Diffusionweighted imaging findings of intracranial tumors relationship between

Diffusion-weighted imaging findings of intracranial tumors relationship between T 2 and diffusibility Hiwatashi A, Moritani T University of Rochester, NY

Introduction Routine magnetic resonance (MR) imaging is sensitive method of detecting tumors of the brain. Diffusion-weighted imaging (DWI) can differentiate between tumor and infection (1 -51) and can help the characterization and grading of brain tumors. This exhibit demonstrates DWI characteristics of intracranial tumors.

Gliomas (Figs 1 -6) The signal intensity of gliomas on DWI is variable and depends mainly on their T 2 and apparent diffusion coefficient (ADC) values (1 -22). Thus, some gliomas are hyperintense on DWI with decreased ADC, which reflects reduced volume of extracellular space. Other gliomas have normal or increased ADC which causes T 2 shinethrough effect.

High Grade Tumors It has been reported that high-grade gliomas tends to show high signal on DWI with decreased ADC (3, 8 -10, 12, 13, 19). High tumor cellularity is probably the major cause (3, 8, 12, 18). There are still controversies regarding how well DWI can differentiate between high grade primary brain tumor and metastasis (6, 18, 46). In lymphoma, ADC have been reported to be lower than in high grade gliomas (16), but in the clinical situation there is often overlap between lymphoma and high-grade glioma.

Peritumoral infiltration The value of DW imaging for the delineation of peritumoral invasion in primary brain tumors is controversial (3, 4, 10 -12, 18, 19). The poor delineation is probably due to conjoined effects of T 2 and ADC. Diffusion tensor imaging may add more information about tumor infiltration.
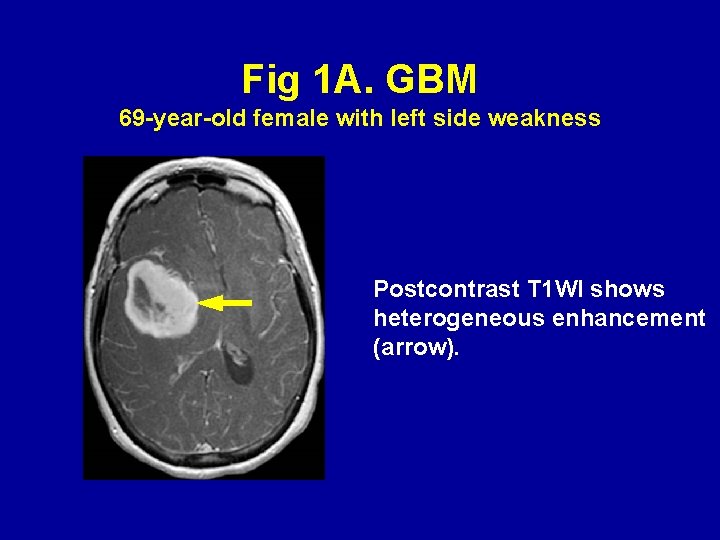
Fig 1 A. GBM 69 -year-old female with left side weakness Postcontrast T 1 WI shows heterogeneous enhancement (arrow).
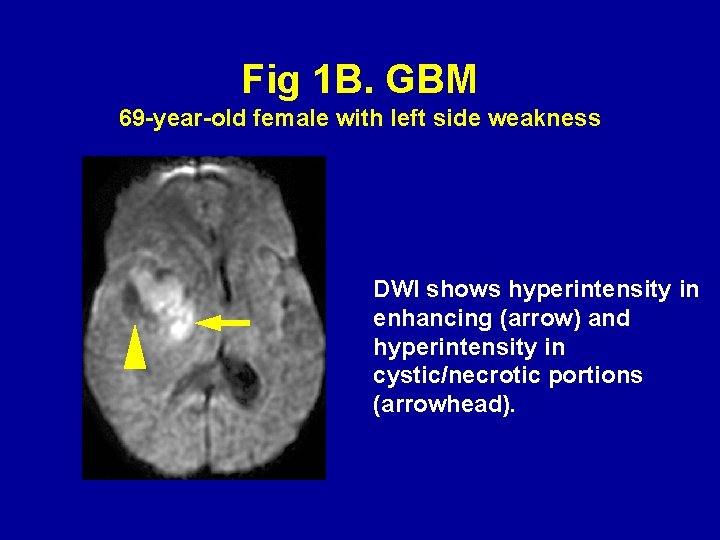
Fig 1 B. GBM 69 -year-old female with left side weakness DWI shows hyperintensity in enhancing (arrow) and hyperintensity in cystic/necrotic portions (arrowhead).
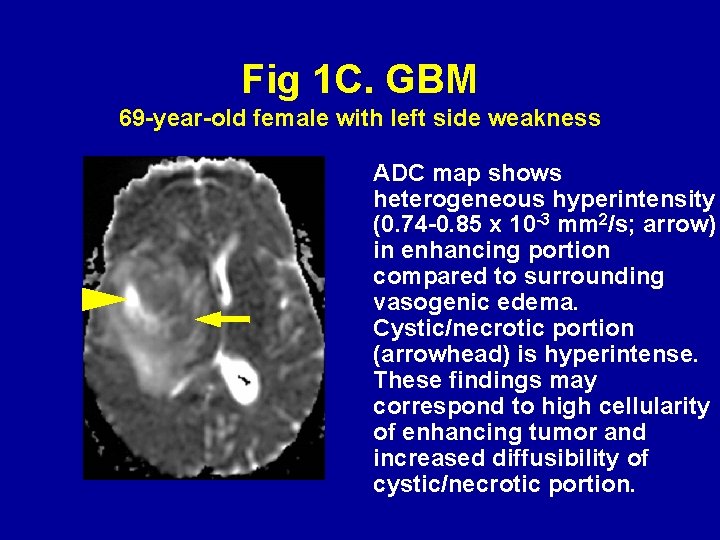
Fig 1 C. GBM 69 -year-old female with left side weakness ADC map shows heterogeneous hyperintensity (0. 74 -0. 85 x 10 -3 mm 2/s; arrow) in enhancing portion compared to surrounding vasogenic edema. Cystic/necrotic portion (arrowhead) is hyperintense. These findings may correspond to high cellularity of enhancing tumor and increased diffusibility of cystic/necrotic portion.
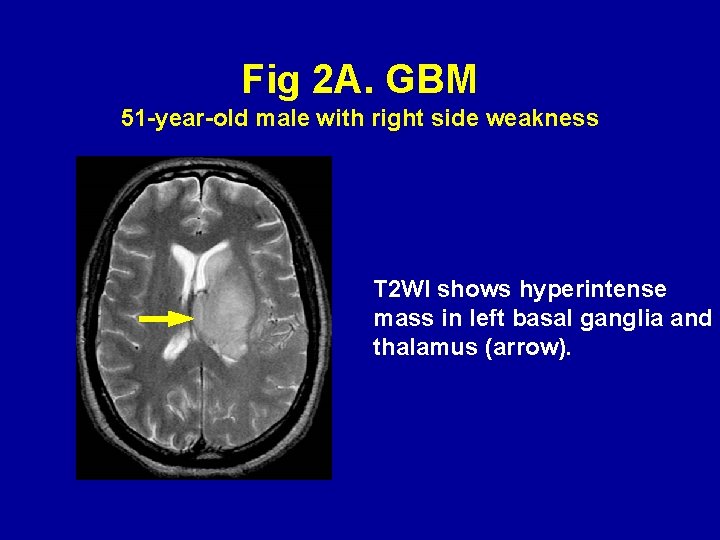
Fig 2 A. GBM 51 -year-old male with right side weakness T 2 WI shows hyperintense mass in left basal ganglia and thalamus (arrow).
