanic (%) 30/8/62 Height (cm) 167. 0 ± 9. 9 Body Weight (kg) 91. 5 ± 18. 8 BMI (kg/m 2) 32. 6 ± 5. 1 A 1 C (%) 8. 5 ± 1. 2 Duration of Diabetes (y) 7± 5 Fasting Triglycerides (mg/d. L) 166 ± 93 FPG (mg/d. L) 178 ± 48 2 -hr PPG (mg/d. L) 245 ± 65 Patients with T 2 D; MET background; Mean ± SD, unless otherwise indicated; BMI indicates body mass index De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 6

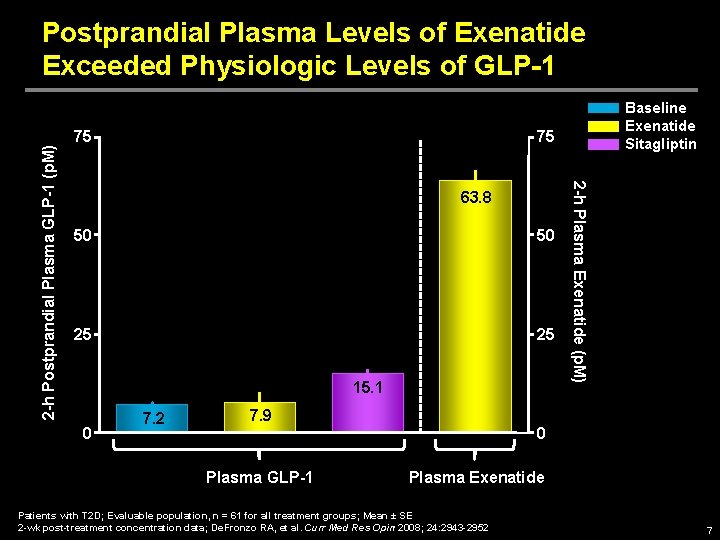
75 75 63. 8 50 50 25 25 15. 1 0 Baseline Exenatide Sitagliptin 7. 2 2 -h Plasma Exenatide (p. M) 2 -h Postprandial Plasma GLP-1 (p. M) Postprandial Plasma Levels of Exenatide Exceeded Physiologic Levels of GLP-1 7. 9 0 Plasma GLP-1 Plasma Exenatide Patients with T 2 D; Evaluable population, n = 61 for all treatment groups; Mean ± SE 2 -wk post-treatment concentration data; De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 7

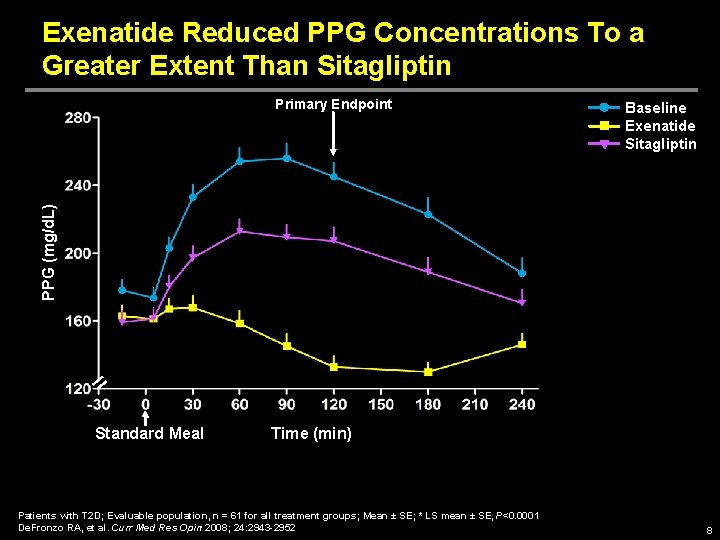
Exenatide Reduced PPG Concentrations To a Greater Extent Than Sitagliptin Baseline Exenatide Sitagliptin PPG (mg/d. L) Primary Endpoint Standard Meal Time (min) Patients with T 2 D; Evaluable population, n = 61 for all treatment groups; Mean ± SE; * LS mean ± SE, P<0. 0001 De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 8

