DIMENSIONAL ANALYSIS also known as FactorLabel Method Steps

DIMENSIONAL ANALYSIS (also known as Factor-Label Method)
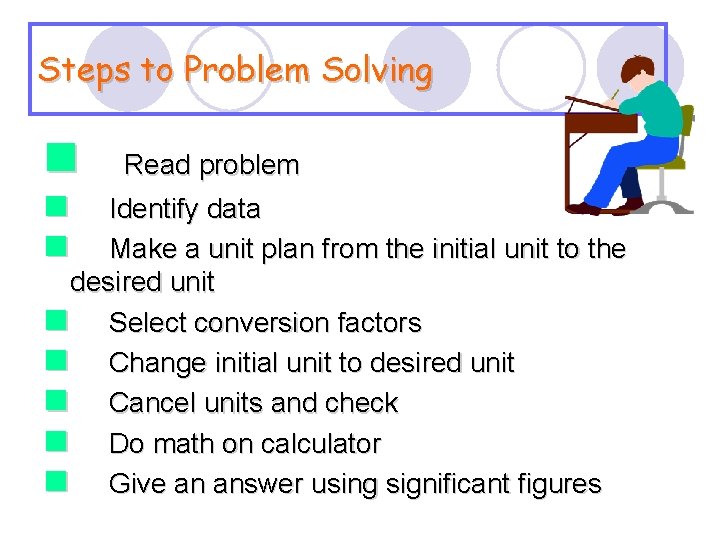
Steps to Problem Solving n Read problem n Identify data n Make a unit plan from the initial unit to the desired unit n Select conversion factors n Change initial unit to desired unit n Cancel units and check n Do math on calculator n Give an answer using significant figures

Conversions l. If you know ONE conversion for each type of measurement, you can convert anything! l. You must memorize and be able to use the metric conversions:

Conversion Factors Fractions in which the numerator and denominator are EQUAL quantities expressed in different units Example: 1 ft = 12 in. Factors: 1 ft. and 12 in 1 ft. Example: 1 g = 102 cg. Factors: 1 g. and 102 cg 1 g

How many minutes are in 2. 5 hours? Conversion factor 2. 5 hr x 60 min 1 hr = 150 min By using dimensional analysis / factor-label method, the UNITS ensure that you have the conversion right side up, and the UNITS are calculated as well as the numbers!

Sample Problem l You have $7. 25 in your pocket in quarters. How many quarters do you have? 7. 25 dollars X 4 quarters = 29 quarters 1 dollar

Sample Problem – Using Metric l You have 200 L of gasoline in your car. How many milliliters do you have? 200 liters 103 milliliters 1 liter = 2 x 103 m. L

Learning Check SET UP THESE PROBLEMS USING DA 2. 22 cg = _______g 1. 7 x 106 ng = ______ g. 288 g = _____mg 1. 5 KL = ______L

Multi-Step Problems You are in Paris and want some peaches for lunch. The sign in the fruit stand indicates that peaches are 11. 5 francs per kilogram. Given that there approximately 5 francs to the dollar, calculate what a pound of peaches will cost in dollars. 1 lb. 455 g 1 lb. 1 Kg 1000 g 11. 5 f 1 Kg 1$ 5 f NOW WHAT? ?

Practice Problems Mrs. Borden has 3 classes of Chemistry. Each class has 25 students in it and they are each required to pay $15. 00 for their lab fee. How much money will she have at the end of the semester if everyone pays their fee? l Students in chemistry are required to write 10 lab reports per year. Each lab report has 6 sections each. Mrs. Borden requires that each section consists of a paragraph with no less than 7 sentences. How many paragraphs will each student write in 1 year time? l

Complex Units Many times a problem will ask for you to convert 2 different units in one problem. Example: Change miles per hour to feet per minute. Given 20 miles per hour, how many feet is that per minute? ( 5280 ft = 1 mile) 20 miles 1 hour 5280 ft. 60 min. 1 mile = 1760 ft. /min.

YOU TRY IT l. The density of gold is 19. 3 g/m. L. What is gold’s density in decigrams per liter?

NEW UNITS l These are units you have never used but you must know for the rest of the year. 1 mole of any element or compound = its mass 1 mole of any element or compound = 6. 02 x 1023 representative particles

CONVERSION FACTORS l 1 mole of Na. Cl = mass of Na. Cl How to calculate mass, using the periodic table: Na = 23. 0 g and Cl = 35. 5 g 23. 0 + 35. 5 therefore 1 mole of Na. Cl = 58. 5 g or 1 mole of Na. Cl 58. 5 g

Practice p. ROBLEM l You are given 3. 5 moles of Na. Cl, how many grams of Na. Cl do you have? 3. 5 moles Na. Cl 58. 5 g Na. Cl = 204. 75 g of Na. Cl 1 mole Na. Cl

CONVERSION FACTORS l 1 mole of Na. Cl = 6. 02 x 1023 particles of Na. Cl Nothing to calculate, the number of particles is the same for every element therefore 1 mole of Na. Cl = 6. 02 x 1023 Particles or 1 mole of Na. Cl 6. 20 x 1023 particles

Practice p. ROBLEM l You are given 3. 5 moles of Na. Cl, how many particles of Na. Cl do you have? 3. 5 moles Na. Cl 6. 20 x 1023 particles of Na. Cl 1 mole Na. Cl = 21. 07 x 1023 particles of Na. Cl or 2. 107 x 1024 particles

Multi-Step Problems l You are given 4. 55 x 1025 particles of oxygen, how many grams of oxygen do you have? 4. 55 x 1025 rp of O 1 mole of O 16. 0 g of O = 12. 09 x 102 grams 6. 02 x 1023 rp 1 mole of O or 1. 209 x 103 grams of O
- Slides: 18