disclosure This presentation has been developed for an

disclosure This presentation has been developed for an educational purpose and represents independent evaluations and opinions of the authors and contributors independently from the operating company sponsoring the present symposium. Before using any medical device, review all relevant package inserts with particular attention to the indications, contraindications, warnings and precautions, and steps for use of the device(s). Michael Rinaldi, MD is compensated by and presenting on behalf of Cordis, and must present information in accordance with applicable regulatory requirements. 100511435 03/18 1

Polymer & Drug Science: Recent Advancements in Understanding Michael Rinaldi, MD Sanger Heart & Vascular Institute Carolinas Medical Center Charlotte, NC

Elu. NIR™ Device Components System component Stent Material Component description Manufactured from L 605 Co. Cr alloy Drug Substance Ridaforolimus, 1. 1µg/mm 2 (total drug load 34 -219µg depending on stent length) Inactive Ingredients Blend of Carbo. Sil 20 55 D and Poly n-Butyl Methacrylate (PBMA) Coating 100511435 03/18 3

Ridaforolimus Drug Substance • Molecular formula: C 53 H 84 NO 14 P • Molecular weight: 990. 22 g/mol • Non-prodrug analog of Rapamycin • Produced by reaction of Rapamycin with dimethyl-phosphinic chloride (single chemical transformation step) 100511435 03/18 4
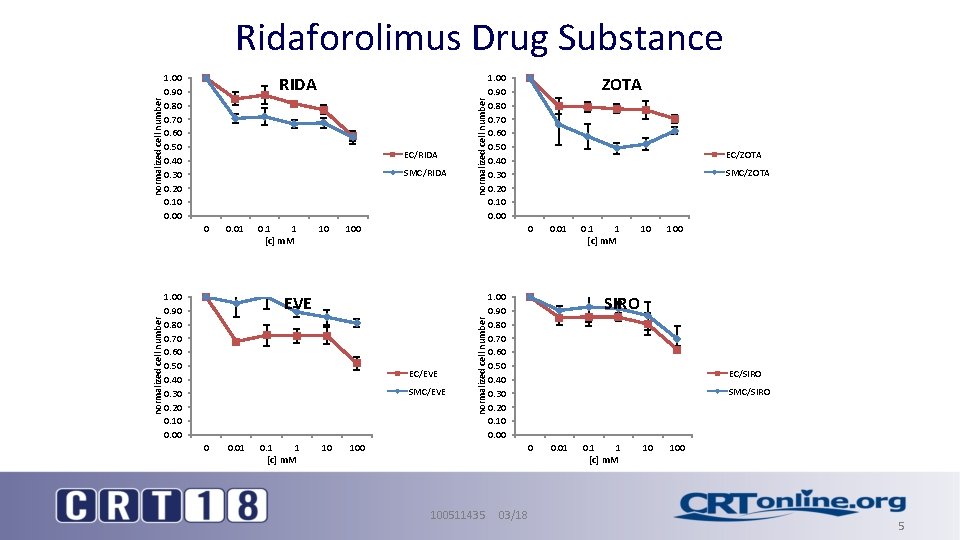