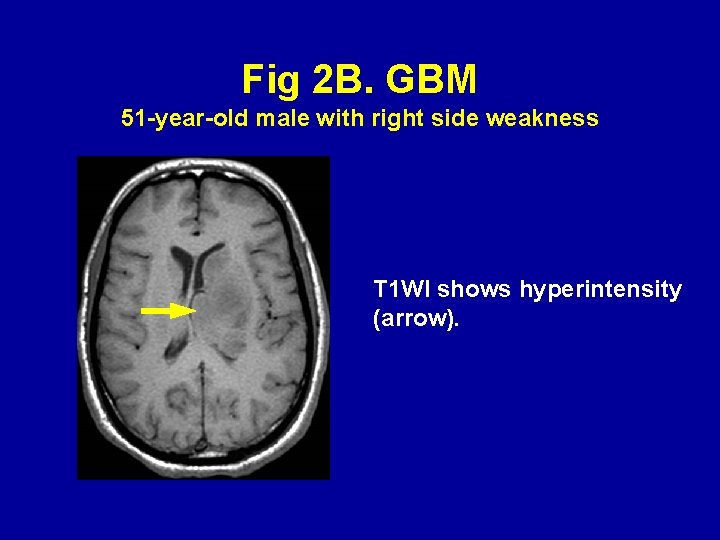
Fig 2 B. GBM 51 -year-old male with right side weakness T 1 WI shows hyperintensity (arrow).
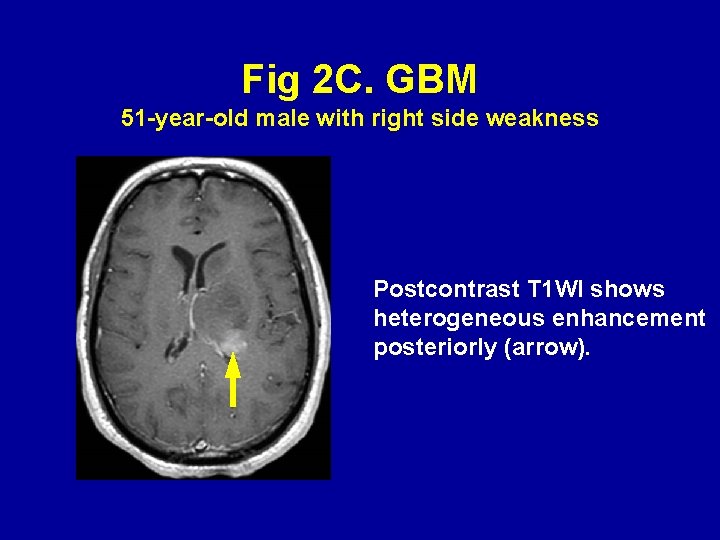
Fig 2 C. GBM 51 -year-old male with right side weakness Postcontrast T 1 WI shows heterogeneous enhancement posteriorly (arrow).
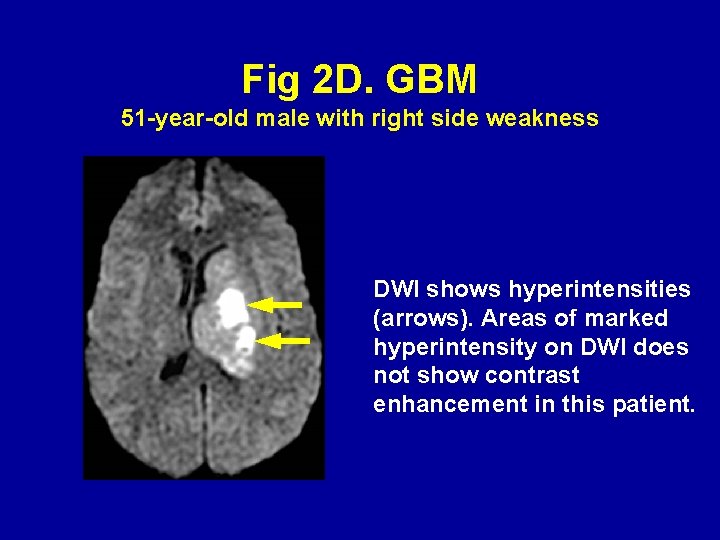
Fig 2 D. GBM 51 -year-old male with right side weakness DWI shows hyperintensities (arrows). Areas of marked hyperintensity on DWI does not show contrast enhancement in this patient.
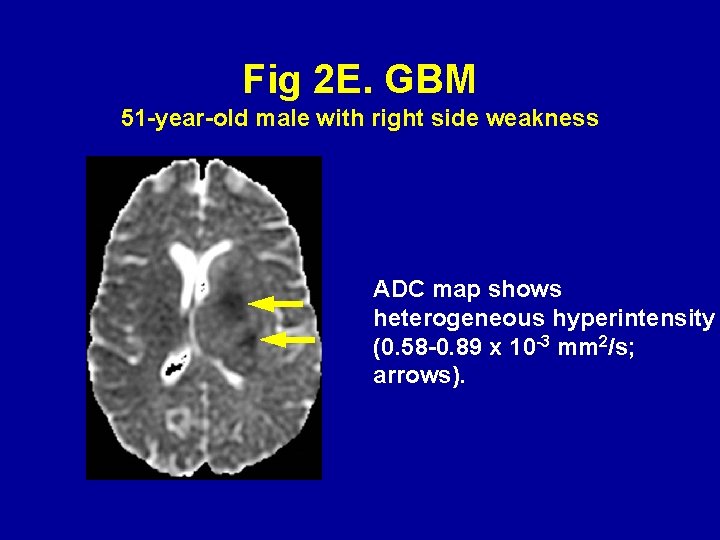
Fig 2 E. GBM 51 -year-old male with right side weakness ADC map shows heterogeneous hyperintensity (0. 58 -0. 89 x 10 -3 mm 2/s; arrows).
