DIOXINAS Y FURANOS Prof Jess Olivero Verbel Ph

DIOXINAS Y FURANOS Prof. Jesús Olivero Verbel. Ph. D. Grupo de Química Ambiental y Computacional Universidad de Cartagena

DIOXINAS, FURANOS PCBs PFOS

¿Qué son los COPs? Tóxicos, Persistentes, Bioacumulables, Contaminantes globales Tóxicas: en muy bajas concentraciones afectan la salud de las personas, animales y el medio ambiente. Persistentes: tienen muy lenta degradación física, química o microbiológica. Bioacumulables: por sus características físico-químicas se acumulan en tejidos grasos, biomagnificándose al subir en la cadena alimenticia. Contaminantes globales: se dispersan por el medio ambiente por medio de corrientes de aire, marinas, ríos y en los seres vivos, encontrándose en el agua, sedimentos, animales y personas incluso en zonas remotas. 3

¿Cuáles son? • Plaguicidas organoclorados: Aldrin, Clordano, DDT, Dieldrin, Endrin, Heptacloro, Hexaclorobenceno, Mirex y Toxafeno, • Bifenilos Policlorados (PCBs) usado como aceite dieléctrico en transformadores y acumuladores y otros usos. • Dioxinas y Furanos: subproductos no intencionales generados por combustión y en algunos procesos industriales cuyos insumos contienen cloro. 4

Convenio de Estocolmo Objetivo: Proteger la salud humana y el medio ambiente frente a los Contaminantes Orgánicos Persistentes (Art. 1) 5

What Are “Dioxins”? A family of structurally related chemicals which have a common mechanism of action and induce a common spectrum of biological responses.
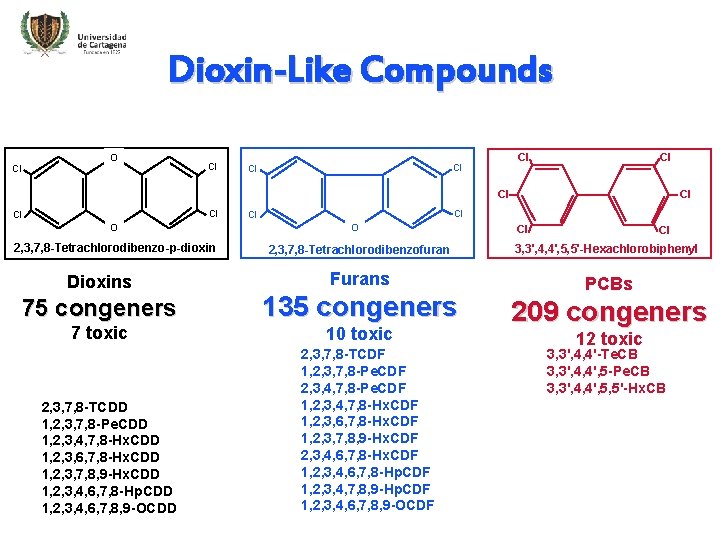
Dioxin-Like Compounds O Cl Cl Cl O 2, 3, 7, 8 -Tetrachlorodibenzo-p-dioxin Dioxins 75 congeners 7 toxic 2, 3, 7, 8 -TCDD 1, 2, 3, 7, 8 -Pe. CDD 1, 2, 3, 4, 7, 8 -Hx. CDD 1, 2, 3, 6, 7, 8 -Hx. CDD 1, 2, 3, 7, 8, 9 -Hx. CDD 1, 2, 3, 4, 6, 7, 8 -Hp. CDD 1, 2, 3, 4, 6, 7, 8, 9 -OCDD Cl Cl Cl O Cl Cl 2, 3, 7, 8 -Tetrachlorodibenzofuran 3, 3', 4, 4', 5, 5'-Hexachlorobiphenyl Furans PCBs 135 congeners 10 toxic 2, 3, 7, 8 -TCDF 1, 2, 3, 7, 8 -Pe. CDF 2, 3, 4, 7, 8 -Pe. CDF 1, 2, 3, 4, 7, 8 -Hx. CDF 1, 2, 3, 6, 7, 8 -Hx. CDF 1, 2, 3, 7, 8, 9 -Hx. CDF 2, 3, 4, 6, 7, 8 -Hx. CDF 1, 2, 3, 4, 6, 7, 8 -Hp. CDF 1, 2, 3, 4, 7, 8, 9 -Hp. CDF 1, 2, 3, 4, 6, 7, 8, 9 -OCDF 209 congeners 12 toxic 3, 3', 4, 4'-Te. CB 3, 3', 4, 4', 5 -Pe. CB 3, 3', 4, 4', 5, 5'-Hx. CB