1. 00 0. 90 0. 80 0. 70 0. 60 0. 50 0. 40 0. 30 0. 20 0. 10 0. 00 EC/RIDA SMC/RIDA normalized cell number 0 0. 01 1. 00 0. 90 0. 80 0. 70 0. 60 0. 50 0. 40 0. 30 0. 20 0. 10 0. 00 0. 1 1 [c] m. M 10 normalized cell number RIDA EC/EVE SMC/EVE 0. 01 0. 1 1 [c] m. M 10 ZOTA EC/ZOTA SMC/ZOTA 0 EVE 0 1. 00 0. 90 0. 80 0. 70 0. 60 0. 50 0. 40 0. 30 0. 20 0. 10 0. 00 100 normalized cell number Ridaforolimus Drug Substance 100 1. 00 0. 90 0. 80 0. 70 0. 60 0. 50 0. 40 0. 30 0. 20 0. 10 0. 00 0. 1 1 [c] m. M 10 100 SIRO EC/SIRO SMC/SIRO 0 100511435 0. 01 03/18 0. 01 0. 1 1 [c] m. M 10 100 5
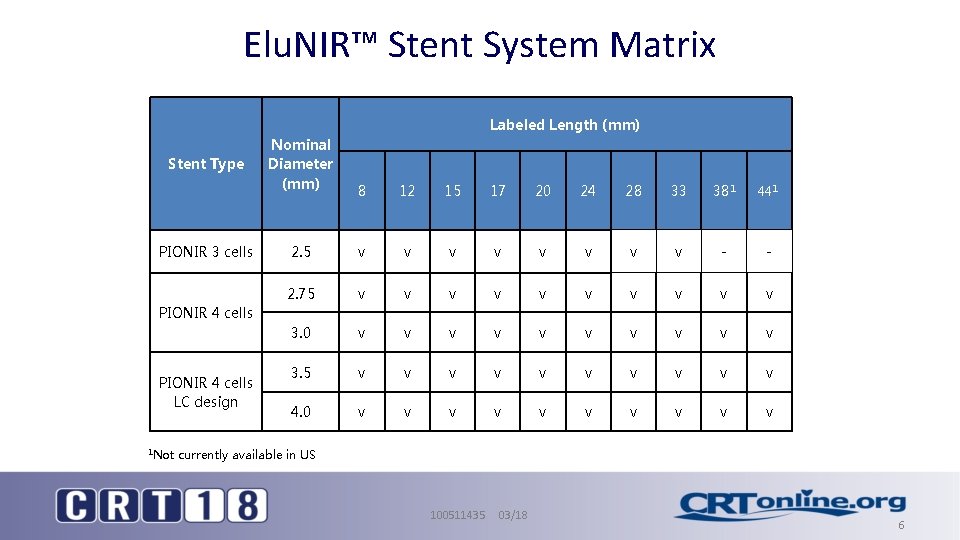
Elu. NIR™ Stent System Matrix Labeled Length (mm) Stent Type PIONIR 3 cells Nominal Diameter (mm) 8 12 15 17 20 24 28 33 381 441 2. 5 v v v v - - 2. 75 v v v v v 3. 0 v v v v v 3. 5 v v v v v 4. 0 v v v v v PIONIR 4 cells LC design 1 Not currently available in US 100511435 03/18 6
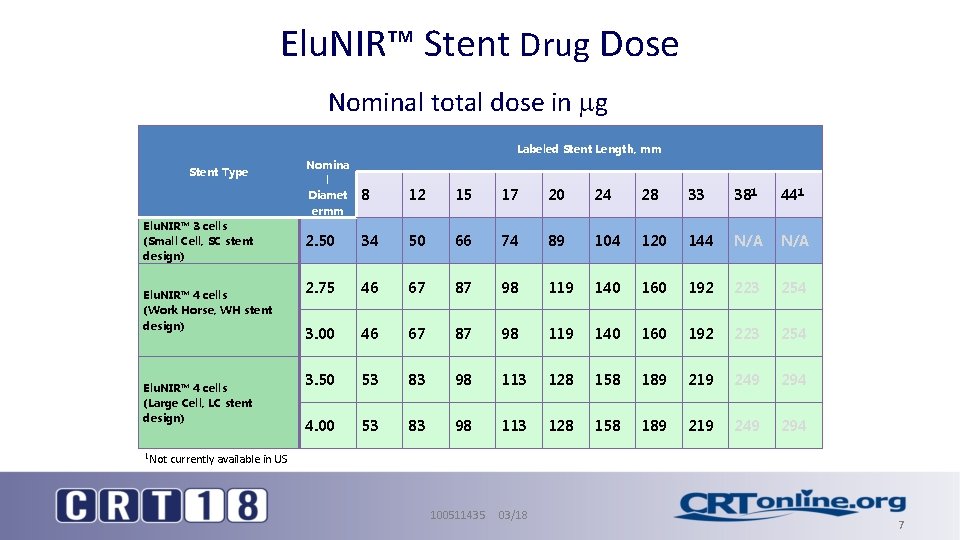
Elu. NIR™ Stent Drug Dose Nominal total dose in g Labeled Stent Length, mm Stent Type Elu. NIR™ 3 cells (Small Cell, SC stent design) Elu. NIR™ 4 cells (Work Horse, WH stent design) Elu. NIR™ 4 cells (Large Cell, LC stent design) 1 Not Nomina l Diamet ermm 8 12 15 17 20 24 28 33 381 441 2. 50 34 50 66 74 89 104 120 144 N/A 2. 75 46 67 87 98 119 140 160 192 223 254 3. 00 46 67 87 98 119 140 160 192 223 254 3. 50 53 83 98 113 128 158 189 219 249 294 4. 00 53 83 98 113 128 158 189 219 249 294 currently available in US 100511435 03/18 7