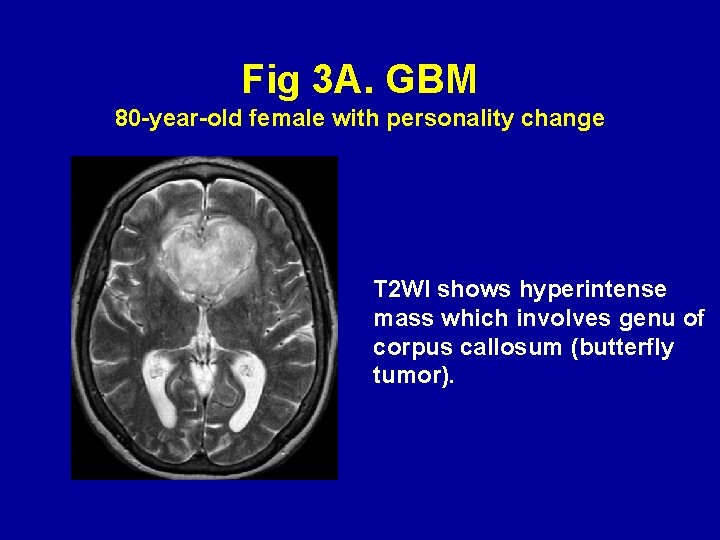
Fig 3 A. GBM 80 -year-old female with personality change T 2 WI shows hyperintense mass which involves genu of corpus callosum (butterfly tumor).
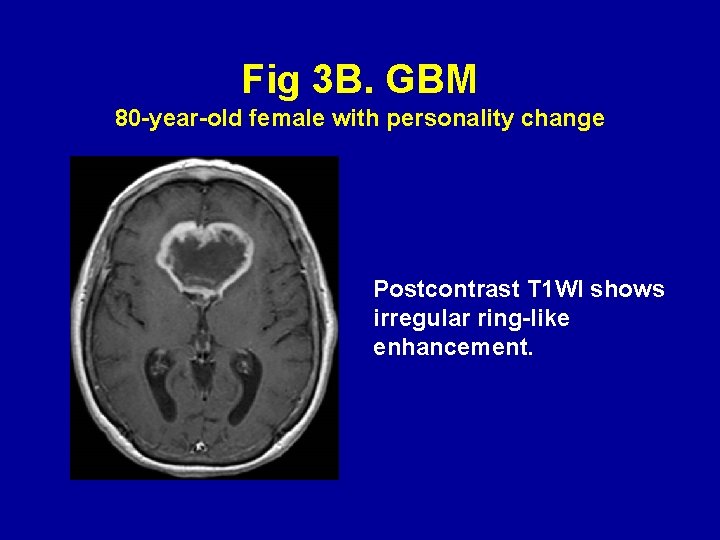
Fig 3 B. GBM 80 -year-old female with personality change Postcontrast T 1 WI shows irregular ring-like enhancement.
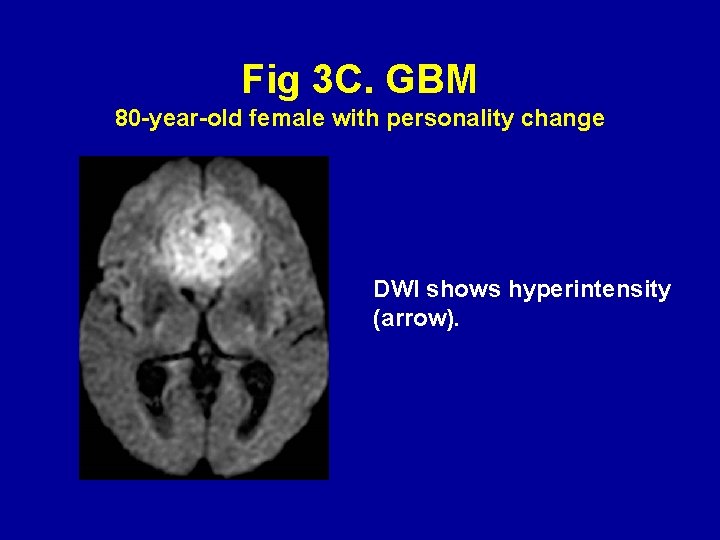
Fig 3 C. GBM 80 -year-old female with personality change DWI shows hyperintensity (arrow).
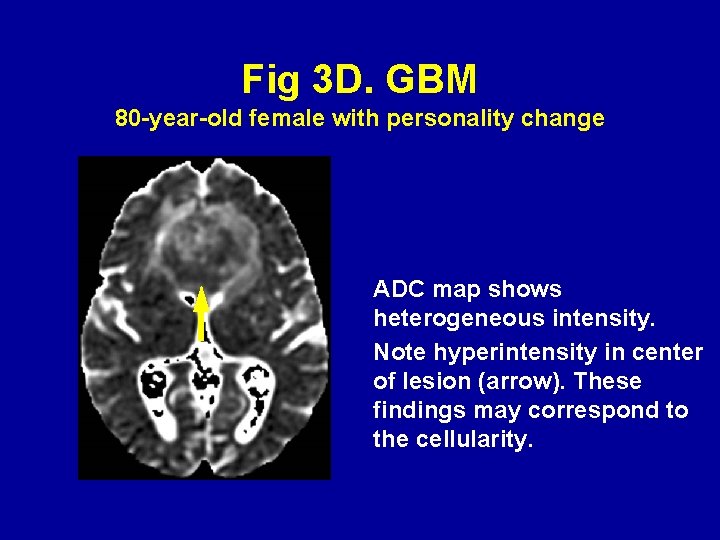
Fig 3 D. GBM 80 -year-old female with personality change ADC map shows heterogeneous intensity. Note hyperintensity in center of lesion (arrow). These findings may correspond to the cellularity.
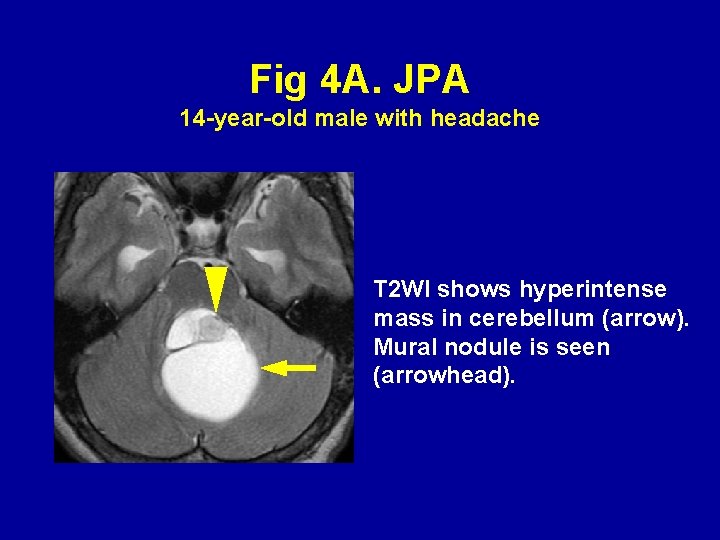
Fig 4 A. JPA 14 -year-old male with headache T 2 WI shows hyperintense mass in cerebellum (arrow). Mural nodule is seen (arrowhead).