Dioxinas
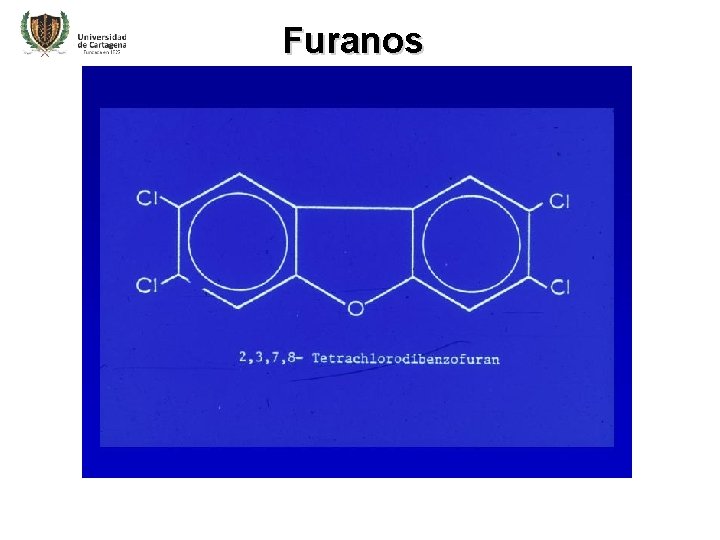
Furanos
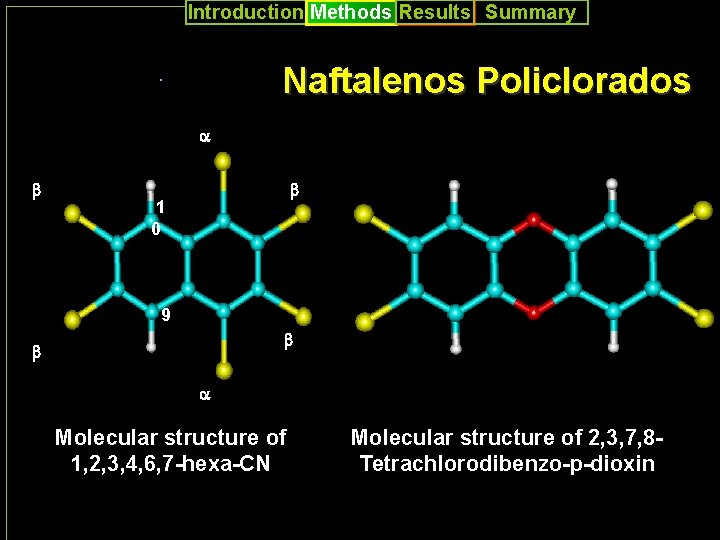
Introduction Methods Results Summary Naftalenos Policlorados Naftalenos Pol . a a b b 1 0 b b b 9 b b b aa Molecular structure of 1, 2, 3, 4, 6, 7 -hexa-CN Molecular structure of 2, 3, 7, 8 Tetrachlorodibenzo-p-dioxin

2, 3, 7, 8 -Tetrachlorodibenzo-p-dioxin “The Most Toxic Man-Made Compound” • Prototype for family of structurally related compounds • Common mechanism of action • Common spectrum of biological responses • Environmentally and biologically persistent (Basis for TEQ approach)