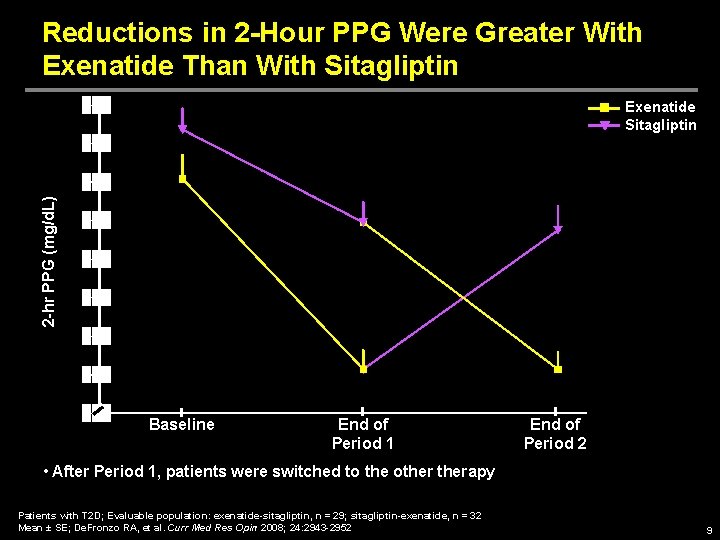
Reductions in 2 -Hour PPG Were Greater With Exenatide Than With Sitagliptin 2 -hr PPG (mg/d. L) Exenatide Sitagliptin Baseline End of Period 1 End of Period 2 • After Period 1, patients were switched to the otherapy Patients with T 2 D; Evaluable population: exenatide-sitagliptin, n = 29; sitagliptin-exenatide, n = 32 Mean ± SE; De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 9

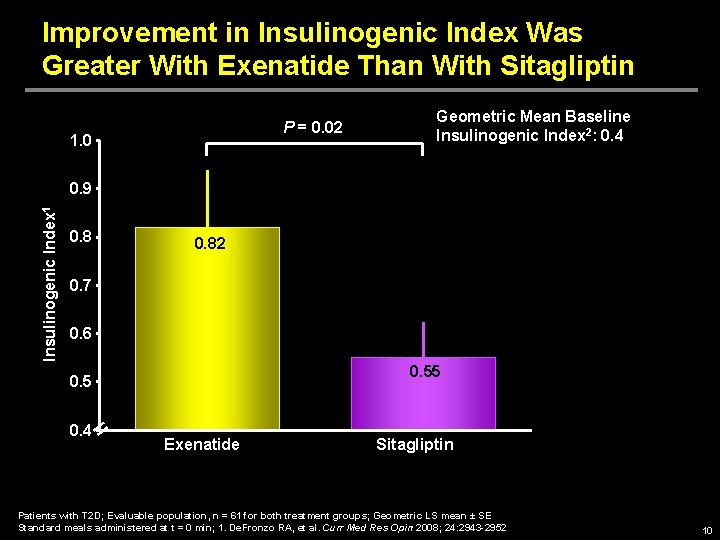
Improvement in Insulinogenic Index Was Greater With Exenatide Than With Sitagliptin P = 0. 02 1. 0 Geometric Mean Baseline Insulinogenic Index 2: 0. 4 Insulinogenic Index 1 0. 9 0. 82 0. 7 0. 6 0. 55 0. 4 Exenatide Sitagliptin Patients with T 2 D; Evaluable population, n = 61 for both treatment groups; Geometric LS mean ± SE Standard meals administered at t = 0 min; 1. De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 10

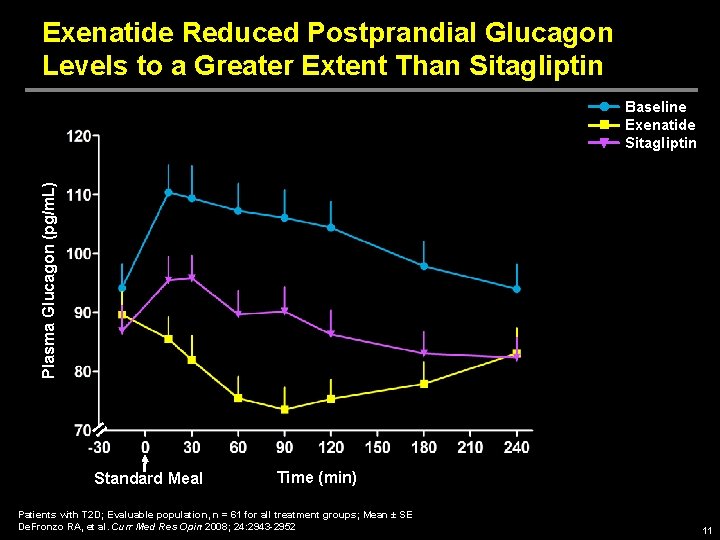
Exenatide Reduced Postprandial Glucagon Levels to a Greater Extent Than Sitagliptin Plasma Glucagon (pg/m. L) Baseline Exenatide Sitagliptin Standard Meal Time (min) Patients with T 2 D; Evaluable population, n = 61 for all treatment groups; Mean ± SE De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 11