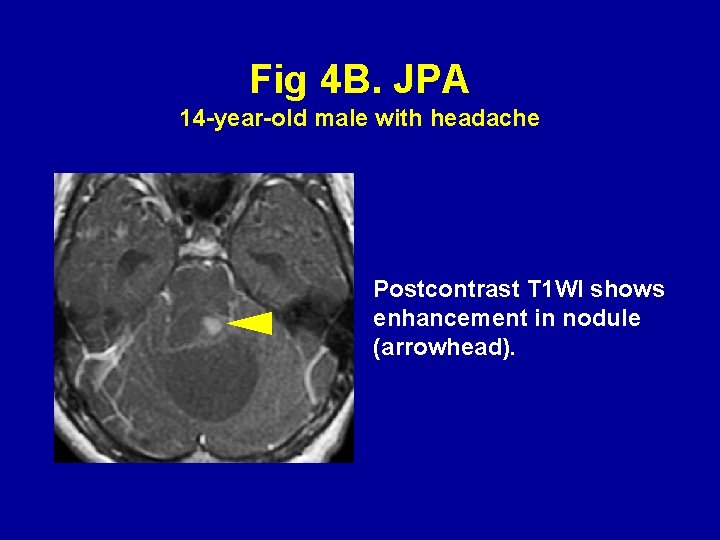
Fig 4 B. JPA 14 -year-old male with headache Postcontrast T 1 WI shows enhancement in nodule (arrowhead).
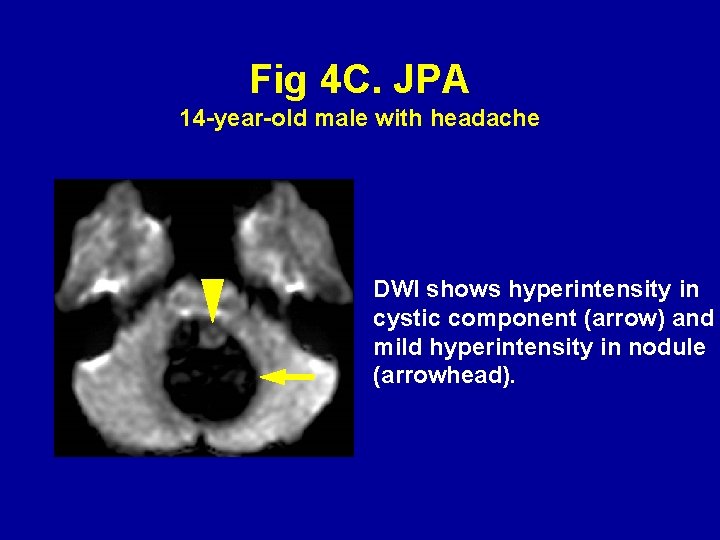
Fig 4 C. JPA 14 -year-old male with headache DWI shows hyperintensity in cystic component (arrow) and mild hyperintensity in nodule (arrowhead).
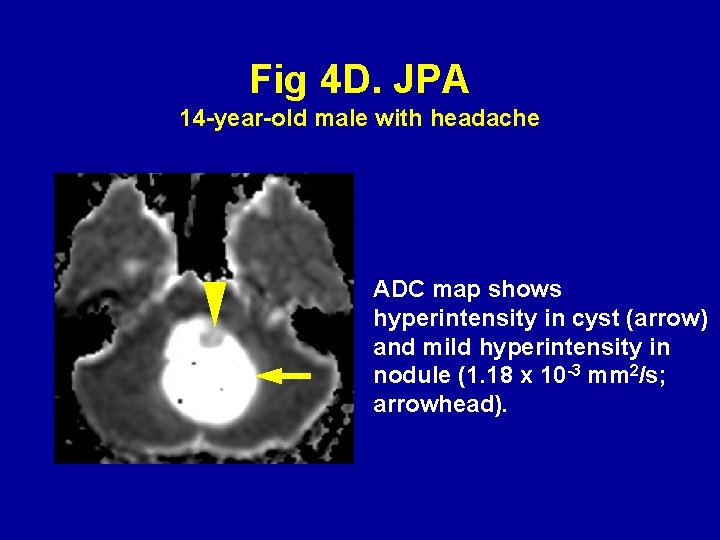
Fig 4 D. JPA 14 -year-old male with headache ADC map shows hyperintensity in cyst (arrow) and mild hyperintensity in nodule (1. 18 x 10 -3 mm 2/s; arrowhead).
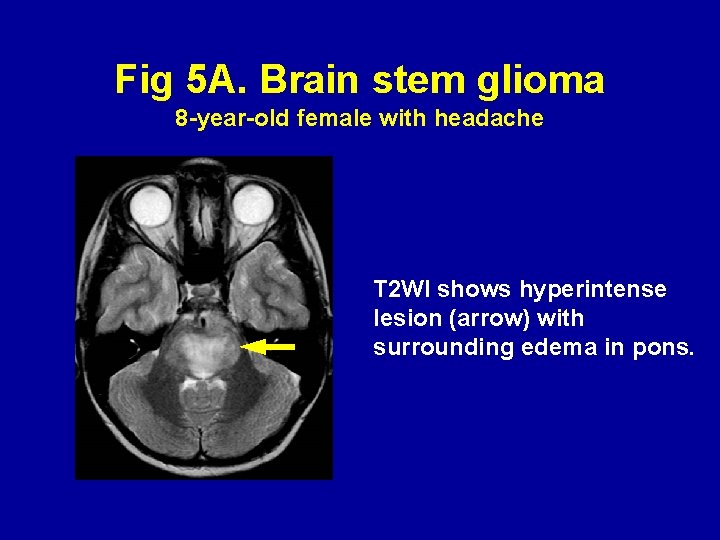
Fig 5 A. Brain stem glioma 8 -year-old female with headache T 2 WI shows hyperintense lesion (arrow) with surrounding edema in pons.
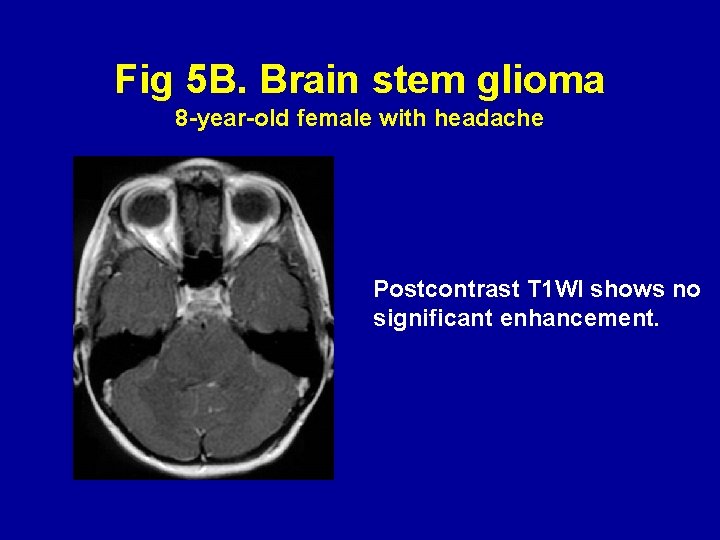
Fig 5 B. Brain stem glioma 8 -year-old female with headache Postcontrast T 1 WI shows no significant enhancement.
