DIGITAL HEALTH FDA BAKUL PATEL June 6 2019

DIGITAL HEALTH @ FDA BAKUL PATEL June 6, 2019 www. fda. gov/digitalhealth
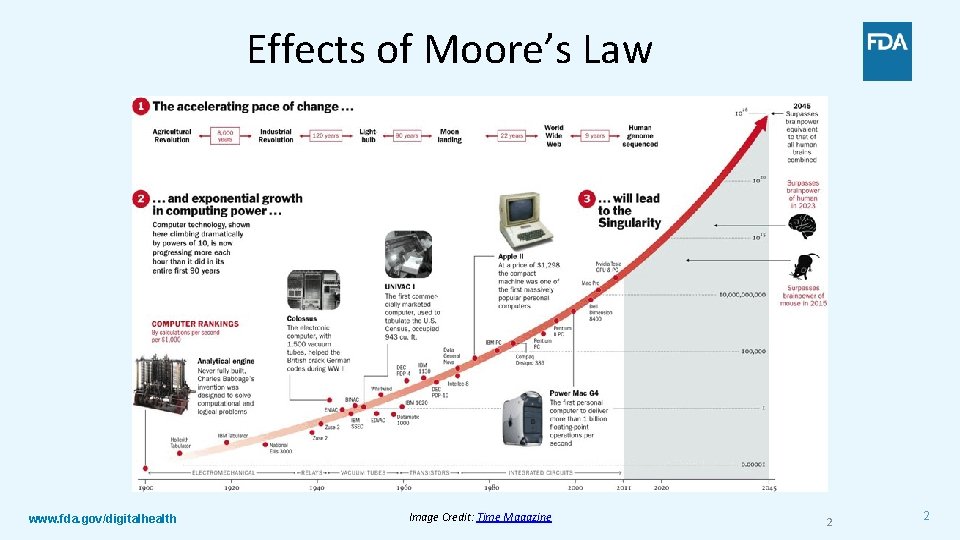
Effects of Moore’s Law www. fda. gov/digitalhealth Image Credit: Time Magazine 2 2
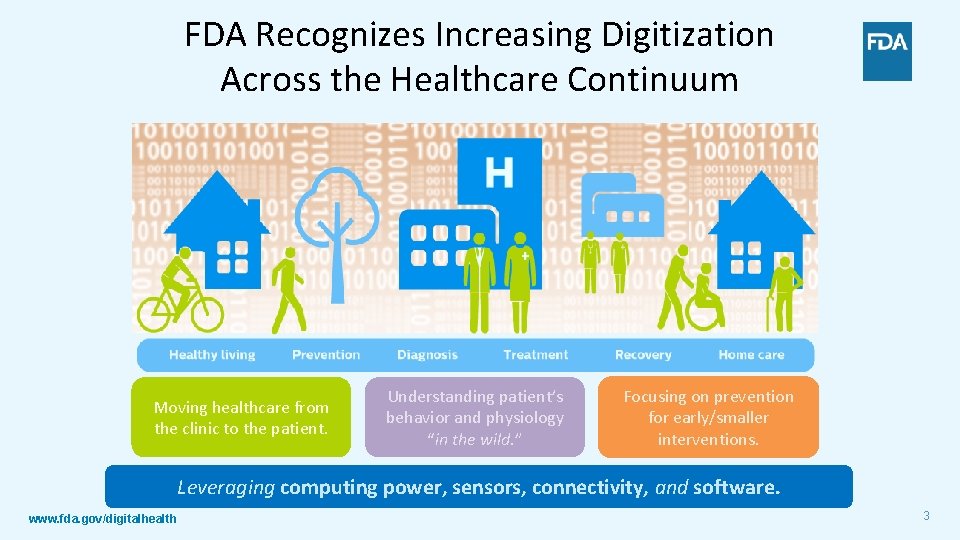
FDA Recognizes Increasing Digitization Across the Healthcare Continuum Moving healthcare from the clinic to the patient. Understanding patient’s behavior and physiology “in the wild. ” Focusing on prevention for early/smaller interventions. Leveraging computing power, sensors, connectivity, and software. www. fda. gov/digitalhealth 3

