Director Italian Branch Cagliari Regional Director for Europe

Director Italian Branch Cagliari Regional Director for Europe Director Ian Donald School for Invasive Procedures WHO Collaborating Centre for Community Control of Hereditary Diseases INVASIVE VS NON-INVASIVE PRENATAL DIAGNOSTIC PROCEDURES Giovanni Monni 12 th TURKISH GYNECOLOGY AND OBSTETRICS CONGRESS Antalya, 15 th – 19 th Maggio 2014

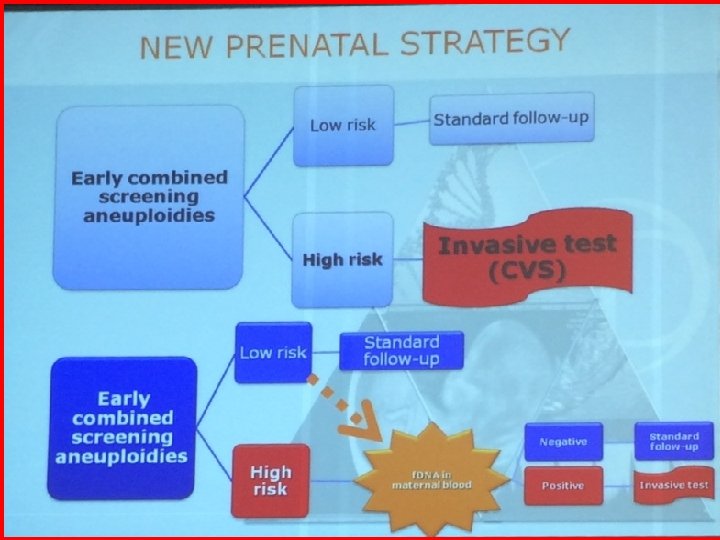
DILEMMAS TO AVOID GENETIC DISORDERS IN THE NEWBORNS Screening based on maternal age alone? Prenatal invasive procedures? First or second trimester ultrasound and biochemical screening? Standard karyotype? a. CGH