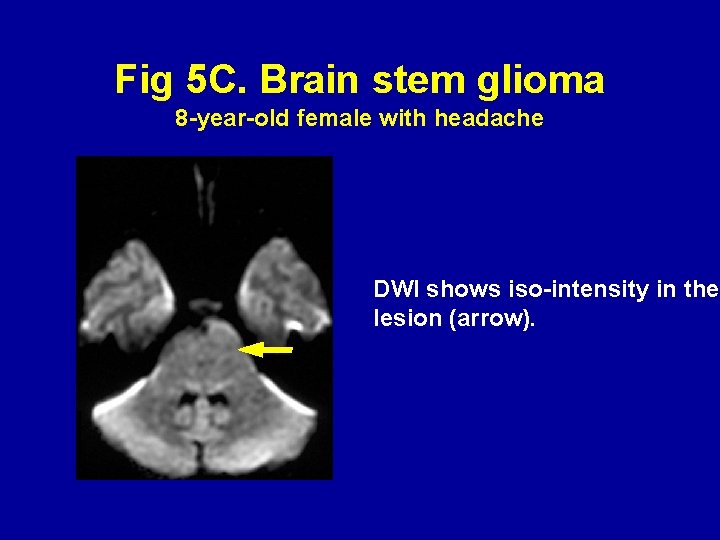
Fig 5 C. Brain stem glioma 8 -year-old female with headache DWI shows iso-intensity in the lesion (arrow).
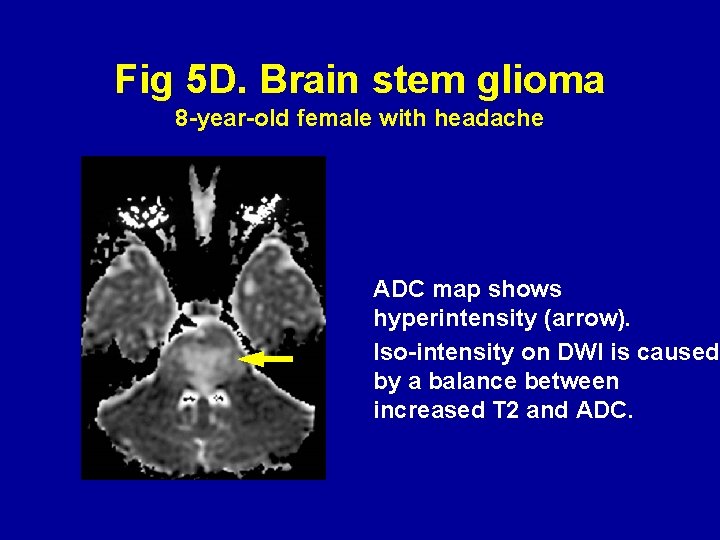
Fig 5 D. Brain stem glioma 8 -year-old female with headache ADC map shows hyperintensity (arrow). Iso-intensity on DWI is caused by a balance between increased T 2 and ADC.
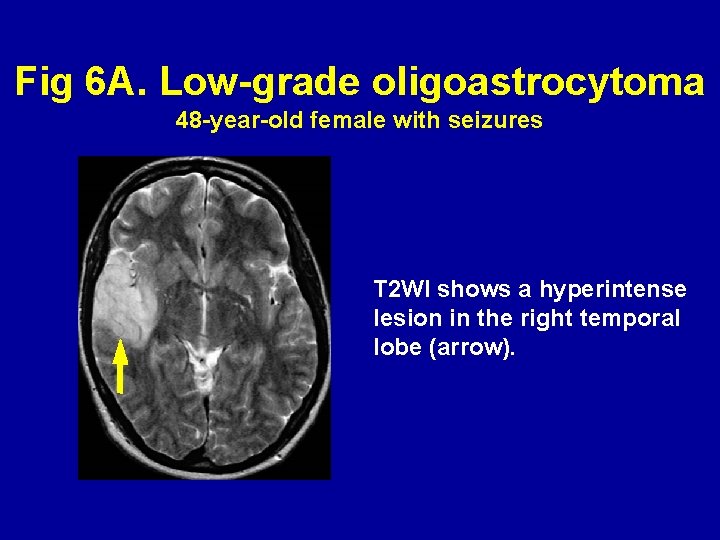
Fig 6 A. Low-grade oligoastrocytoma 48 -year-old female with seizures T 2 WI shows a hyperintense lesion in the right temporal lobe (arrow).
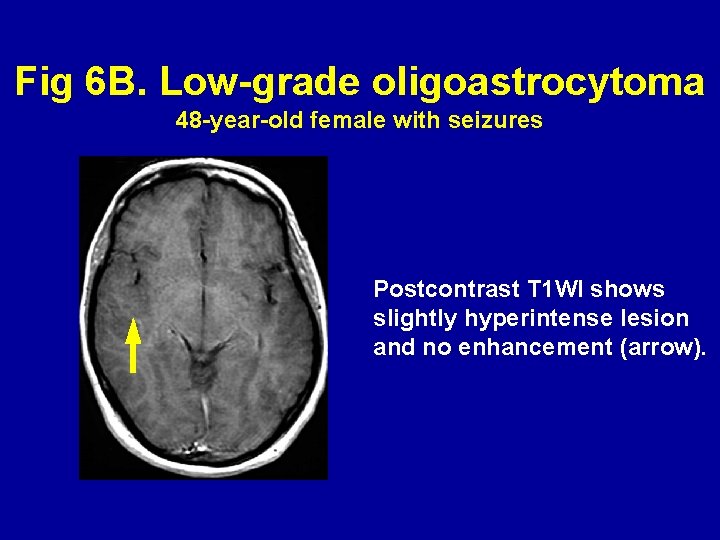
Fig 6 B. Low-grade oligoastrocytoma 48 -year-old female with seizures Postcontrast T 1 WI shows slightly hyperintense lesion and no enhancement (arrow).
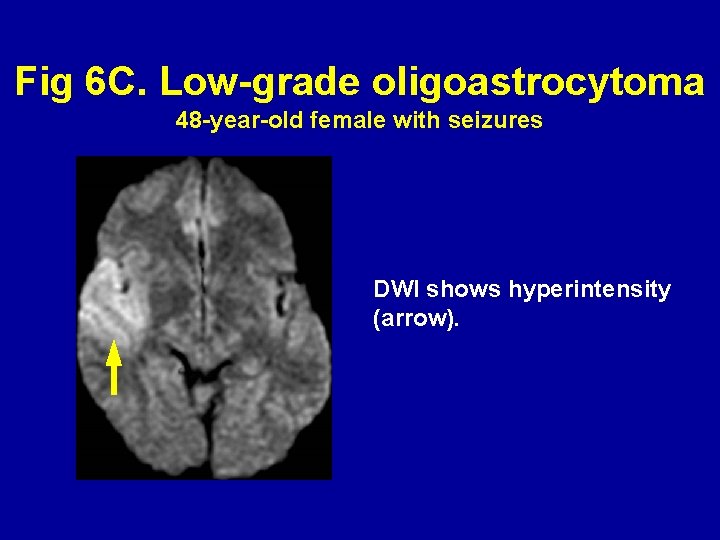
Fig 6 C. Low-grade oligoastrocytoma 48 -year-old female with seizures DWI shows hyperintensity (arrow).