Why the Interest in Dioxins? • 1899 – Chloracne Characterized • 1929 – PCBs produced commercially • 1947 – “X” Disease in cattle • 1957 – Chick Edema Disease; TCDD identified in TCPs • 1962 -1970 – Agent Orange use in Southeast Asia • 1971 – Times Beach; TCDD causes birth defects in mice • 1976 – Seveso, Italy • 1978 – Kociba rat cancer study • 1981 – Capacitor fire in Binghamton, NY • 1985 – 1 st US EPA health assessment of TCDD • 1991 – NIOSH cancer mortality study of US workers • 1999 – Belgium dioxin poisoning; Viennese poisoning

“Dioxins” Polyhalogenated Dibenzo-p-dioxins and furans Never produced intentionally Unwanted byproducts of industrial and combustion processes Polyhalogenated Biphenyls, Naphthalenes, Azo/azoxybenzenes Commercially produced Major industrial chemicals Limited number of congeners have dioxin-like properties Lateral halogenation > 3 Halogens Chlorinated, brominated, and mixed chloro-bromo congeners

Dioxin-Like Compounds • • • Semivolatile Lipophilic Hydrophobic Persistent Bioaccumulating Toxics

PCBs • Large Family of Chemicals – 209 Possible Congeners – Small Subset Are “Dioxins” – NEVER have PCBs without Dioxin-like PCBs • Majority Have Own, Inherent, Toxicities – Multiple, Overlapping, Structural Classes – Can Interact Additively, Synergistically, and/or Antagonistically With Dioxins and With Other PCB Congeners

TCDD is NEVER Found Alone • Complex Mixtures Exist both Environmentally and in Animal and Human Tissues • TCDD is only a Small Part of Total Chemical Mass • We have the Most Toxicological Information about TCDD.

Toxic Equivalency Factors (TEFs) • Developed for Risk Assessment • Interpret Complex Database Derived from Analysis of Samples Containing Mixtures of Dioxin-like Chemicals • Express Quantitatively the Toxicity of a Chemical in terms of an Equivalent concentration of TCDD (Relative Potency) • ∑([Chemical] x TEF)PCDD/PCDF/PCB=TEQ


Five Compounds Make up about 80+% of the Total TEQ in Human Tissues • Four of 17 Toxic PCDD/PCDF Congeners – 2, 3, 7, 8 -TCDD – 1, 2, 4, 7, 8, -Pe. CDD – 1, 2, 3, 6, 7, 8 -Hx. CDD – 2, 3, 4, 7, 8 -Pe. CDF • One of the 12 Toxic PCBs – PCB 126


Major Past Sources of Dioxins (20 th Century Problem) • Chloralkali Facilities • Chlorinated herbicide and biocide Production. • Leaded Gasoline • Municipal, Medical, and Hazardous Waste Incineration. • Chlorine Bleaching of Paper and Pulp Products.

Recently Identified Sources • Open Burning of Household Waste • Uncontrolled Combustion – Forest Fires and Volcanos • Metal Refining


From Factory to the Fetus Dioxins and PCBs: Pathways of Exposure and Neurodevelopmental Effects PCBs AIR Dioxins: PVC Manufacturing Medical/Municipal Incinerator Dioxins PCB s SOIL WATE R FOOD PCBs: Transformers Landfills Hazardous Waste Sites

How do Dioxins Move in the Environment • If emitted into air, undergo atmospheric transport and deposition on land or water. • If emitted into water, bind to sediment. • Recycle in environment. • Bioaccumulate up the food chain. • Resistance to physical, chemical, and biological degradation.

Dioxin Environmental Mobility • Dioxins are less mobile than Hg or the more volatile PCBs. • Dioxins do not appear to exhibit global retort, or strong “grasshopper” effect. • Dioxins are continually exchanged among media and should be viewed as a complex system of stocks and flows.