analysis? Preimplantation genetic diagnosis? Diagnostic ultrasonography (1 st-2 nd trimester)? Fetal cell free-fetal DNA (cff- DNA) in maternal blood (general or contingent) ?
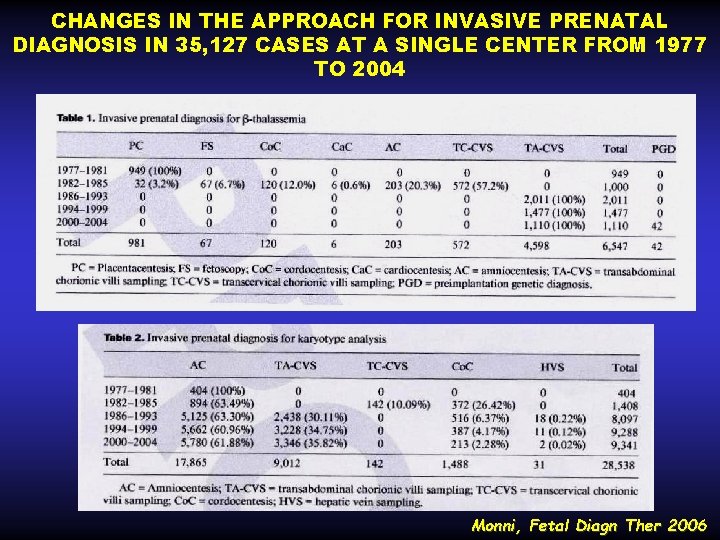
CHANGES IN THE APPROACH FOR INVASIVE PRENATAL DIAGNOSIS IN 35, 127 CASES AT A SINGLE CENTER FROM 1977 TO 2004 • DIAGNOSI SEMPRE Più PRECOCE Monni, Fetal Diagn Ther 2006
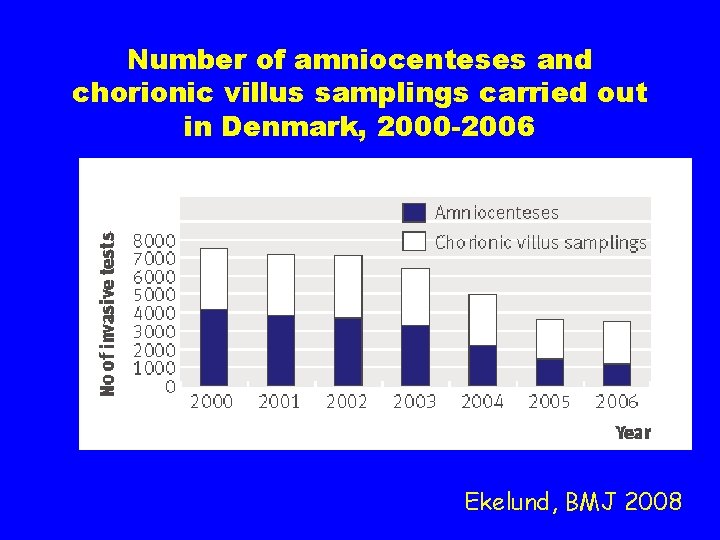
Number of amniocenteses and chorionic villus samplings carried out in Denmark, 2000 -2006 Ekelund, BMJ 2008
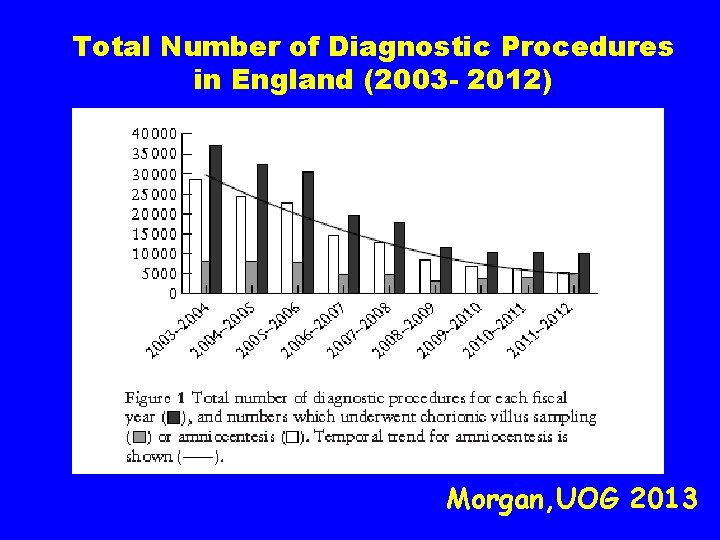
Total Number of Diagnostic Procedures in England (2003 - 2012) Morgan, UOG 2013

