Disclaimer Panion BF Biotech Inc The Company Disclaimer


Disclaimer Panion & BF Biotech Inc. (The Company) Disclaimer • The presentation and the relevant information mentioned in this material, including operating performance, financial performance and the business outlook, have been compiled from both internal and external resources. • These forward looking statements involve known and unknown risks, uncertainties and other factors, including price variation, competition, global economy, exchange rate movement and market demand, which may cause actual results to differ materially from those implied by such forward-looking statements. • This Presentation should not be considered as the giving of investment advice by the Company or any of its shareholders, directors, officers, agents, employees or advisers. Each party to whom this Presentation is made available must make its own independent assessment of the Company after making such investigations and taking such advice as may be deemed necessary. In particular, any estimates or projections or opinions contained herein necessarily involve significant elements of subjective judgment, analysis and assumptions and each recipient should satisfy itself in relation to such matters. • The forward looking statements expressed in this material reflect the Company’s current view about the future as of today. The Company is not responsible for any updates if there any changes in the future.





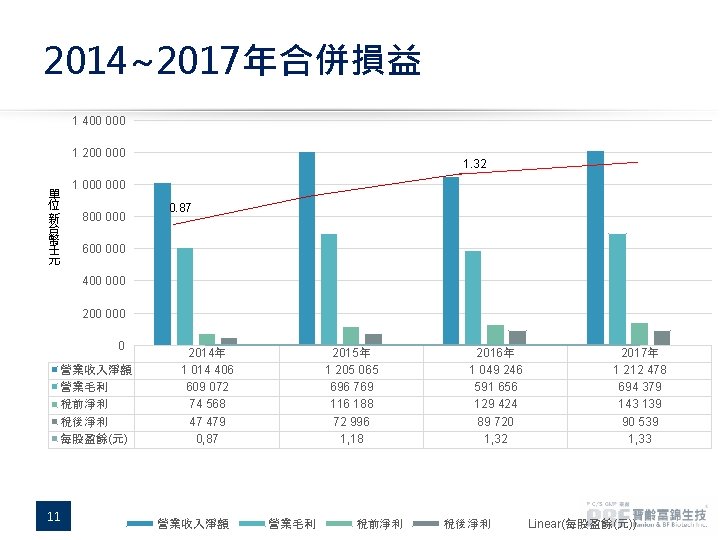
2014~2017年合併損益 1 400 000 單位 新台幣千元 1 200 000 單 位 新 台 幣 千 元 1. 32 1 000 800 0. 87 600 000 400 000 200 0 營業收入淨額 營業毛利 稅前淨利 稅後淨利 每股盈餘(元) 11 2014年 1 014 406 609 072 74 568 47 479 0, 87 營業收入淨額 2015年 1 205 065 696 769 116 188 72 996 1, 18 營業毛利 稅前淨利 2016年 1 049 246 591 656 129 424 89 720 1, 32 稅後淨利 2017年 1 212 478 694 379 143 139 90 539 1, 33 Linear(每股盈餘(元))


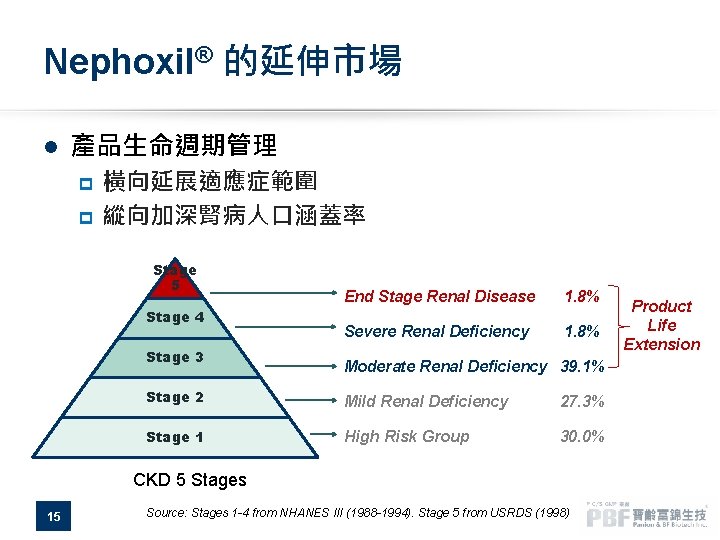
Nephoxil® 的延伸市場 l 產品生命週期管理 橫向延展適應症範圍 p 縱向加深腎病人口涵蓋率 p Stage 5 Stage 4 Stage 3 End Stage Renal Disease 1. 8% Severe Renal Deficiency 1. 8% Moderate Renal Deficiency 39. 1% Stage 2 Mild Renal Deficiency 27. 3% Stage 1 High Risk Group 30. 0% CKD 5 Stages 15 Source: Stages 1 -4 from NHANES III (1988 -1994). Stage 5 from USRDS (1998) Product Life Extension