_ _ Digital Health Team Vision Prepare the FDA for the Digital Health future by promoting innovation and ensuring all Americans have timely access to high-quality, safe and effective digital health products. www. fda. gov/digitalhealth 4
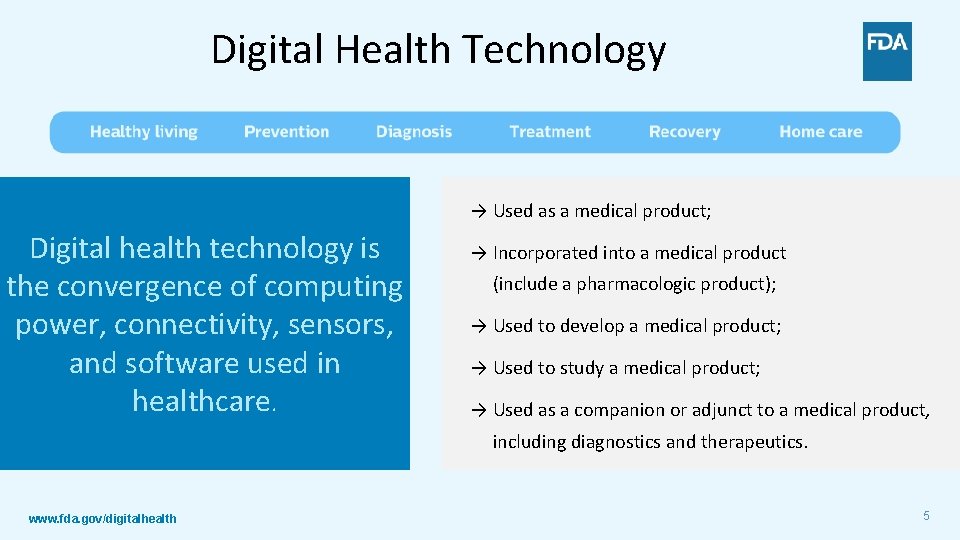
Digital Health Technology → Used as a medical product; Digital health technology is the convergence of computing power, connectivity, sensors, and software used in healthcare. → Incorporated into a medical product (include a pharmacologic product); → Used to develop a medical product; → Used to study a medical product; → Used as a companion or adjunct to a medical product, including diagnostics and therapeutics. www. fda. gov/digitalhealth 5
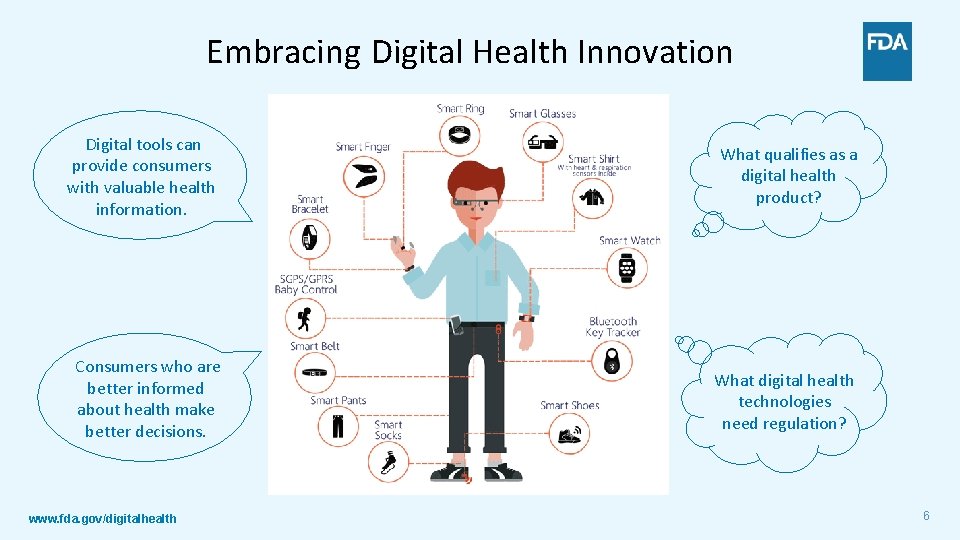
Embracing Digital Health Innovation Digital tools can provide consumers with valuable health information. What qualifies as a digital health product? Consumers who are better informed about health make better decisions. What digital health technologies need regulation? www. fda. gov/digitalhealth 6