UK national policy study of aneuploidy screening after the implementation of the combined test 1. Reduction of false positive rate from 6% to 3% without significant change of DR of Down Syndrome 2. Progressive reduction in the number of screen-positive cases 3. Significant reduction in the number of invasive prenatal diagnostic procedures Morgan, Ultrsound Obstet Gynecol 2013

UK NATIONAL POLICY STUDY ANEUPLOIDY SCREENING The odds of the fetus being affected after a positive combined test in the first trimester were much greater than were the odds based on advanced maternal age alone (1: 20 vs 1: 75). So a significantly higher probability of an invasive test would confirm an abnormal fetal karyotype. Morgan, Ultrsound Obstet Gynecol 2013

REDUCTION IN THE FETAL NUMBER OF INVASIVE PROCEDURES PERFORMED FOR PRENATAL KARYOTYPE Redistribution of the proportion of procedures performed by amnio and CVS - Denmark: in 2006 CVS in 66% of cases - UK: in 2003 Amnio/CVS 3: 1 in 2011 Amnio/CVS 1: 1 Monni, Zoppi, Ultrasound Obstet Gynecol 2013: Opinion

FIRST TRIMESTER EUROPEAN NATIONAL POLICY FOR PRENATAL DOWN SYNDROME SCREENING Denmark (BMJ 2008) and UK (UOG 2013) Studies - Decrease in Fetal Loss due to a reduction in invasive diagnostic procedures Earlier Diagnosis of Chromosomal Aneuploidies Monni, Zoppi, Ultrasound Obstet Gynecol 2013: Opinion

FIRST TRIMESTER SCREENING AND INVERSION OF THE PYRAMID OF PRENATAL CARE Nicolaides, Prenat Diagn 2012
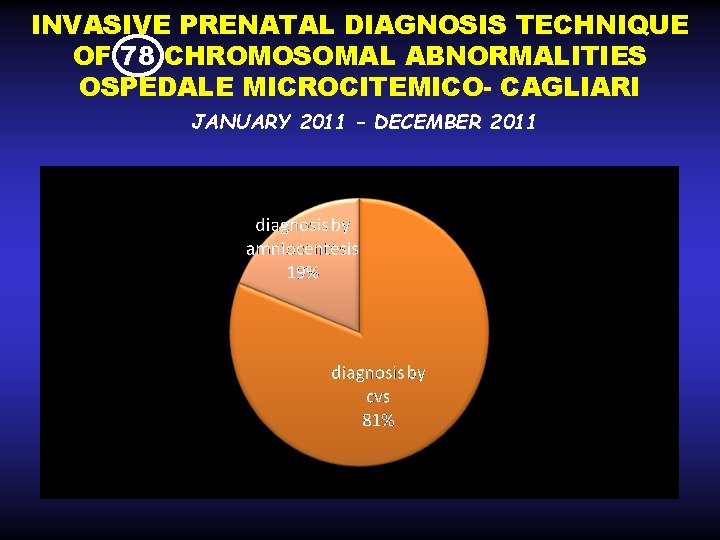
INVASIVE PRENATAL DIAGNOSIS TECHNIQUE OF 78 CHROMOSOMAL ABNORMALITIES OSPEDALE MICROCITEMICO- CAGLIARI JANUARY 2011 – DECEMBER 2011

Distribution of number of fellows for CVS training at the Ospedale Microcitemico - Cagliari Period No. % *1983 - 1996 42 28 **1997 - 2012 109 72 151 100 * BEFORE NT SCREENING ** AFTER NT SCREENING
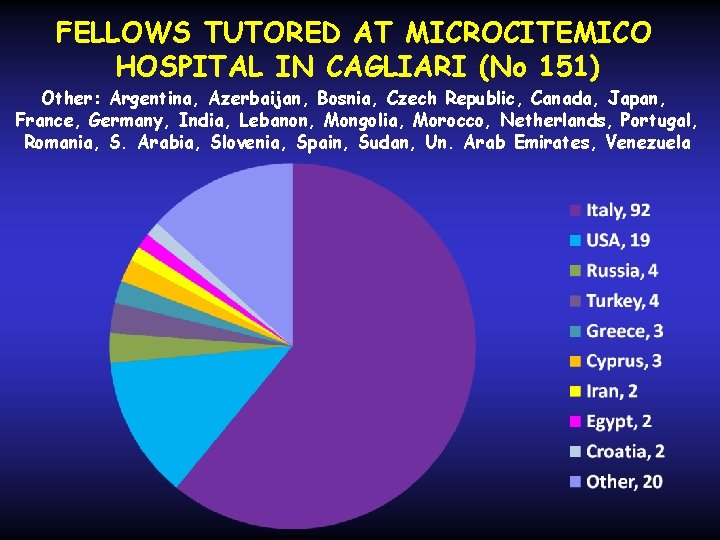
FELLOWS TUTORED AT MICROCITEMICO HOSPITAL IN CAGLIARI (No 151) Other: Argentina, Azerbaijan, Bosnia, Czech Republic, Canada, Japan, France, Germany, India, Lebanon, Mongolia, Morocco, Netherlands, Portugal, Romania, S. Arabia, Slovenia, Spain, Sudan, Un. Arab Emirates, Venezuela



NEW LABORATORY TECHNIQUES • Fluorescent in situ hybridization (FISH) • Amplification of polymorphic chromosome-specific markers by polymerase chain reaction (PCR) • Most laboratories offer a rapid test (PCR or FISH) to detect trisomy 21, 18, 13 and sex chromosome aneuploidies, as well as tissue culture to provide a full karyotype • Array comparative genomic hybridization (a-CGH): in cases of multiple congenital abnormalities at ultrasound or for clinical diagnosis?