How are People Exposed? • Dioxins are omnipresent • Majority of exposure (>95%) is via microcontamination of food – Meat, fish, dairy. • Sensitive Subpopulations with High Exposure – Subsistence Fishers and Hunters – Nursing Infants – Occupational Workers • Oral, dermal, and inhalation exposures

How You are Exposed Makes Little Difference • Dioxins are well absorbed from the GI tract and lungs – Skin absorption is limited and slow. • Dioxins primarily lodge in the liver and fat. • Dioxins are primarily eliminated after metabolism, which is VERY slow.

Why do the Body Burdens Increase Over Time? • Persistence – Resistance to Biological, Chemical, and Physical Degradation – Long Half-Lives in Animals and People • More Body Fat- Longer Half-Life • Half-Life is Dose-Dependent. • Bioaccumulation – Due to Persistence in Animal tissues Higher Trophic Organisms have Higher Concentrations. – Older Organisms have Higher Body Burdens than Young.

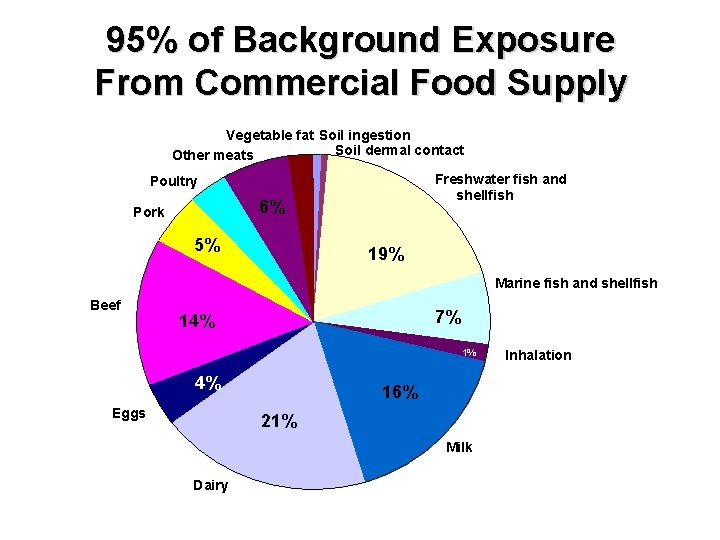
95% of Background Exposure From Commercial Food Supply Vegetable fat Soil ingestion Soil dermal contact Other meats Freshwater fish and shellfish Poultry 6% Pork 5% 19% Marine fish and shellfish Beef 7% 14% 1% 4% Eggs 16% 21% Milk Dairy Inhalation

Dioxin/PCB Exposure Trends • Environmental Levels – Peaked in late ’ 60 s/early ’ 70 s – decline since confirmed by sediment data – Decline also supported by Emissions Inventory – shows significant decrease from ’ 87 to ‘; 95 (~80%) • Human tissue data suggest mid-90 s levels approximately half of 1980 – 55 25 ppt TEQ lipid (~5 ng/kg ww) – Decrease continues • Success of Regulatory Agenda!
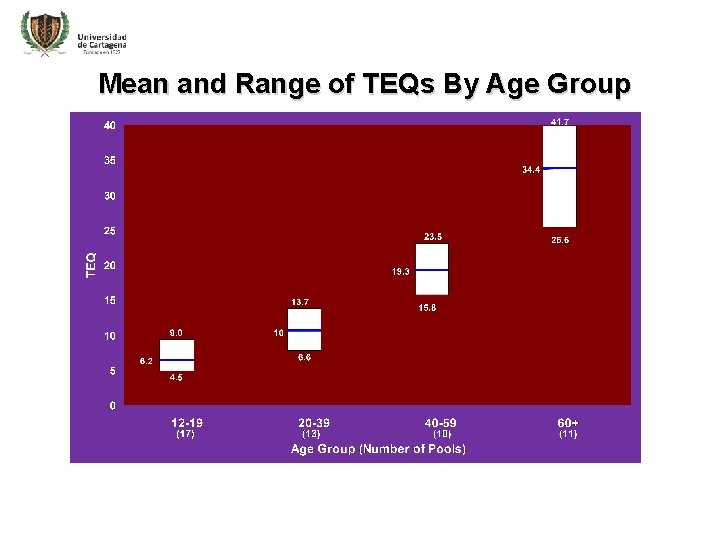
Mean and Range of TEQs By Age Group

