Diphtheria Tetanus Pertussis Med Ch 401 Lecture 3


























- Slides: 27

Diphtheria, Tetanus, Pertussis Med. Ch 401 Lecture 3 17 May 06 KL Vadheim Lecture 3 1

Adjuvant • Substances that enhance the immune response • Two categories: – vehicles – immunomodulators 17 May 06 KL Vadheim Lecture 3 2

Adjuvants functioning as vehicles I • Human use: – Alum compounds • Aluminum hydroxide and phosphate • the only licensed adjuvants in U. S. – MF 59 • Oil and water emulsion • Marketed in Europe 17 May 06 KL Vadheim Lecture 3 3

Adjuvants functioning as vehicles II • Animal use: – Freund’s Complete Adjuvant (CFA) • dessicated Mycobacterium butyricum, mineral oil and an emulsifying agent, mannide monooleate • causes potentially secere local inflammatory lesions, chronic granulomas, abscesses, and tissue sloughs. Injected into the murine footpad, it can cause chronic lameness and arthritis; injected intraperitoneally, it can cause peritonitis – Freund’s Incomplete Adjuvant • Mineral oil and Mannide monooleate • Fewer side effects, adequate for boosting 17 May 06 KL Vadheim Lecture 3 4

Immunomodulatory Adjuvants • Purified Protein Derivative (PPD) • Lipopolysaccharide (LPS; bacterial endotoxin) • Lipid A - lipid portion of LPS • Cholera toxin B subunit • Cp. G 17 May 06 KL Vadheim Lecture 3 5

Diphtheria vaccine • Detoxified bacterial, protein toxin • Injectable, IM administration • Toxigenic Corynebacterium diphtheriae (infected with b phage) • Primarily a childhood disease 17 May 06 KL Vadheim Lecture 3 6

Diphtheria Transmission • Person-to-person by respiratory droplet • No animal reservoir 17 May 06 KL Vadheim Lecture 3 7

Manufacturing Process • Toxigenic strain of C. diphtheriae grown in Fenton medium with a bovine extract • After suitable growth, toxin purified from cells by centrifugation • Toxoided by incubation with formaldehyde for several weeks • Concentrated with ultrafiltration • Purified by precipitation, dialysis and sterile filtered • Adsorbed onto aluminum hydroxide, Al(OH)3 17 May 06 KL Vadheim Lecture 3 8

Excellent Vaccine Efficacy • Mortality – 1860 -1897 46 -196/100, 000 people – Case fatality rates >50% during outbreaks – 1920 15/100, 000 people • Cases – 1971 - 1981 - 1, 288 in U. S. – 1980 - 1995 - 41 – occasional in developing countries and Native populations in U. S. and Canada 17 May 06 KL Vadheim Lecture 3 9

Tetanus Vaccine • • Detoxified bacterial, protein toxin Clostridium tetani Injectable dosage form IM 17 May 06 KL Vadheim Lecture 3 10

Tetanus Transmission • Not a communicable disease • The only vaccine-preventable infection that is not communicable • Disease acquired through exposure to bacterial spores in the environment – inoculation of bacterial spores into body by puncture or deep cut 17 May 06 KL Vadheim Lecture 3 11

Transmission II • Neonatal tetanus most common worldwide • Case fatality increases with age (~50% for 80+ years of age) • Disease reservoirs – Soil – Animal feces 17 May 06 KL Vadheim Lecture 3 12

Tetanus toxins • Tetanolysin - possible role in establishing infection at inoculation site • Tetanospasm – accumulates intracellularly during log-phase growth – released into medium upon autolysis – Minimum human lethal dose ~ 2. 5 ng/kg 17 May 06 KL Vadheim Lecture 3 13

Tetanus disease • Tetanospasms – localized - spasm of muscles close to site of injection; weeks to months duration; rare but may precede generalized symptoms – generalized - 80% of cases • Complications of the spasms: – fractures of the long bones and vertebrae – asphyxia from glottic obstruction 17 May 06 KL Vadheim Lecture 3 14

Nervous system effects • Toxin travels up nerve endings by intraaxonal transport • Gains entry to neuromuscular junctions by binding to gangliosides • Interferes with release of neurotransmitters from presynaptic inhibitory fibers • Excitatory reflexes multiply unchecked, causing spasms 17 May 06 KL Vadheim Lecture 3 15