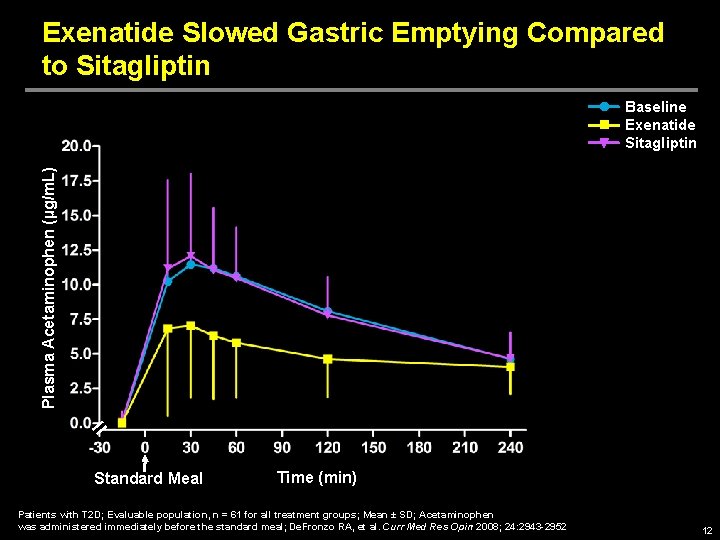
Exenatide Slowed Gastric Emptying Compared to Sitagliptin Plasma Acetaminophen (µg/m. L) Baseline Exenatide Sitagliptin Standard Meal Time (min) Patients with T 2 D; Evaluable population, n = 61 for all treatment groups; Mean ± SD; Acetaminophen was administered immediately before the standard meal; De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 12

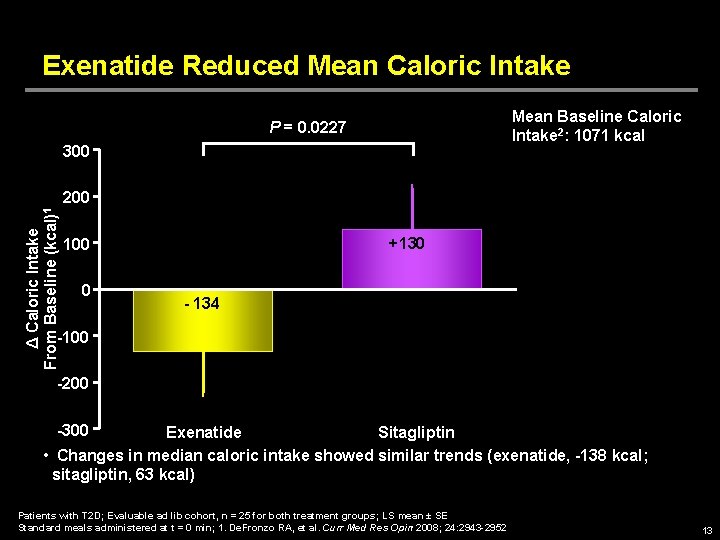
Exenatide Reduced Mean Caloric Intake Mean Baseline Caloric Intake 2: 1071 kcal P = 0. 0227 300 Δ Caloric Intake From Baseline (kcal)1 200 +130 100 0 -100 - 134 -200 -300 Exenatide Sitagliptin • Changes in median caloric intake showed similar trends (exenatide, -138 kcal; sitagliptin, 63 kcal) Patients with T 2 D; Evaluable ad lib cohort, n = 25 for both treatment groups; LS mean ± SE Standard meals administered at t = 0 min; 1. De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 13

Exenatide vs Sitagliptin: Safety • Both exenatide and sitagliptin were generally well tolerated 1 – Overall incidence of AEs was low 2 • Mild-to-moderate nausea and vomiting were the most frequently reported events 1 – Nausea: exenatide, 34%; sitagliptin, 12% – Vomiting: exenatide, 24%; sitagliptin, 3% • Study withdrawals due to AEs 1 – 2 patients on exenatide (nausea and moderate hypoglycemia symptoms) – 1 patient on sitagliptin (dizziness) • No major hypoglycemic events were reported 1 Patients with type 2 diabetes; ITT population, N = 95; AE indicates adverse event 1. De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952; 2. Data on file, Amylin Pharmaceuticals, Inc. 14

Exenatide vs Sitagliptin MOA Study: Conclusions • 2 -hr PPG concentration was significantly reduced with exenatide compared with sitagliptin • Compared with sitagliptin treatment, exenatide treatment led to – Greater reductions in § PPG concentrations over time § Postprandial glucose excursions § Postprandial glucagon levels – Improved insulinogenic index – Delayed gastric emptying – Decreased caloric intake • Changes in FPG concentrations were comparable with exenatide and sitagliptin • Both exenatide and sitagliptin were generally well tolerated De. Fronzo RA, et al. Curr Med Res Opin 2008; 24: 2943 -2952 15