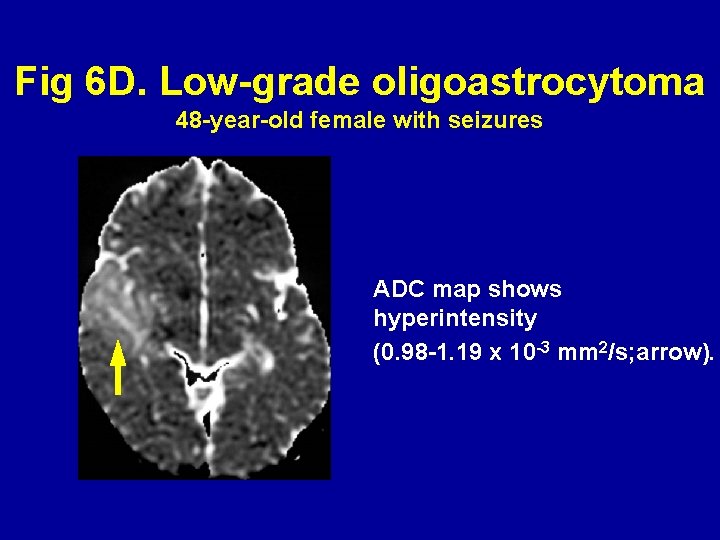
Fig 6 D. Low-grade oligoastrocytoma 48 -year-old female with seizures ADC map shows hyperintensity (0. 98 -1. 19 x 10 -3 mm 2/s; arrow).

Epidermoid tumor (Fig 7) Epidermoid tumors are benign neoplasms of ectodermal origin with stratified squamous epithelium and keratinaceous debris (13, 24 -31). They are hyperintense on DWI with decreased ADC. The ADC of epidermoid tumors has been reported to be lower than cerebrospinal fluid and equal or higher than brain parenchyma (24 -26, 28, 29, 31). Therefore, the hyperintensity of epidermoid tumors on DWI is primarily caused by T 2 shine-through effect.
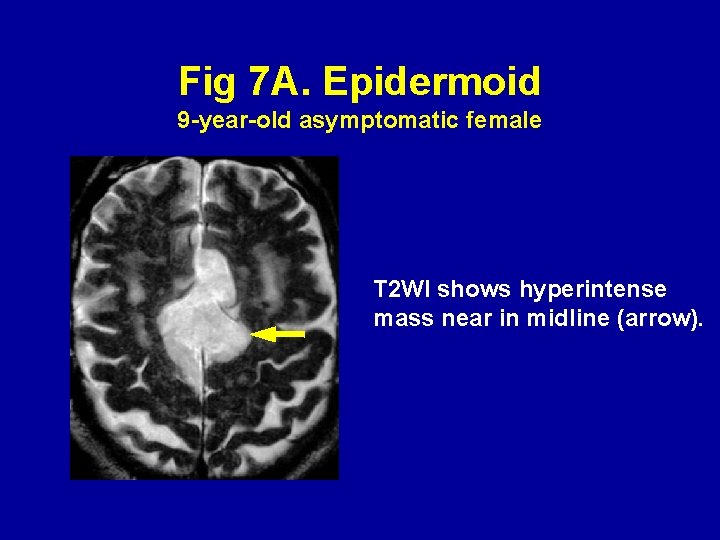
Fig 7 A. Epidermoid 9 -year-old asymptomatic female T 2 WI shows hyperintense mass near in midline (arrow).
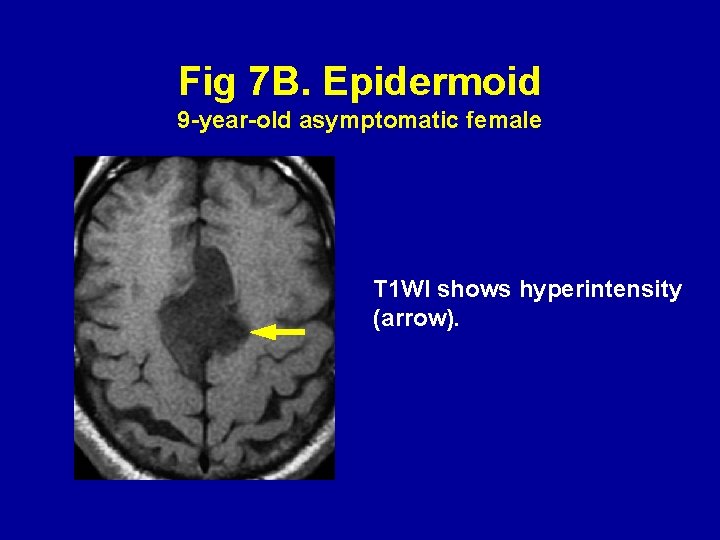
Fig 7 B. Epidermoid 9 -year-old asymptomatic female T 1 WI shows hyperintensity (arrow).
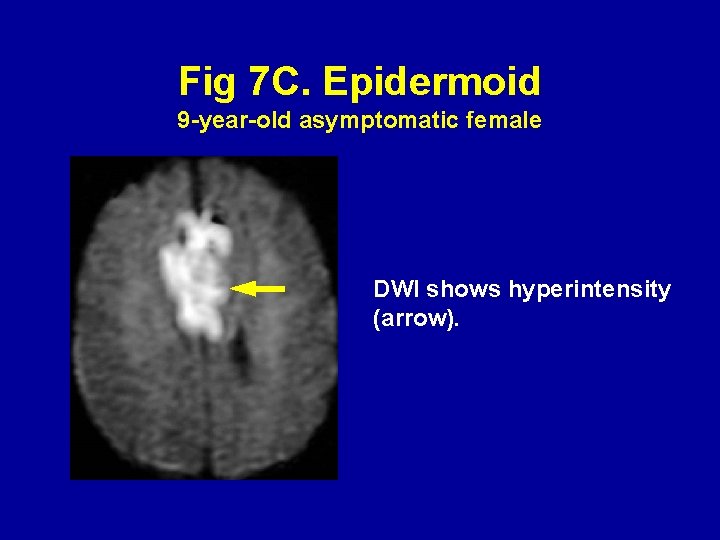
Fig 7 C. Epidermoid 9 -year-old asymptomatic female DWI shows hyperintensity (arrow).