Elastomeric Durable Polymer • Carbo. Sil® - Thermoplastic Silicone-Polycarbonate Urethane Elastomer – Elastic, non-cracking coating – Enables predictable and uniform release of drug – Enables controlled and uniform drug deposition to the vessel wall – Designed to reduce surface irregularities and deformations 100511435 03/18 8
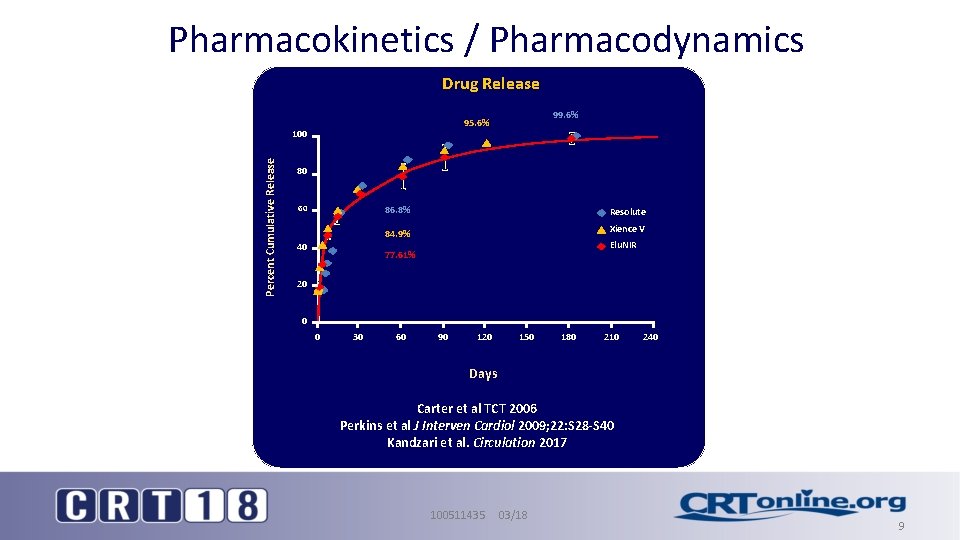
Pharmacokinetics / Pharmacodynamics Drug Release 100 Percent Cumulative Release 99. 6% 95. 6% 80 60 86. 8% Resolute Xience V 84. 9% 40 Elu. NIR 77. 61% 20 0 0 30 60 90 120 150 180 210 240 Days Carter et al TCT 2006 Perkins et al J Interven Cardiol 2009; 22: S 28 -S 40 Kandzari et al. Circulation 2017 100511435 03/18 9
![Drug Deposition Arterial Drug Concentration [ng/mg] 25 Resolute 20 Elu. NIR 15 10 5 Drug Deposition Arterial Drug Concentration [ng/mg] 25 Resolute 20 Elu. NIR 15 10 5](http://slidetodoc.com/presentation_image_h2/811f79cb1b86e69a18ea7e7e24ab2cd1/image-10.jpg)
Drug Deposition Arterial Drug Concentration [ng/mg] 25 Resolute 20 Elu. NIR 15 10 5 0 0 20 40 60 80 100 Days Yazdani et al J Invasive Cardiol 2013 100511435 03/18 10
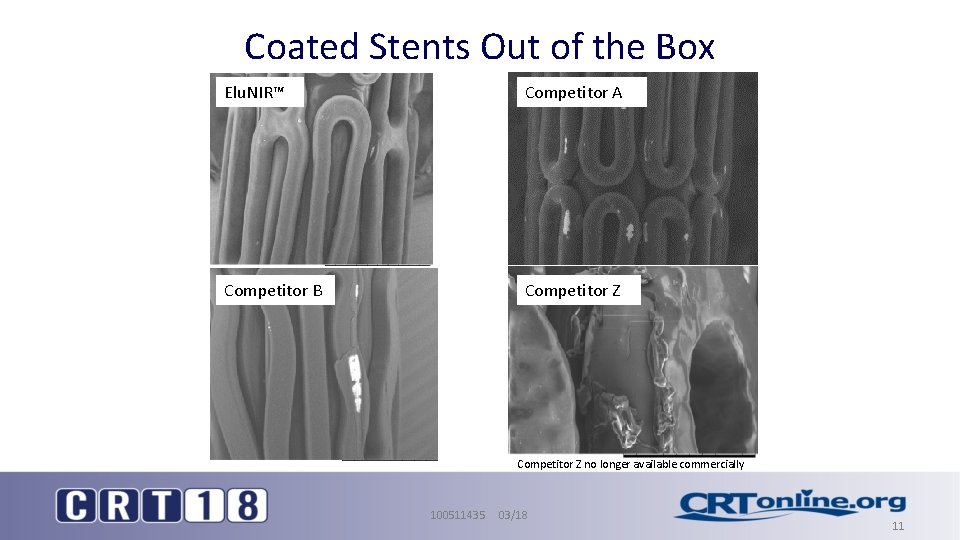
Coated Stents Out of the Box Elu. NIR™ Competitor A Competitor B Competitor Z no longer available commercially 100511435 03/18 11