Goals for a Tailored Regulatory Framework Fostering Responsible Digital Health Innovation Enhance patients access to high quality digital medical products Enable manufacturers to rapidly improve software products with minor changes Maintain a reasonable assurance of safety and effectiveness Minimally burdensome www. fda. gov/digitalhealth 7

FDA approach to regulation of digital health products www. fda. gov/digitalhealth 8
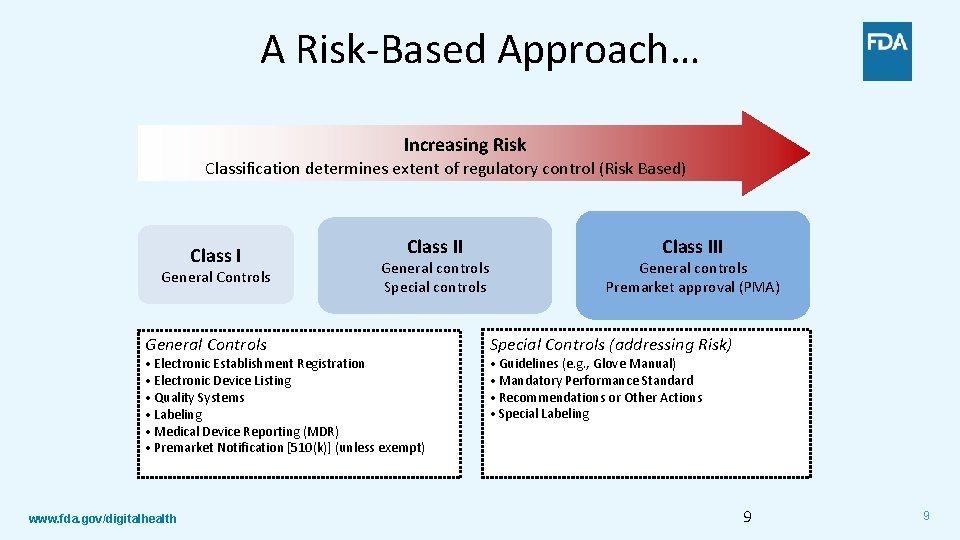
A Risk-Based Approach… Increasing Risk Classification determines extent of regulatory control (Risk Based) Class I General Controls Class II General controls Special controls • Electronic Establishment Registration • Electronic Device Listing • Quality Systems • Labeling • Medical Device Reporting (MDR) • Premarket Notification [510(k)] (unless exempt) www. fda. gov/digitalhealth Class III General controls Premarket approval (PMA) Special Controls (addressing Risk) • Guidelines (e. g. , Glove Manual) • Mandatory Performance Standard • Recommendations or Other Actions • Special Labeling 9 9