Advantages of array Comparative Genomic Hybridization (a. CGH) or Chromosomal Microarray Analysis (CMA) • a. CGH allows detection of smaller pathogenic chromosomal variants that are undetectable using standard cytogenetic analyses (G-band karyotyping)

DISADVANTAGES OF ACGH • a. CGH does not allow detection of balanced chromosomal rearrangements triploidy and some instances of mosaicism • The biggest challenge presented by a. CGH is the detection of chromosomal variants of unknown clinical significance (VOUS)

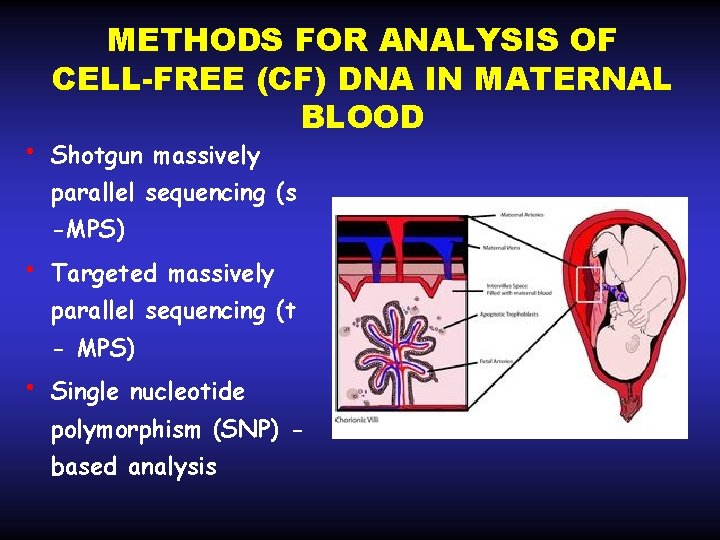
• METHODS FOR ANALYSIS OF CELL-FREE (CF) DNA IN MATERNAL BLOOD Shotgun massively parallel sequencing (s -MPS) • Targeted massively parallel sequencing (t - MPS) • Single nucleotide polymorphism (SNP) based analysis

CFDNA ANALYSIS FOR T 21: A META-ANALYSIS (18 CITATIONS 2011 - 2013) • Individual studies: – Detection Rate (DR) ranges: 94. 4 -100% – False Positive Rate (FPR) ranges: 0 - 2. 05% • Pooled weighted: – DR: 99. 0% (95% CI 98. 2 - 99. 6) – FPR: 0. 08% (95% CI 0. 03 - 0. 14) Gil et Nicolaides, Fetal Diag Ther 2014
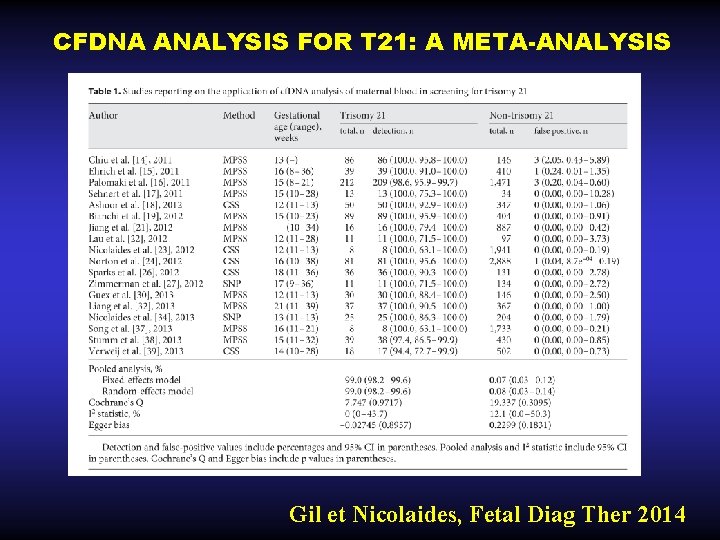
CFDNA ANALYSIS FOR T 21: A META-ANALYSIS Gil et Nicolaides, Fetal Diag Ther 2014