Adverse Effects Wildlife and Domestic Animals Great Lakes fish, birds, mammals Baltic seals, Dolphins Developmental/reproductiv e effects Immunological effects Effects observed at environmental levels Cows, Horses, Sheep, Chickens Effects observed during poisoning episodes. Laboratory Animals Fish Amphibians Turtles Birds Rats Mice Guinea Pigs Hamsters Rabbits Dogs Non-human primates

Effects of Dioxins BIOCHEMICAL • Induction of Drug Metabolizing Genes – Cyp 1 A 1/2, 1 B 1; GST; UDPGT; ALDH… • Induction of Proliferation Genes • Induction of Cytokines – TNF, IL-6, IL-1β • Induction of Oxidative Stress • Induction of Growth factors/receptors – TGFs, EGFR… • Modulation of Hormones/Receptors TOXIC • Lethality/Wasting • Gonadal/Lymphoid Atrophy • Hyperplasia/Metaplasia • Endocrine Disruption • Carcinogenicity • Repro/Developmental toxicity • Functional Devpt. Toxicity • Dermal Toxicity • Immunotoxicity • Neurotoxicity • Hepatic Toxicity • Cardiovascular Toxicity • Bone/Teeth Toxicity

Effects of Dioxins • • • Multiple Effects Multiple Tissues Both Sexes Multiple Species Throughout Vertebrata • Molecular/ Biochemical • Metabolic/ Cellular • Tissue/Organ • Growth/ Differentiation • Wasting/Death

Dioxin Effects Require the “Ah Receptor” • Highly conserved protein – throughout Vertebrates – Related Proteins in Invertebrates • Member of Growing Family of Key Regulatory Proteins – Development, Aging, Hypoxia, Daily Rhythms • Necessary, but Not Sufficient, for All of the Effects of Dioxins
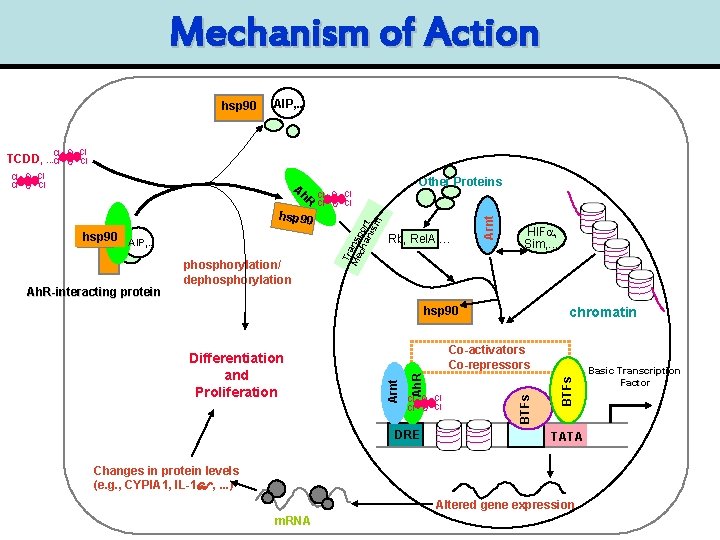
Mechanism of Action hsp 90 AIP, . . Cl O Cl TCDD, . . . Cl hsp 90 AIP, . . Ah. R-interacting protein phosphorylation/ dephosphorylation Other Proteins Cl O Cl Rb, Rel. A, … HIFa, Sim, . . . hsp 90 Cl O Cl DRE BTFs Ah. R Co-activators Co-repressors Arnt Differentiation and Proliferation chromatin BTFs h. R Arnt A Tr Me ansp ch or an t ism Cl O Cl TATA Changes in protein levels (e. g. , CYPIA 1, IL-1 , . . . ) Altered gene expression m. RNA Basic Transcription Factor
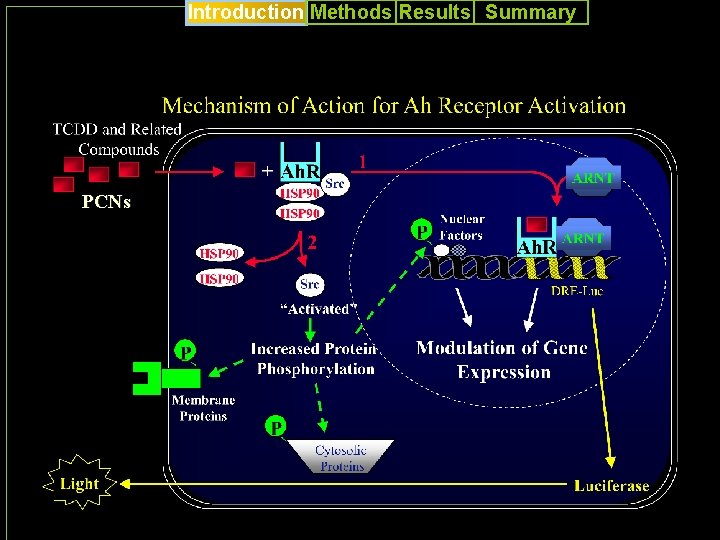
Introduction Methods Results Summary PCNs