Under preparation Ongoing Under development A. 新適應症擴充: IDA-NDD-CKD l 2017年 11月8日,USFDA核准新適應症擴充(s. NDA),Ferric citrate 成 為美國透析前慢性腎病市場中唯一治療缺鐵性貧血口服製劑。 ~$2. 8 to $4. 9 B Market 16 ~$2. 2 B Market
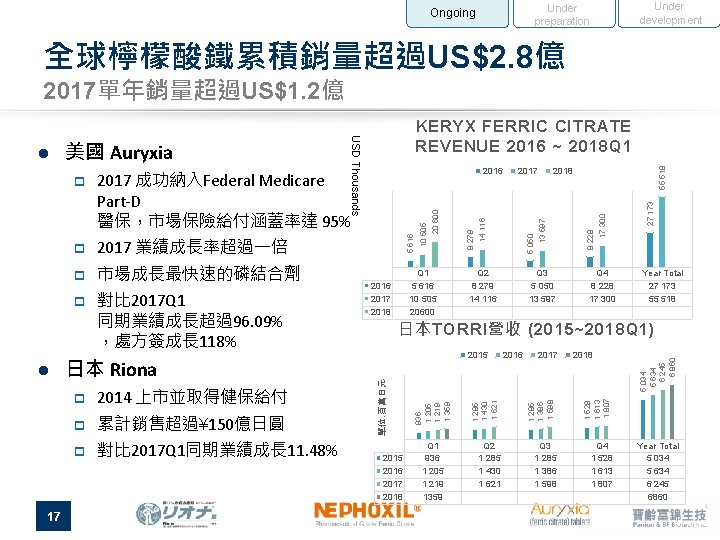
Under development Under preparation Ongoing 全球檸檬酸鐵累積銷量超過US$2. 8億 2017單年銷量超過US$1. 2億 17 2014 上市並取得健保給付 p 累計銷售超過¥ 150億日圓 p 對比 2017 Q 1同期業績成長 11. 48% 55 518 27 173 17 300 13 597 5 050 14 116 20 600 8 228 Q 4 8 228 17 300 Year Total 27 173 55 518 日本TORRI營收 (2015~2018 Q 1) 2015 2016 2017 2018 Q 1 936 1 205 1 219 1359 2016 2017 2018 Q 2 1 285 1 430 1 621 Q 3 1 285 1 386 1 598 Q 4 1 528 1 613 1 807 5 034 5 634 6 245 6 860 p Q 3 5 050 13 597 1 528 1 613 1 807 日本 Riona Q 2 8 279 14 116 2018 1 285 1 386 1 598 l 對比 2017 Q 1 同期業績成長超過96. 09% ,處方簽成長 118% Q 1 5 616 10 505 20600 2016 2017 2018 8 279 市場成長最快速的磷結合劑 2017 1 285 1 430 1 621 p 5 616 2017 業績成長率超過一倍 936 1 205 1 219 1 359 p p 2016 2017 成功納入Federal Medicare Part-D 醫保,市場保險給付涵蓋率達 95% 單位: 百萬日元 p 10 505 美國 Auryxia USD Thousands l KERYX FERRIC CITRATE REVENUE 2016 ~ 2018 Q 1 Year Total 5 034 5 634 6 245 6860
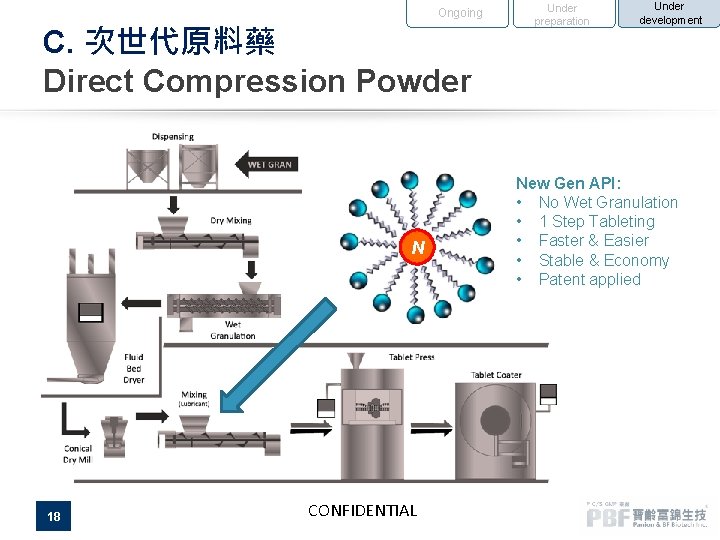
Ongoing C. 次世代原料藥 Direct Compression Powder N 18 CONFIDENTIAL Under preparation Under development New Gen API: • No Wet Granulation • 1 Step Tableting • Faster & Easier • Stable & Economy • Patent applied