CFDNA ANALYSIS FOR T 18, 13, MONOSOMY X : A META-ANALYSIS Trisomy 18 Trisomy 13 Monosomy X Detection rate False positive rate 96. 8% 0. 15% (95% CI 94. 5 - 98. 4) (95% CI 0. 08 - 0. 25) 92. 1% 0. 20% (95% CI 85. 9 - 96. 7) (95% CI 0. 04 - 0. 46) 88. 6% 0. 12% The poor performance of cf. DNA analysis in screening for (95% CI 83. 093. 1) (95% 0. 05 - 0. 24) trisomy 13 and monosomy X could be due to the CI highly variable amplification of chromosome X and 13 because of a lower guanosine- cytosine content Gil et Nicolaides, Fetal Diag Ther 2014

CFDNA ANALYSIS FOR SEX CHROMOSOME ANEUPLOIDIES OTHER THAN MONOSOMY X • Pooled weighted: – DR: 93. 8% (95% CI 85. 9 - 98. 7) – FPR: 0. 12% (95% CI 0. 02 - 0. 28) Gil et Nicolaides, Fetal Diag Ther 2014

CFDNA ANALYSIS FOR TRIPLOIDY • • Diandric (paternal): • Placenta enlarged and partially molar • NT enlarged • Free- beta h. CG very high (10 times higher Digynic (maternal): • Placenta very small • Fetus severely growth restricted • Normal NT • Free- beta h. CG and PAPP-A very than normal) low The SNP method for cf. DNA testing is the only one at present that can detect triploidy because it analyses allele distributions 4 out 8 cases of diandric triploidy have been detected, and suspicion raised for a case of diagynic triploidy Utility of cf. DNA as first-line method of screening because identification of triploidy would be beneficial (diandric triploidy can cause maternal complications including early- onset preeclamsia and choriocarcinoma) Gil et Nicolaides, Fetal Diag Ther 2014

LIMITATIONS OF CFDNA TESTING • Failure to provide results • Receiving • Cost results in 1 - 2 weeks

FAILURE TO PROVIDE RESULTS In 1 - 5% of cases no results is given after first sampling • • • Problems with sample collection or with transportation to the laboratory (on repeat sampling result is obtained in about 100%) Assay failure (on repeat sampling result is obtained in about 75%) Low fetal fraction (on repeat sampling result is obtained in about 50%); if it is a consequence of maternal obesity this problem is difficult to overcome Gil et Nicolaides, Fetal Diag Ther 2014

RECEIVING RESULTS IN 1 - 2 WEEKS • Average interval 10 calendar days • In 95 - 98% of cases a result • available within 14 days In 2% of cases a result may not be available in less than 3 -4 weeks Such delay may reverse the beneficial shift in screening and diagnosis of aneuploidies from the second to the first trimester Gil et Nicolaides, Fetal Diag Ther 2014

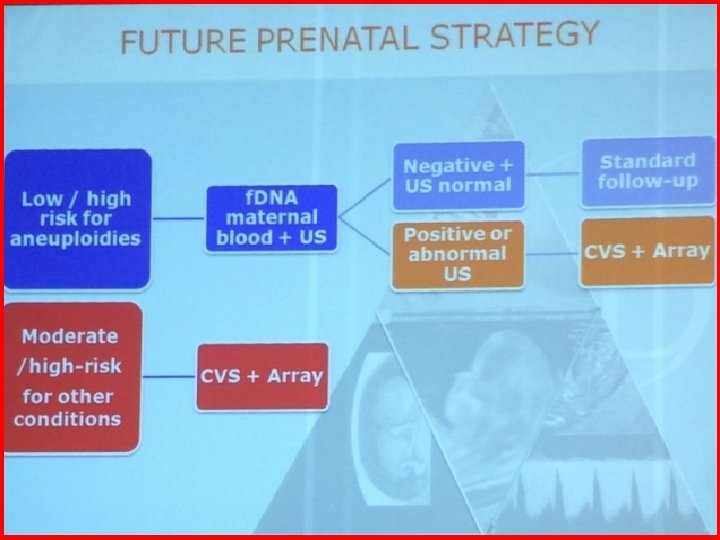
MODELS FOR CLINICAL IMPLEMENTATION OF CFDNA TESTING IN MATERNAL BLOOD • Routine screening for whole population • Contingent screening based on the result of first trimester combined test

