DIRECT MEASUREMENTS OF RAT INTRACRANIAL PRESSURE Saratov State









- Slides: 12

DIRECT MEASUREMENTS OF RAT INTRACRANIAL PRESSURE Saratov State University SFM 2020 V. B. Ageev, I. V. Fedosov, I. A. Blokhina, O. V. Semyachkina-Glushkovskaya

Abstract Accurate and reproducible measurements of intracranial pressure during sleep are а key to the understanding of brain clearing through the circulation of cerebrospinal fluid. Existing methods are based on the use of a flexible catheter or fiber light guide sensor that makes sealing difficult and reduces measurement accuracy. We have proposed a method that uses two integral solid state sensors, one of which measures atmospheric pressure, and the other directly closes the opening in the animal's skull to measure the cerebrospinal fluid pressure in the subarachnoid space. The paper presents the results of measuring the intracranial pressure in a rat during sleep and wakefulness; it was found that intracranial pressure increases upon awakening, and also changes when the head and trunk are raised, when the animal turns over on its back. The proposed method provides high sensitivity; minimizes measurement errors; and practically eliminates trauma to the brain during the installation and operation of the sensor; our ICP data are in good numerical agreement with the data of other authors.

Significance Previously, attempts were made to record the dynamics of intracranial pressure using recording system with an external sensor [1]. Such a system incorporates flexible catheter filled with a fluid that transfers intracranial pressure to the sensor. The systems cannot directly measure intracranial pressure, but it can quite successfully register changes pressure changes caused by catheter deformations. At the same time, there is a high probability to injure animal's brain during the installation of a flexible tube in the skull. The volume of the catheter may be comparable to the volume of the animal's skull [2]. The low rigidity and tightness of the hydraulic system creates the potential for leaks. The pressure in the system may vary from external impacts on the system [3].

Aim The purpose of our work is to eliminate the shortcomings mentioned above. We will get rid of the system with flexible tubes and attach the pressure sensor to the animal's skull for direct measurement of intracranial pressure, with minimal brain injury while attaching the sensor to the animal's skull.

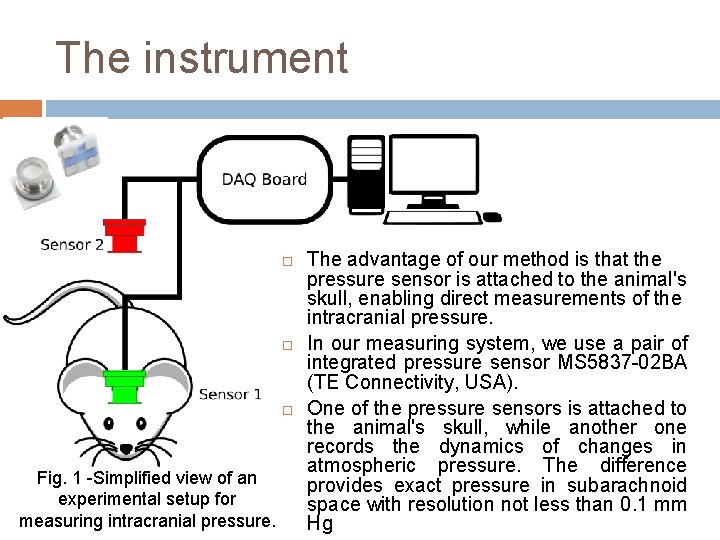
The instrument Fig. 1 -Simplified view of an experimental setup for measuring intracranial pressure. The advantage of our method is that the pressure sensor is attached to the animal's skull, enabling direct measurements of the intracranial pressure. In our measuring system, we use a pair of integrated pressure sensor MS 5837 -02 BA (TE Connectivity, USA). One of the pressure sensors is attached to the animal's skull, while another one records the dynamics of changes in atmospheric pressure. The difference provides exact pressure in subarachnoid space with resolution not less than 0. 1 mm Hg

Method The pressure recording started when the pressure sensors had atmospheric pressure and showed a pressure difference of 0 mm Hg. , after 5 minutes, the sensor was fixed and the mounting screws were tightened. When the rat was turned over on its back, the pressure rose to 9 mm Hg (the rat was turned over at the 11 th minute, see figure 2). there was also a good increase in pressure when lifting the back of the body and a decrease when lifting the head. (Jump up and down immediately after the explosion in the 11 th minute) at rest, the pressure was restored. The sensitivity of the sensor is 0. 05 mm. Hg. Under anesthesia, the ICP was about 3. 5 mm Hg (a sensor on the top of the skull) and remained unchanged for about an hour. At the 75 th minute, there was a sharp jumpthe animal was lifted and placed in an electroencephalographic playpen. Before waking up, after 140 minutes (17 h 40 min), a Series of jumps, after waking up (160 min, 18 00), the pressure rose to 6 mm Hg. when the rat was active, pressure jumps were observed.