…Aimed At Smart Regulation • • Risk Based Narrowly Tailored Functionality Focused Platform Independent www. fda. gov/digitalhealth 10
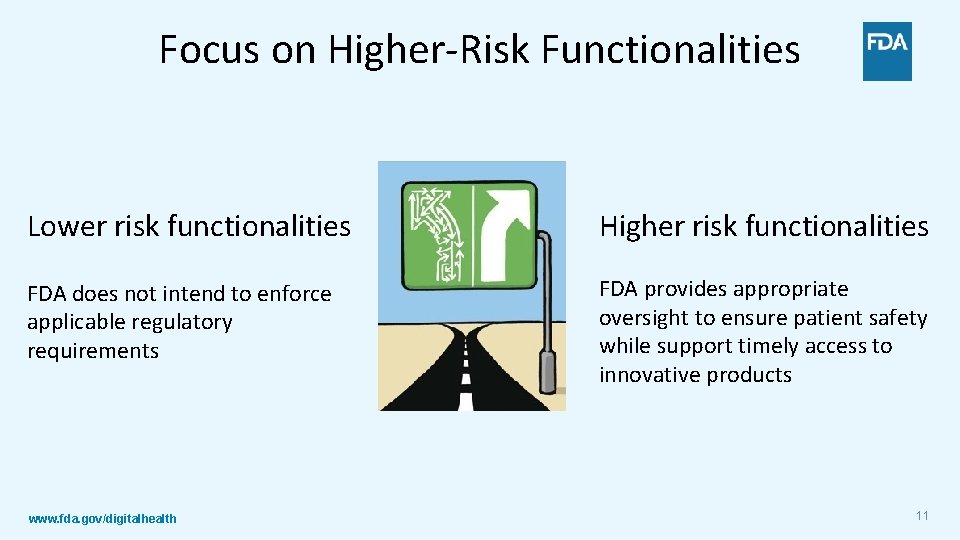
Focus on Higher-Risk Functionalities Lower risk functionalities Higher risk functionalities FDA does not intend to enforce applicable regulatory requirements FDA provides appropriate oversight to ensure patient safety while support timely access to innovative products www. fda. gov/digitalhealth 11
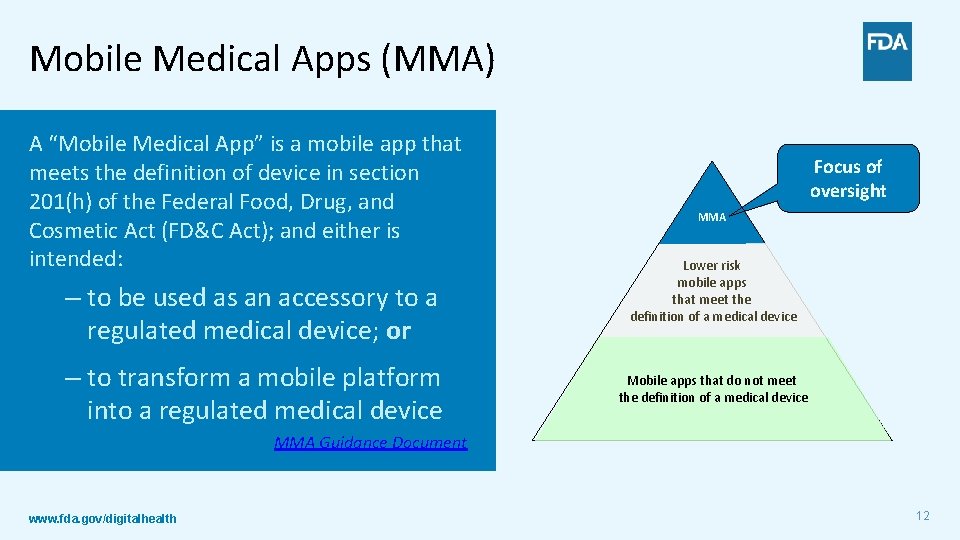
Mobile Medical Apps (MMA) A “Mobile Medical App” is a mobile app that meets the definition of device in section 201(h) of the Federal Food, Drug, and Cosmetic Act (FD&C Act); and either is intended: – to be used as an accessory to a regulated medical device; or – to transform a mobile platform into a regulated medical device Focus of oversight MMA Lower risk mobile apps that meet the definition of a medical device Mobile apps that do not meet the definition of a medical device MMA Guidance Document www. fda. gov/digitalhealth 12