MODELS FOR CLINICAL IMPLEMENTATION OF CFDNA TESTING AS FIRST-LINE METHOD FOR ALL PREGNANCIES • 10 • 12 • • • weeks, maternal blood to all weeks first trimester us Expected: 99% DR of trisomy 21 95% DR of trisomy 18 and 13 1% Invasive testing rate Gil et Nicolaides, Fetal Diag Ther 2014

MODELS FOR CLINICAL IMPLEMENTATION OF CFDNA TESTING AS CONTINGENT SCREENING HIGH RISK GROUP • • Maternal blood in the high risk group (> 1: 100) Expected: 86% DR of tris. 21 89% DR of tris. 18 /13 0. 4% Invasive test. rate cf. DNA testing could not detect other aneuploidies Gil et Nicolaides, Fetal Diag Ther 2014

• • MODELS FOR CLINICAL IMPLEMENTATION OF CFDNA TESTING AS CONTINGENT SCREENING INTERMEDIATE RISK GROUP Maternal blood in the Intermediate Risk Group (>1: 11<1: 2, 500) Expected: 97. 6% DR of tris. 21 98. 1% DR of tris. 18/13 0. 8% Invasive test. rate cf. DNA testing could not detect other aneuploidies Gil et Nicolaides, Fetal Diag Ther 2014

• • • PRENATAL NONINVASIVE DIAGNOSIS FOR MONOGENIC DISEASE: ACTUALLY VALIDATED USE Fetal sex determination (X-linked diseases in order to avoid invasive procedure in female fetuses) or for congenital adrenal hyperplasia (CAH) for therapeutic options RH blood group, D antigen Paternal inherited autosomal dominant diseases or de- novo after ultrasound suspicion (chondrodysplasias) SIGU 2014, Document on the indications of use of performing noninvasive prenatal research

PRENATAL NONINVASIVE DIAGNOSIS FOR MONOGENIC DISEASE: NOT YET VALIDATED USE • Autosomal • X recessive diseases linked diseases • Autosomal origin dominat diseases of maternal SIGU 2014, Document on the indications of use of performing noninvasive prenatal research