Tetanus • Toxin-mediated disease • Symptoms may progress clinically despite use of parenteral antibiotics • Treatment: – hyperimmune serum – supportive care • Recovery may depend on development of new functional nerve connections 17 May 06 KL Vadheim Lecture 3 16

Manufacturing Process • Growth of C. tetani in modified Latham broth in fermenters • Harvest extracellular toxin by filtration • Purify • Detoxify with formaldehyde for ~3 weeks • Adsorb with Alum adjuvant • Diafiltration 17 May 06 KL Vadheim Lecture 3 17

Pertussis (Whooping Couth) • Bordetella pertussis • Whole cell and acellular vaccines – traditional and recombinant • Injectable dosage form • IM 17 May 06 KL Vadheim Lecture 3 18

Pertussis Transmission • Person-to-person • Respiratory droplets 17 May 06 KL Vadheim Lecture 3 19

Pertussis (whooping cough) • Killed Whole cell – old, not licensed in U. S. or Europe – still used in developing countries – relatively cheap • Acellular (a. P) – currently licensed in U. S. , Japan and Europe – some are recombinant – expensive 17 May 06 KL Vadheim Lecture 3 20

B. pertussis virulence factors • • • Pertussis toxin (PT) Filamentous hemagglutinin (FHA) Fimbriae (fimbrial agglutinogens) (Fim) Pertactin (PRN) Adenylate cyclase Tracheal cytotoxin Dermonecrotic or heat-labile toxin Brk. A Endotoxin (LPS) 17 May 06 KL Vadheim Lecture 3 21

Vaccine Efficacy • 1914 (Pre-vaccine) – ~270, 000 cases, 10, 000 deaths annually – killed >5 of every 1000 children born in U. S. • 1976 - 1, 010 cases • 1996 - 7, 796 • 2002 – 8, 296 cases – 3. 01/100, 000 pop. 17 May 06 KL Vadheim Lecture 3 22

Vaccine History • • 1914 1948 1991 - 2002 2005 17 May 06 First whole-cell vaccine (P) DTP DTa. P for children Tdap for adults 19 -64 KL Vadheim Lecture 3 23

Manufacturing • B. pertussis cultures grown in modified Stainer-Scholte broth • Acellular antigens: – purified from culture medium (PT and FHA) or – extracted from cells (PRN, FIM) • Formaldehyde and/or glutaraldehyde detoxification of PT • Aluminum adjuvants 17 May 06 KL Vadheim Lecture 3 24

Manufacturing II • Novartis-Behring (Chiron) - Recombinant – Genetically detoxified PT – Two other components – Not available in U. S. yet 17 May 06 KL Vadheim Lecture 3 25

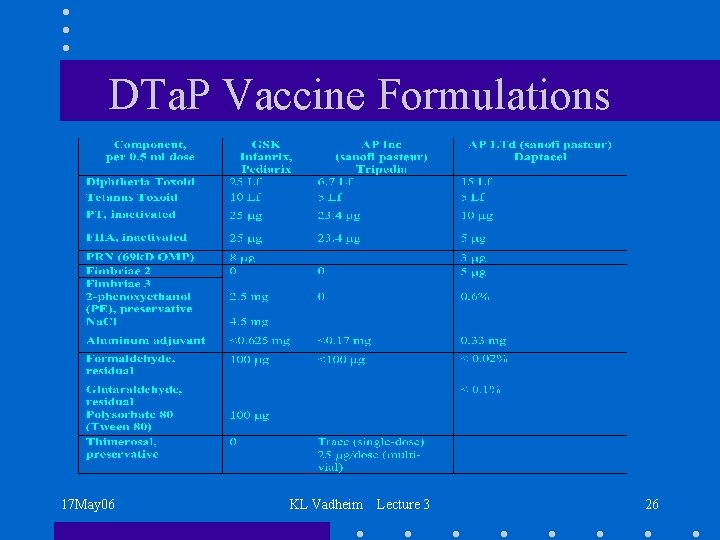
DTa. P Vaccine Formulations 17 May 06 KL Vadheim Lecture 3 26

Licensed DTa. P Vaccines 17 May 06 KL Vadheim Lecture 3 27