Dioxins’ Effects in People • Cardiovascular Disease • Diabetes • Cancer • Porphyria • Endometriosis • Decreased Testosterone • Chloracne • Biochemical – Enzyme Induction – Receptor Changes • Developmental – – – – Thyroid Status Immune Status Neurobehavior Cognition Dentition Reproductive Effects Altered Sex Ratio Delayed Breast Devpt

Unfortunate Poisoning Episodes • PCBs/PCDFs – Japan (“Yusho”) – Taiwan (“Yucheng”) • PBBs/PBNs – Michigan • TCDD – Seveso, Italy – Vienna, Austria – Ukraine • Clear Evidence of Adverse Health Effects

Chloracne Classic Toxic Effect • • “Hallmark of Dioxin Toxicity” High-Dose Response Genetic Polymorphism Occurs in People, Monkeys, Cows, Rabbits, and Mice • Associated with multiple problems with skin, teeth, hair and nails following prenatal exposure
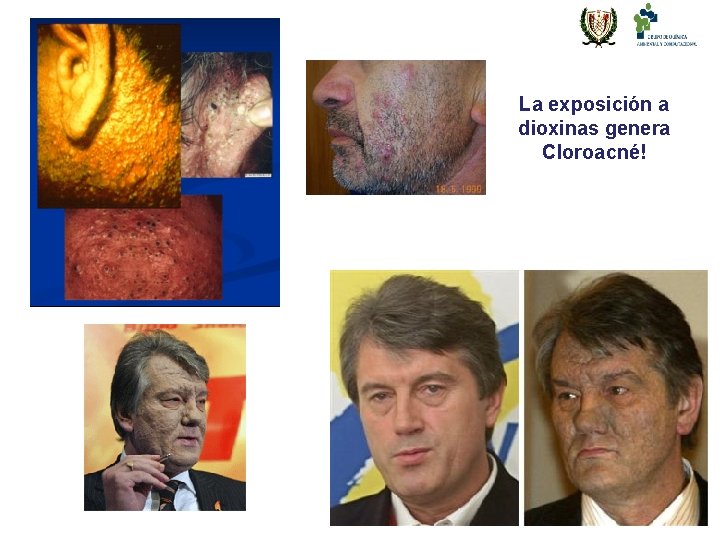
La exposición a dioxinas genera Cloroacné!