Competitor A after 50 days in plasma 100511435 03/18 12
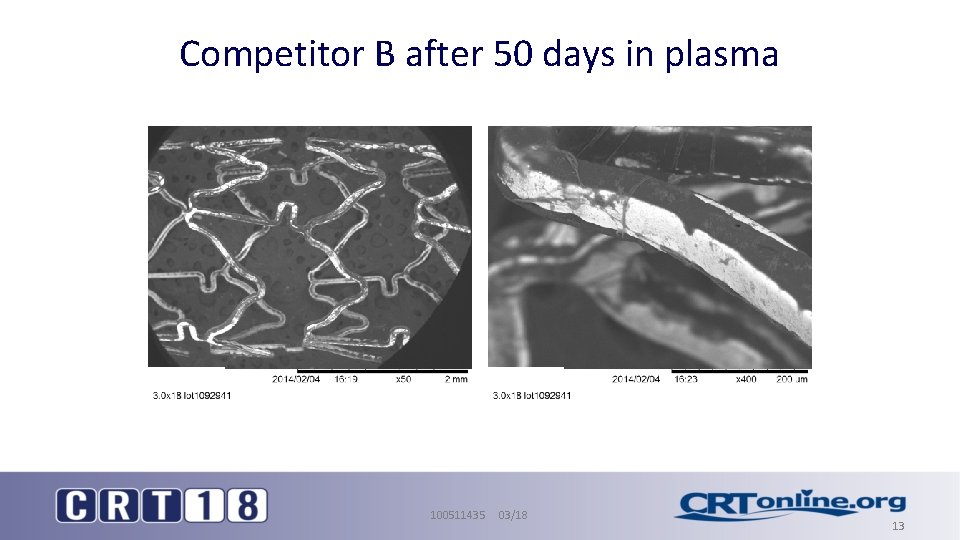
Competitor B after 50 days in plasma 100511435 03/18 13