MAIN FEATURES OF FREE DNA IN MATERNAL PLASMA • • • Free DNA is always present in peripheral blood with a magnitude of between 145 and 201 bp Pregnancy causes an increase in the size of circulating DNA of maternal origin and a progressive increase in the concentration of Fetal DNA that is smaller The origin of circulating Fetal DNA in maternal plasma is due to placental apoptotic processes of the syncytium trophoblast The Fetal DNA is during pregnancy. circulating plasma with the maternal present since the 7 th week of pregnancy and increases In 10 weeks increases to about 5 or 10% of the total DNA. The fraction of fetal tissue correlates negatively weight The presence of Fetal DNA in maternal plasma is no longer detected two hours after giving birth. The average half-life of 16. 3 minutes (range 4 -30 minutes)
FEASIBILITY STUDY OF -THALASSEMIA NIPD IN SARDINIA BY BENCHTOP NEXT GEN SEQUENCING APPARATUS (PGM LIFE TECHNOLOGIES)
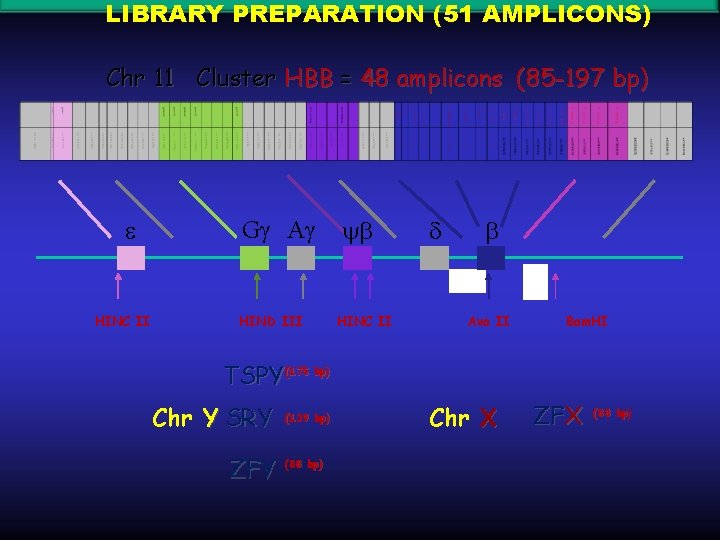
LIBRARY PREPARATION (51 AMPLICONS) Chr 11 Cluster HBB = 48 amplicons (85 -197 bp) HINC II G A HIND III HINC II Ava II Bam. HI TSPY (175 bp) Chr Y SRY ZFY (139 bp) (88 bp) Chr X ZFX (88 bp)

TAKE HOME MESSAGES (1) • • • Maternal age should no longer be the sole criterium for set the parental choice of invasive prenatal diagnosis First trimester combined screening reduces the number of invasive prenatal diagnostic procedures After a positive combined test, a significantly high probability of an invasive test would confirm an abnormal fetal karyotype First trimester combined test induces reversing the traditional pyramid of prenatal care Educational organizations have faced new challenges in providing training for invasive procedures

TAKE HOME MESSAGES (2) 1) a. CGH is not a substitute for conventional karyotyping; 2) a. CGH should be used for specific diagnostic purposes in selected pregnancies and not for general screening in all pregnancies;

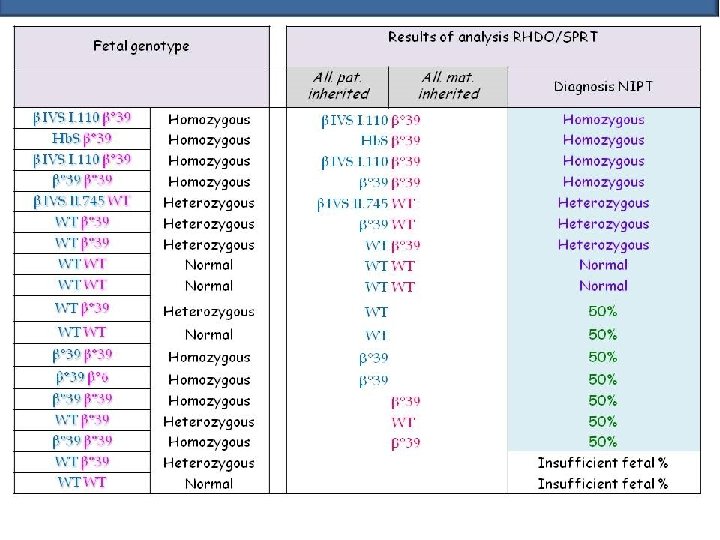
TAKE HOME MESSAGES (3) 1) cff- DNA for NIPT has the role of a screening test 2) Evidence from high risk population 3) Necessity of implementation of cff. DNA in low risk series 4) Genetic counselling is mandatory before and after NIPT

NON-INVASIVE PRENATAL TEST (NIPT) The expectations regarding cff-DNA for fetal genetic anomalies are very high because it may have the potential to change the landscape of prenatal diagnosis. However, to the disappointment of many, cff-DNA does not have the ability to function as a diagnostic test but is considered at present time as a “super” screening test. Monni, Journal of Perinatal Medicine 2014
- Slides: 46