HEALTH EFFECTS IN “HIGHLY” EXPOSED POPULATIONS • • Exposures Are Not As High As We Once Thought: 10 -100 X Background (“Ambient”) Occupational Populations – Chloracne, Cancer, Heart Disease, Diabetes, . . . • Poisoning Episodes – Chloracne. Cancer, Heart Disease, Diabetes, Reproductive, Developmental, Hormonal and Immune Effects

EFFECTS SEEN IN ADULTS AT BACKGROUND EXPOSURES • Type II Diabetes – Decreased Glucose Tolerance – Hyperinsulinemia – Mechanistic Plausibility • Endometriosis – Hormone Disruption and Immune Suppression – Animal Models • Cancer? ?

HEALTH OUTCOMES IN PRENATALLY-EXPOSED CHILDREN • • • Studies in the US (Michigan, North Carolina, Lake Oswego); Japan; the Netherlands; Sweden; Finland Low Birthweight Cognitive and Behavioral Impairment Immune System Effects Hormonal Changes (Thyroid Effects) Altered Dentition

Dioxin Effects of Greatest Concern • Developmental Alterations Occurring at “High End” of Background Population • Decreased neuro-optimality and IQ • Altered Behavior • Altered Immune System • Altered Hormone Systems • Altered Growth

ORGANOFLUORINES F is the most electronegative element. C-F bond is the strongest of known covalent bonds. e. g. , C-F bond can withstand boiling with 100% sulfuric acid without any defluorination. Perfluorinated (fully fluorinated) alkanes are anthropogenic.
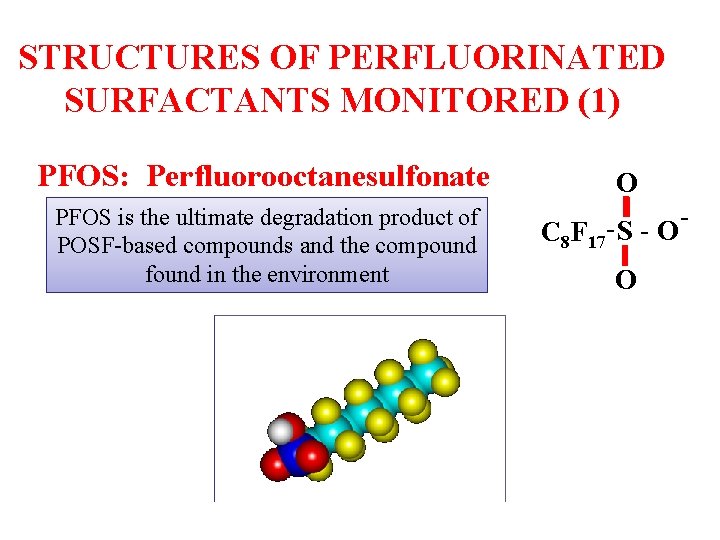
STRUCTURES OF PERFLUORINATED SURFACTANTS MONITORED (1) PFOS: Perfluorooctanesulfonate PFOS is the ultimate degradation product of POSF-based compounds and the compound found in the environment O C 8 F 17 S O O

ORGANOFLUORINE SURFACTANTS Surfactants: Surface-active agents; due to selective adsorption at the interface. Amphiphilic or amphipathic : Implies attraction to two different kinds of media. Surfactant structure consists of ‘hydrophilic’ and ‘hydrophobic’ parts. The hydrophobic portion repels not only water but also oil and fat. So, fluorinated surfactants exhibit both water and oil repellency when adsorbed on substrates such as textiles or paper.

USES OF FLUORINATED SURFACTANTS (1) Adhesives, antifogging, antistatic agents, cement and paint additives, insulators for cables, electroplating, herbicides and insecticides, cleaners for hard surfaces (automotive waxes), cosmetics (hair conditioning formulations). Fire-Fighting Foams: Formulated to float on flammable liquids and extinguish flames. FS in Aqueous Film-Forming Foams (AFFF) reduce the surface tension of water and form a film on the fuel surface.

USES OF FLUORINATED SURFACTANTS (2) Paper: Used in folding cartons for snack foods, carry-out fast food, cake mixes, margarine, candy, bakery products and pet foods. Polishes, Waxes, Leather: In floor waxes to provide dust repellency. Textiles: Carpets, Polyester, etc. to impart soil, oil and water repellency.