Tools available from FDA https: //www. fda. gov/Medical. Devices/Digital. Health/ucm 562577. htm www. fda. gov/digitalhealth 13
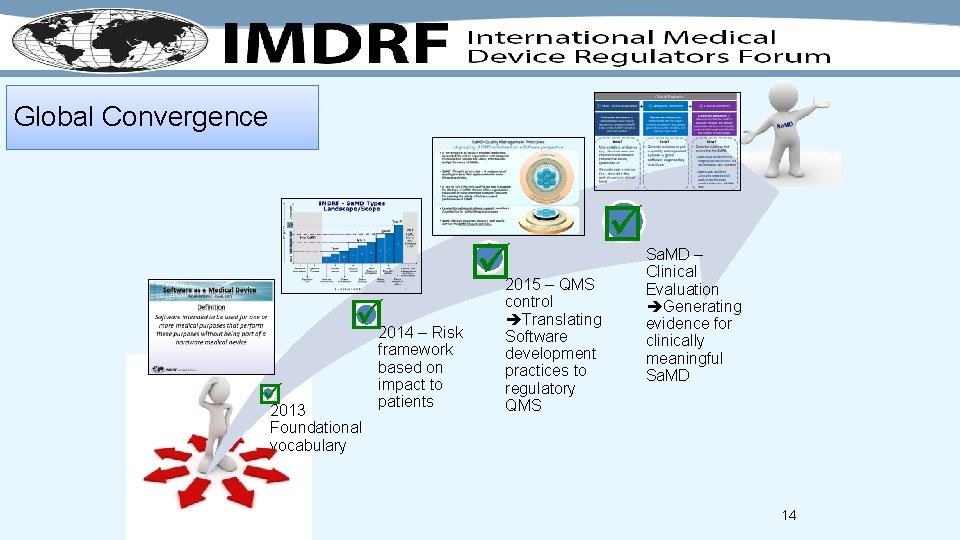
Global Convergence 2013 2014 – Risk framework based on impact to patients 2015 – QMS control Translating Software development practices to regulatory QMS Sa. MD – Clinical Evaluation Generating evidence for clinically meaningful Sa. MD Foundational vocabulary 14

21 st Century Cures Act Codifies current policies and excludes certain software products from medical device definition: • • Medical device data systems EHR certified by ONC General wellness products Administrative products For products with multiple functions, allows FDA to regulate only those functions that meet the definition of a medical device www. fda. gov/digitalhealth Excludes certain types of clinical decision support products from the definition of a medical device. Excluded products must meet all of the following criteria: • Does not acquire or analyze physiological signals; • Is intended to display information about a patient; • Provides recommendations to physicians; and • Allows health care professionals to independently review the basis for such recommendations. 15

FDA Pre-Cert Program An organization-based streamlined regulatory approach for Software as a Medical Device (Sa. MD) that relies on a demonstrated Culture of Quality and Organizational Excellence www. fda. gov/digitalhealth 16

Five Excellence Principles Proposed Patient Safety Product Quality Demonstration of a commitment to providing a safe patient experience, and to emphasizing patient safety as a critical factor in all decision-making processes. Demonstration of a commitment to the development, testing, and maintenance necessary to deliver Sa. MD products at the highest level of quality. Clinical Responsibility Demonstration of a commitment to responsibly conduct clinical evaluation and to ensure that patient-centric issues including labeling and human factors are appropriately addressed. Cybersecurity Responsibility Demonstration of a commitment to protect cybersecurity, and to proactively address cybersecurity issues through active engagement with stakeholders and peers. Proactive Culture Demonstration of a commitment to a proactive approach to surveillance, assessment of user needs, and continuous learning. www. fda. gov/digitalhealth 17