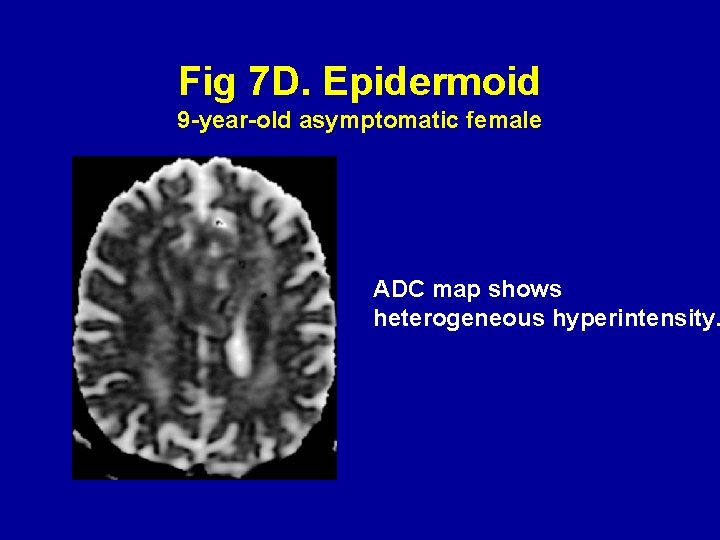
Fig 7 D. Epidermoid 9 -year-old asymptomatic female ADC map shows heterogeneous hyperintensity.

Arachnoid cysts (Fig 8) Arachnoid cysts have a similar appearance on routine MR imaging as epidermoid tumors, but it is well-known that DWI can distinguish the two (24 -31). Arachnoid cysts are hyperintense on DWI and their MR signal characteristics are similar to cerebrospinal fluid.
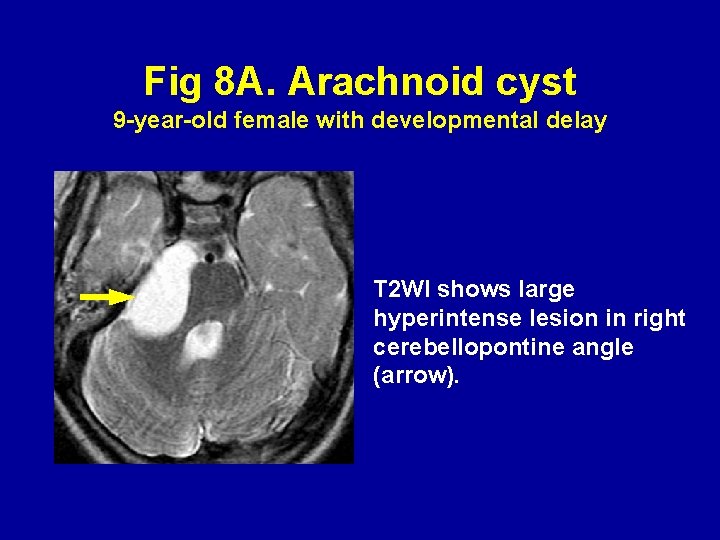
Fig 8 A. Arachnoid cyst 9 -year-old female with developmental delay T 2 WI shows large hyperintense lesion in right cerebellopontine angle (arrow).
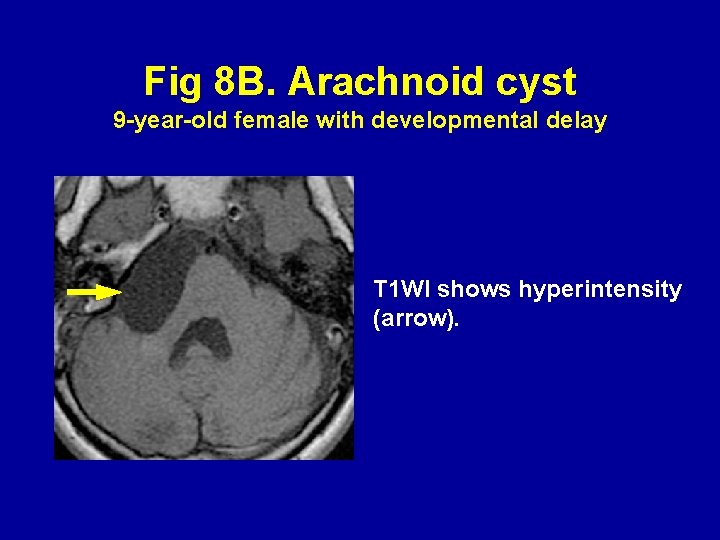
Fig 8 B. Arachnoid cyst 9 -year-old female with developmental delay T 1 WI shows hyperintensity (arrow).

Fig 8 C. Arachnoid cyst 9 -year-old female with developmental delay DWI shows hyperintensity (arrow).
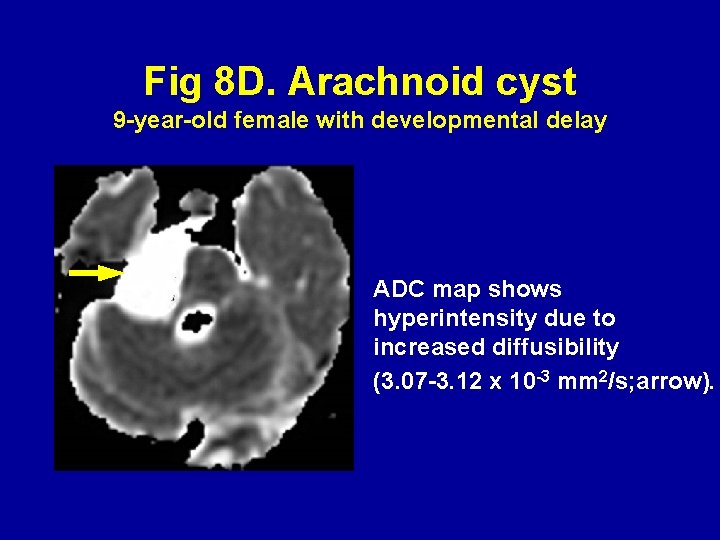
Fig 8 D. Arachnoid cyst 9 -year-old female with developmental delay ADC map shows hyperintensity due to increased diffusibility (3. 07 -3. 12 x 10 -3 mm 2/s; arrow).

Primitive neuroectodermal tumor (PNET) (Fig. 9) PNET is a group of histologically similar tumors that occur mostly in children. They include embryonal, largely undifferentiated tumors, such as medulloblastoma, neuroblastoma, pineoblastoma, ependymoblastoma and medulloepithelioma. These tumors have a high cellular density and are typically hyperintensity on DWI with decreased ADC (32 -35).