Results We obtained data on changes in intracranial pressure during sleep and wake. In this case, the moment when the animal wakes up is expressed by an increase in ICP. We have experimentally established that intracranial pressure changes from the hydrostatic pressure of the brain, when lifting the head and trunk, as well as when turning the animal on its back. The data we obtained numerical ICP in good agreement with the data published in the article of Derek M. Nusbaum [1]. Fig. 2 - General view of the experimental setup and the integrated Fig. 3 -An animal with pressure sensor installed

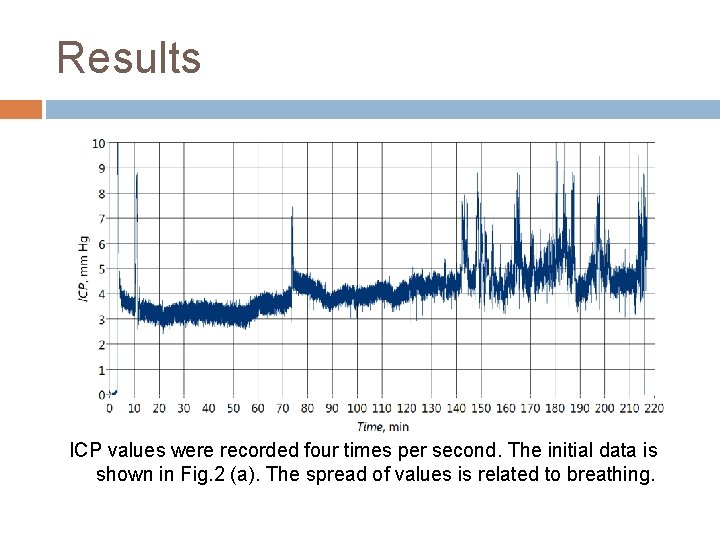
Results ICP values were recorded four times per second. The initial data is shown in Fig. 2 (a). The spread of values is related to breathing.

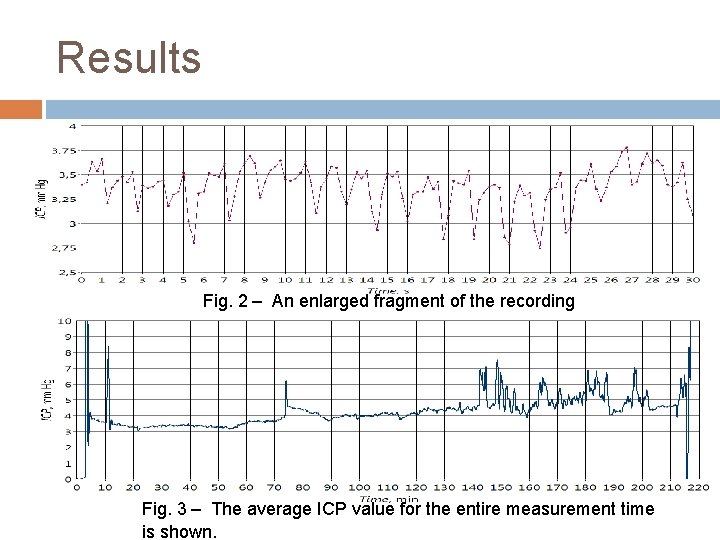
Results Fig. 2 – An enlarged fragment of the recording (b). Fig. 3 – The average ICP value for the entire measurement time is shown.

Conclusion The proposed method provides high sensitivity; minimizes measurement errors; and practically eliminates trauma to the brain during the installation and operation of the sensor; our ICP data are in good numerical agreement with the data of other authors. The pressure recording started when the pressure sensors had atmospheric pressure and showed a pressure difference of 0 mm Hg. , after 5 minutes, the sensor was fixed and the mounting screws were tightened. When the rat was turned over on its back, the pressure rose to 9 mm Hg (the rat was turned over at the 11 th minute, see figure 2). there was also a good increase in pressure when lifting the back of the body and a decrease when lifting the head. (Jump up and down immediately after the explosion in the 11 th minute) at rest, the pressure was restored. The sensitivity of the sensor is 0. 05 mm. Hg. Under anesthesia, the ICP was about 3. 5 mm Hg (a sensor on the top of the skull) and remained unchanged for about an hour. At the 75 th minute, there was a sharp jump-the animal was lifted and placed in an electroencephalographic playpen. Before waking up, after 140 minutes (17 h 40 min), a Series of jumps, after waking up (160 min, 18 00), the pressure rose to 6 mm Hg. when the rat was active, pressure jumps were observed.

Acknowledge Russian Science Foundation 20 -15 -00090 RF Goverment Grant 075 -15 -2019 -1885

References 1. Nusbaum D. M. , Wu S. M. , Frankfort B. J. Elevated intracranial pressure causes optic nerve and retinal ganglion cell degeneration in mice //Experimental eye research. – 2015. – Т. 136. – С. 38 -44. 2. Guild S. J. , Mc. Bryde F. D. , Malpas S. C. Recording of intracranial pressure in conscious rats via telemetry //Journal of Applied Physiology. – 2015. – Т. 119. – №. 5. – С. 576 -581. 3. Feiler S. et al. Standardized induction of subarachnoid hemorrhage in mice by intracranial pressure monitoring //Journal of neuroscience methods. – 2010. – Т. 190. – №. 2. – С. 164 -170.