Our Goals For a New Model How can a pre-certification program address the evolving needs of Sa. MD products? Enable a tailored, pragmatic, and least burdensome regulatory oversight that 1. Assesses organizations to establish trust that they have a culture of quality and organizational excellence such that they can develop high quality Sa. MD products; 2. Leverages transparency of organizational excellence and product performance across the entire lifecycle of Sa. MD; 3. Uses a tailored streamlined premarket review; 4. Leverages unique postmarket opportunities available in software to verify the continued safety, effectiveness, and performance of Sa. MD in the real world. www. fda. gov/digitalhealth 18
Digital Health Center of Excellence “ We’re building our Digital Health Center of Excellence to develop more efficient ways to ensure the safety and effectiveness of technologies like smart watches with medical apps. Our Software Precertification Pilot Program is allowing us to test a new approach for product review. ” Dr. Scott Gottlieb, FDA Commissioner April 2, 2019 www. fda. gov/digitalhealth 19
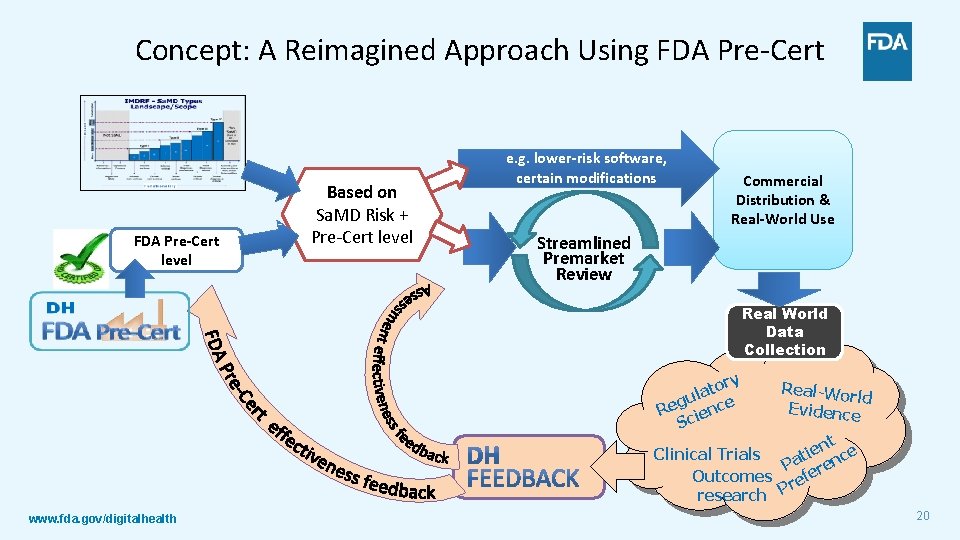
Concept: A Reimagined Approach Using FDA Pre-Cert level Based on Sa. MD Risk + Pre-Cert level e. g. lower-risk software, certain modifications Commercial Distribution & Real-World Use Streamlined Premarket Review Real World Data Collection ory t a gul nce e R e Sci Real-Wo rld Evidence nt e e i Clinical Trials t c Pa eren Outcomes ref P research www. fda. gov/digitalhealth 20
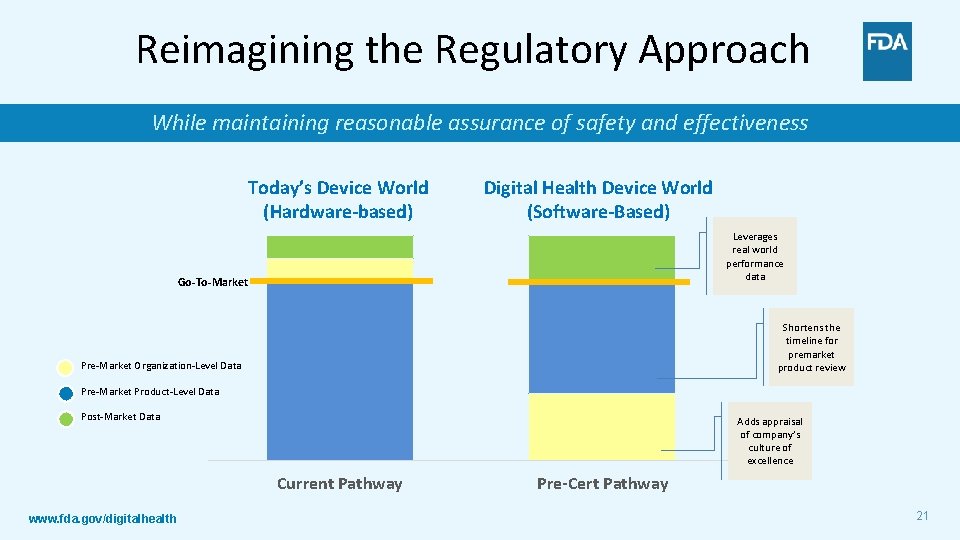
Reimagining the Regulatory Approach While maintaining reasonable assurance of safety and effectiveness Today’s Device World (Hardware-based) Digital Health Device World (Software-Based) Leverages real world performance data Go-To-Market Shortens the timeline for premarket product review Pre-Market Organization-Level Data Pre-Market Product-Level Data Post-Market Data Adds appraisal of company’s culture of excellence Current Pathway www. fda. gov/digitalhealth Pre-Cert Pathway 21

Get More Information www. fda. gov/digitalhealth Digital. Health@fda. hhs. gov Bakul Patel Bakul. Patel@fda. hhs. gov Follow me on Twitter @_Bakul. Patel www. fda. gov/digitalhealth 22
- Slides: 22