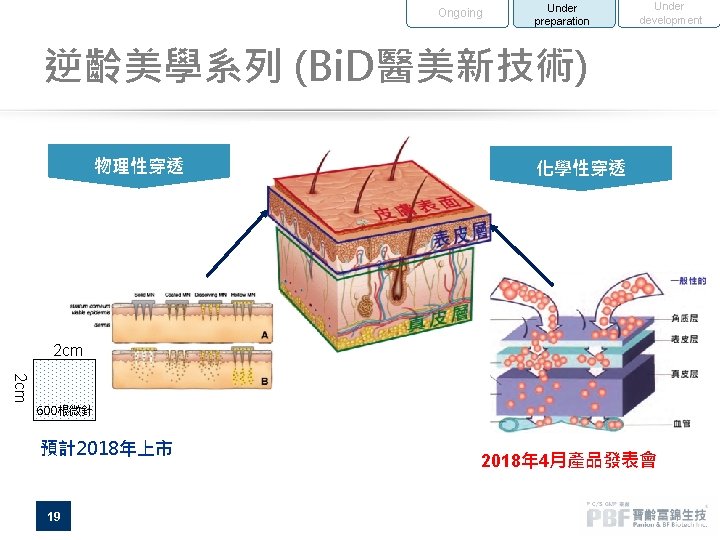
Ongoing Under preparation Under development 逆齡美學系列 (Bi. D醫美新技術) 物理性穿透 化學性穿透 2 cm 600根微針 預計2018年上市 19 2018年 4月產品發表會
Ongoing Under preparation 檢驗試劑事業部門 改良性 + 突破性產品 Under development

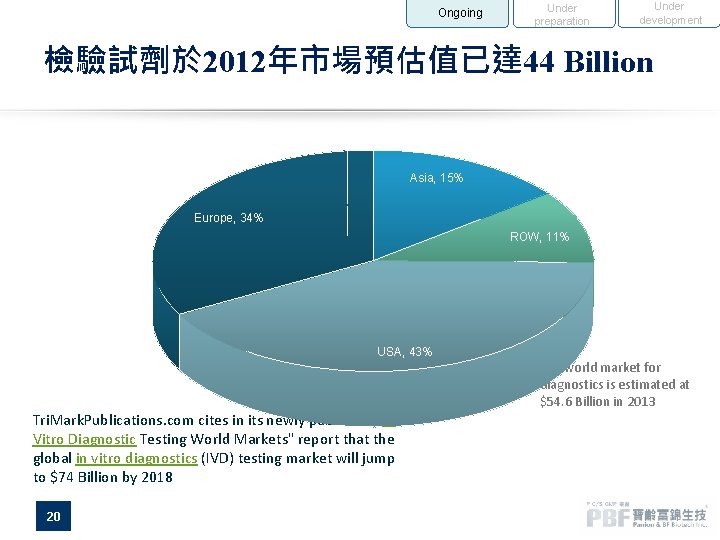
Ongoing Under preparation Under development 檢驗試劑於 2012年市場預估值已達 44 Billion Asia, 15% Europe, 34% ROW, 11% USA, 43% Tri. Mark. Publications. com cites in its newly published "In Vitro Diagnostic Testing World Markets" report that the global in vitro diagnostics (IVD) testing market will jump to $74 Billion by 2018 20 The world market for diagnostics is estimated at $54. 6 Billion in 2013

Ongoing Under preparation Diagnostic Products 檢驗試劑產品 三個呼吸道 三個消化道 21 Under development
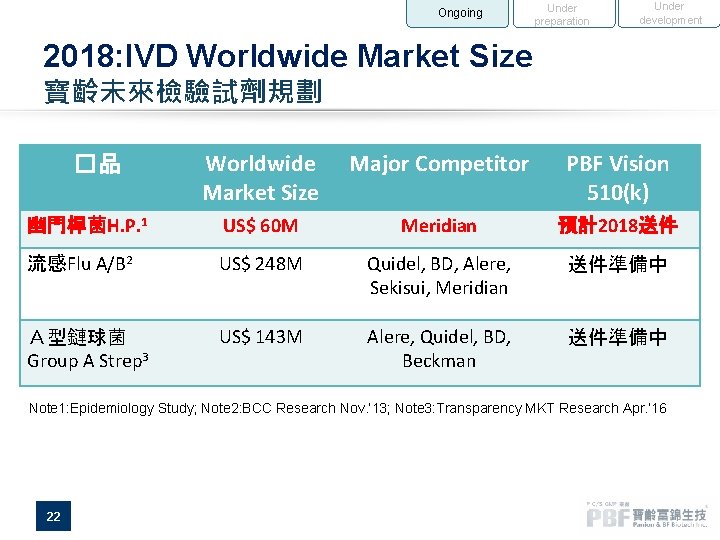
Ongoing Under preparation Under development 2018: IVD Worldwide Market Size 寶齡未來檢驗試劑規劃 Worldwide Market Size Major Competitor PBF Vision 510(k) 幽門桿菌H. P. 1 US$ 60 M Meridian 預計2018送件 流感Flu A/B 2 US$ 248 M Quidel, BD, Alere, Sekisui, Meridian 送件準備中 A型鏈球菌 Group A Strep 3 US$ 143 M Alere, Quidel, BD, Beckman 送件準備中 �品 Note 1: Epidemiology Study; Note 2: BCC Research Nov. ’ 13; Note 3: Transparency MKT Research Apr. ’ 16 22



- Slides: 31