Competitor C after 50 days in plasma 100511435 03/18 14

Competitor D after 45 days in plasma 100511435 03/18 15

Competitor E after 45 days in plasma 100511435 03/18 16
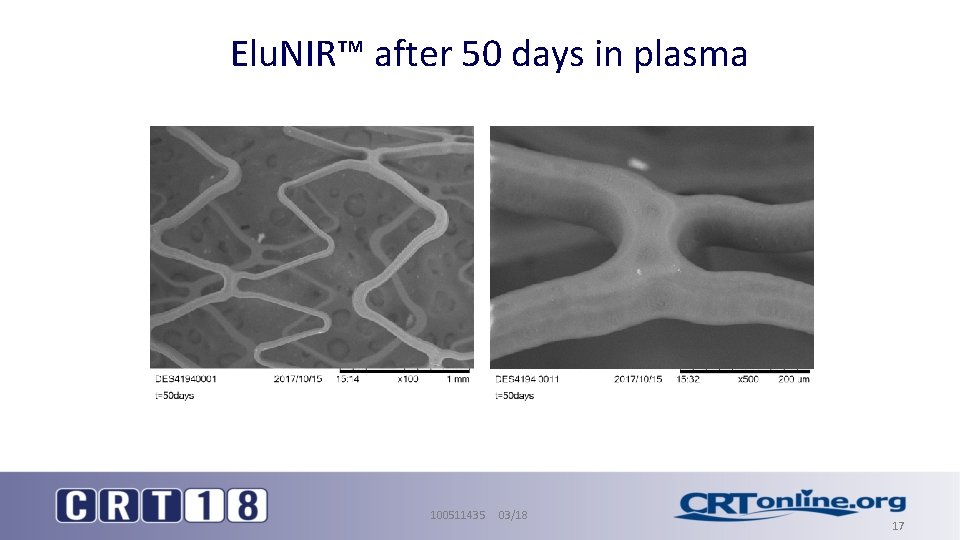
Elu. NIR™ after 50 days in plasma 100511435 03/18 17
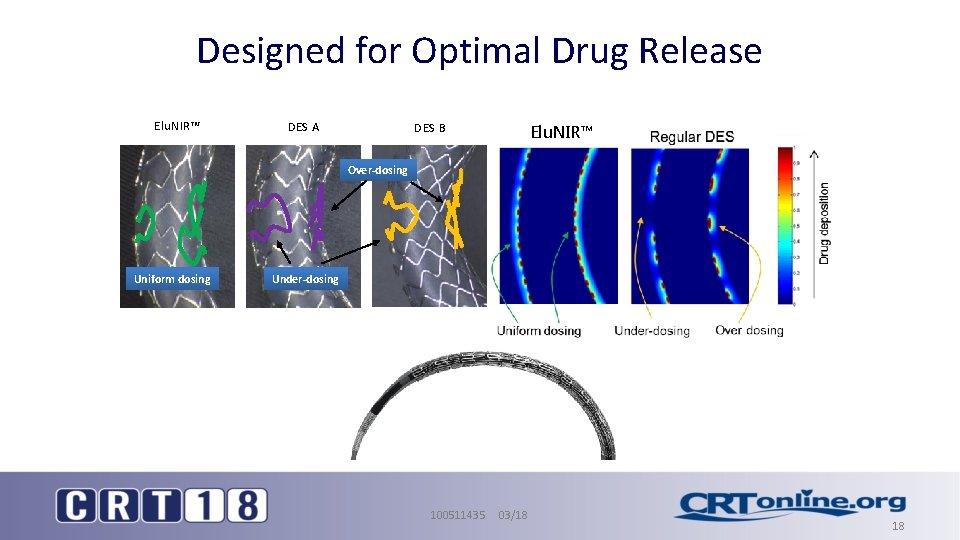
Designed for Optimal Drug Release Elu. NIR™ DES A DES B Elu. NIR™ Over-dosing Uniform dosing Under-dosing 100511435 03/18 18
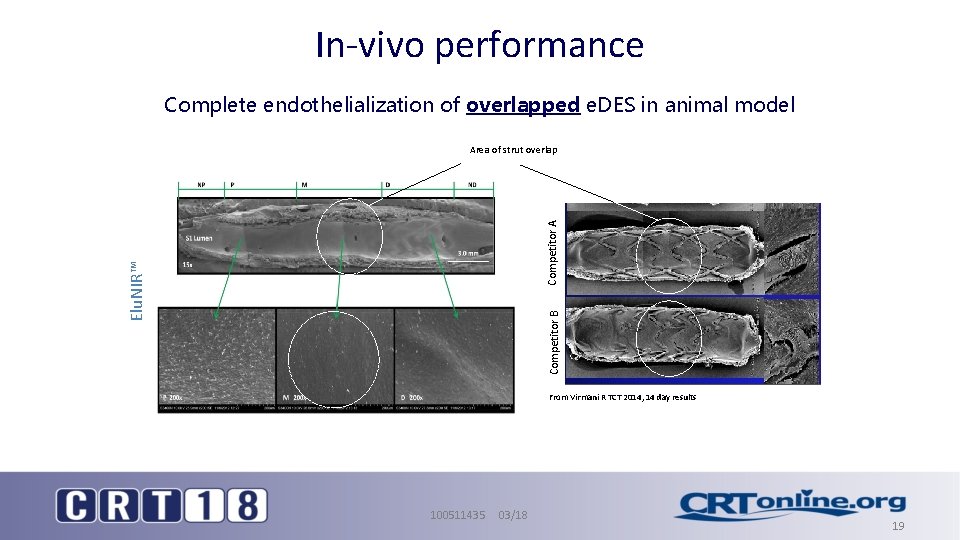
In-vivo performance Complete endothelialization of overlapped e. DES in animal model Competitor B Elu. NIR™ Competitor A at 30 Areadays of strut overlap From Virmani R TCT 2014, 14 day results Elu. NIR™ Stent BMS Control 100511435 03/18 19

Summary • Elu. NIR™ Stent has key design elements aimed at optimizing drug deposition – Controlled and uniform deposition of Ridaforolimus – Use of a novel elastomeric polymer designed to reduce surface irregularities and deformations, providing a controlled drug-elution – Dual pattern strut design maintains uniform scaffolding in a bend • All of these elements contribute to an in-vivo performance demonstrating endothelialization and intimal maturation to be on par with BMS controls. 1 1. From Virmani R TCT 2014, 14 day results 100511435 03/18 20

Thank You
- Slides: 21