Diggin That Density A Look at Density and





















- Slides: 22

Diggin’ That Density A Look at Density and How to Calculate It.

Remember Back ö Volume – Amount of Space something takes up. It is 3 Dimensional ö Mass- Amount of matter something is made of.

Remember Back ö Volume – Measured in milliliters (m. L) or in cubic centimeters (cm³) ö Mass- Measured in grams (g).

Remember Back ö So volume is ö Mass is how the amount of space you have and…. much of something you have.

Bring it Together ö Density is a combination of volume and mass. ö Density is the amount of matter in a given volume. w. The amount of “stuff” in a certain “space”

Figuring it Out ö There is a mathematical formula for calculating density. ö Density = Mass Volume D=M V

Units for Density ö Just like length, volume and mass density has to be labeled with the correct unit. ö g/cm³ or g/m. L is the standard unit for density. ö Remember that a cm³ is

D. Density ö An object has a volume of 825 cm 3 and a density of 13. 6 g/cm 3. Find its mass. GIVEN: WORK: V = 825 cm 3 D = 13. 6 g/cm 3 M=? M = DV M = (13. 6 g/cm 3)(825 cm 3) M = 11, 220 g

D. Density 1) A liquid has a density of 0. 87 g/m. L. What volume is occupied by 25 g of the liquid? GIVEN: WORK: D = 0. 87 g/m. L V=? M = 25 g V=M D V= 25 g 0. 87 g/m. L V = 28. 7 m. L

D. Density 2) You have a sample with a mass of 620 g & a volume of 753 cm 3. Find density. GIVEN: WORK: M = 620 g V = 753 cm 3 D=? D=M V D= 620 g 753 cm 3 D = 0. 82 g/cm 3

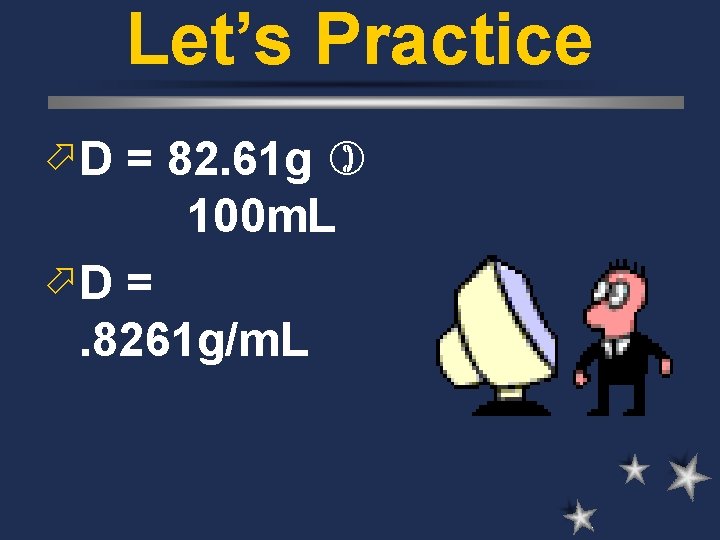
Let’s Practice ö Liquid E had a mass of 82. 61 g. You massed 100 m. L of this liquid. ö Plug it into the formula.

Let’s Practice ö D = 82. 61 g 100 m. L öD = . 8261 g/m. L

Will it Float? ö Density can predict if something will float. How?

Will it Float? ö The object with a higher density than water sinks. If it has lower density it floats.

Density of Water ö Using our results from yesterday, what is the density of sea water? ö 100. 84 g 100 m. L = ö 1. 0084 g/m. L.

Measuring water ö If you wanted to measure 25 ml of water, what instrument would you use?

Density of Water ö One milliliter of water ö Has a mass of 1 gram ö Density = Mass ÷ Volume w Calculate the density of water 1 g ÷ 1 ml = 1 g/ml

Measuring water ö If you wanted to measure 25 ml of water, what instrument would you use?

Measuring water 2 ö If you wanted to measure 25 grams of water, what 2 instruments could you use?

Measuring volume of a solid by water displacement

One cubic centimeter = one milliliter This cube’s sides are each 3 cm. What is the volume of the cube? Using water displacement, how many milliliters do you expect the water to raise when the cube is lowered?

Graduated cylinder measurement ö Volume is measured using a graduated cylinder. ö When reading a graduated cylinder, read the bottom of the meniscus.