Concentrations (ng/m. L) of PFOS in blood and sera of seals: Spatial differences Location Baltic Sea Ringed Gray Seal 16 -475 [110] 14 -76 [37] Canadian Arctic <3 -12 Norwegian Arctic 5 -14 [9] 11 -49 [28] NA Values in brackets are mean; n = 10 to 50
![PFOS IN POLAR BEARS : ALASKAN ARCTIC Liver Blood 82 -680 [350] ng/g, wet PFOS IN POLAR BEARS : ALASKAN ARCTIC Liver Blood 82 -680 [350] ng/g, wet](http://slidetodoc.com/presentation_image_h2/b7c0c2f968248fd1599a0f58967588b1/image-54.jpg)
PFOS IN POLAR BEARS : ALASKAN ARCTIC Liver Blood 82 -680 [350] ng/g, wet wt <2 -52 [34] ng/m. L Values in brackets are mean; n = 10 to 40
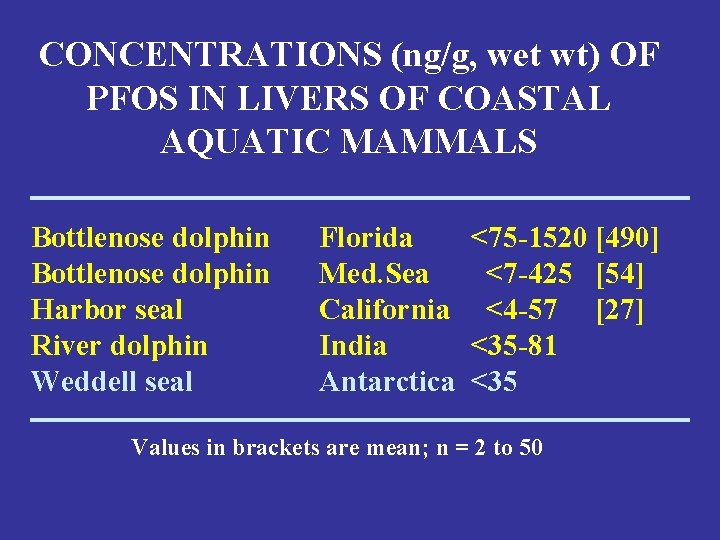
CONCENTRATIONS (ng/g, wet wt) OF PFOS IN LIVERS OF COASTAL AQUATIC MAMMALS Bottlenose dolphin Harbor seal River dolphin Weddell seal Florida Med. Sea California India Antarctica <75 -1520 [490] <7 -425 [54] <4 -57 [27] <35 -81 <35 Values in brackets are mean; n = 2 to 50

BIOMAGNIFICATION OF PFOS IN MINK (LIVERS) Species Mink liver n 18 conc. 2630 Salmon and whitefish liver Liver-BMF 15 80 33 Concentrations are in ng/g, wet wt.

ENVIRONMENTAL FATE PFOS is relatively water soluble, low vapor pressure and tends to bind to particulates While PFOS and related perfluorinated alkanes are persistent and can be accumulated into and retained in biota, their environmental fate processes are fundamentally different from organochlorines and paradigms and models developed for OCs do not apply

TOXIC EFFECTS OF PERFLUOROCHEMICALS Peroxisome proliferation (interference with fatty acid binding and transport) Mitochondrial bioenergetics (stimulation of mitochondrial respiration) Inhibits cell-cell communication Reproductive and developmental effects: rat LD 50 = 250 mg/kg Neuro-endocrinal effects

CONCLUSIONS PFOS is a ubiquitous global environmental pollutant: present both in urban and remote locations. Distribution of PFOA, FOSA and PFHS is localized. PFOS bioaccumulates in humans and wildlife; biomagnifies in the food chain. Age or sex-related accumulation is not prominent Transport pathways and environmental fate of fluorochemicals are the subjects of further/current investigation.
- Slides: 59