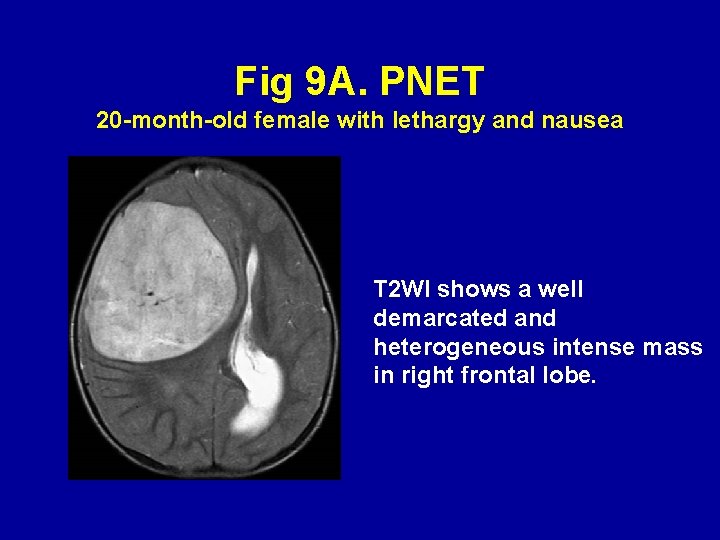
Fig 9 A. PNET 20 -month-old female with lethargy and nausea T 2 WI shows a well demarcated and heterogeneous intense mass in right frontal lobe.
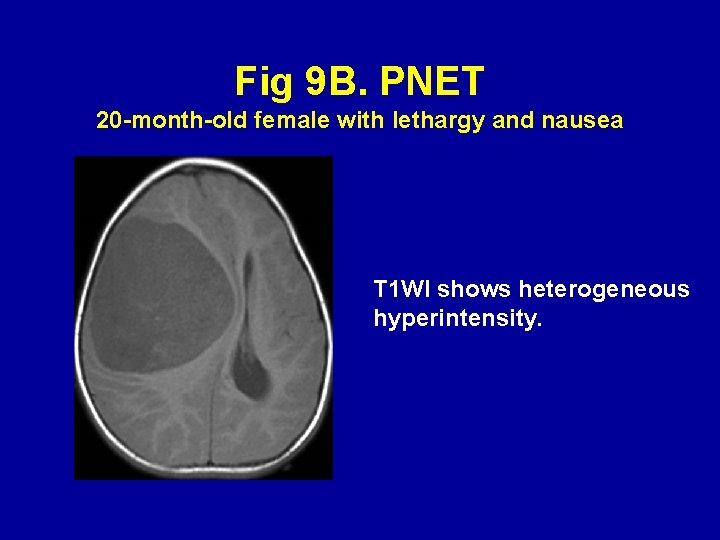
Fig 9 B. PNET 20 -month-old female with lethargy and nausea T 1 WI shows heterogeneous hyperintensity.
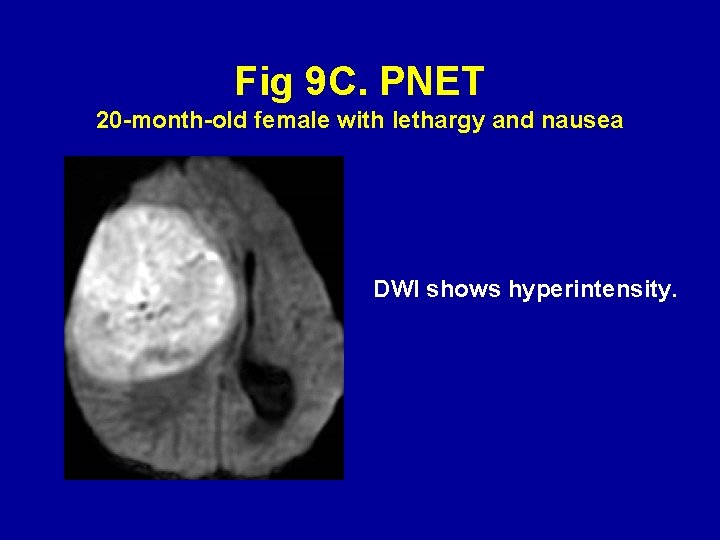
Fig 9 C. PNET 20 -month-old female with lethargy and nausea DWI shows hyperintensity.
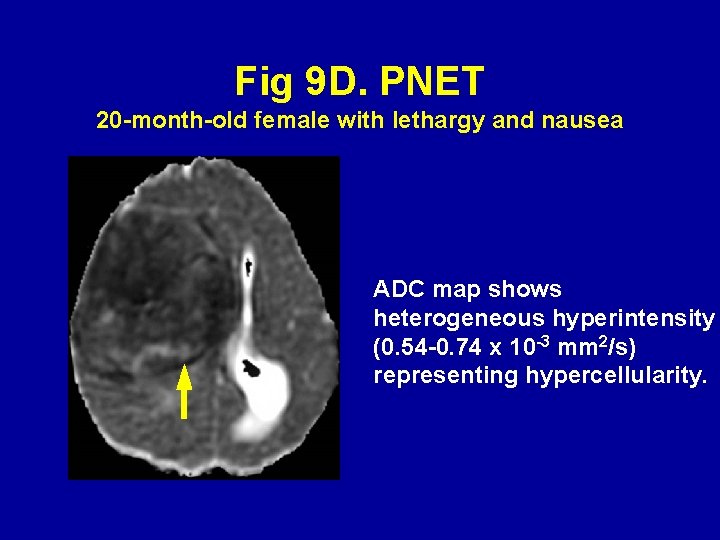
Fig 9 D. PNET 20 -month-old female with lethargy and nausea ADC map shows heterogeneous hyperintensity (0. 54 -0. 74 x 10 -3 mm 2/s) representing hypercellularity.

Meningiomas (Figs 10, 11) Most benign meningiomas are iso-intense on DWI and ADC maps, but some are slightly hyperintense on both DWI and ADC maps. Malignant or atypical meningiomas have markedly increased signal intensity on DWI and decreased ADC due to a high tumor cellularity (12, 18, 36). However, other factors, such as multifocal areas of necrosis, numerous abnormal mitoses and cytologic pleomorphism may also cause the high DWI signal (11, 12, 18, 36, 37).
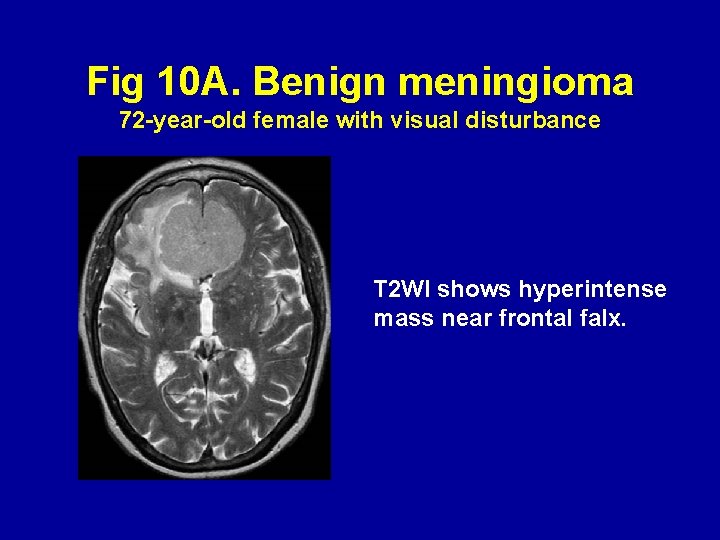
Fig 10 A. Benign meningioma 72 -year-old female with visual disturbance T 2 WI shows hyperintense mass near frontal falx.
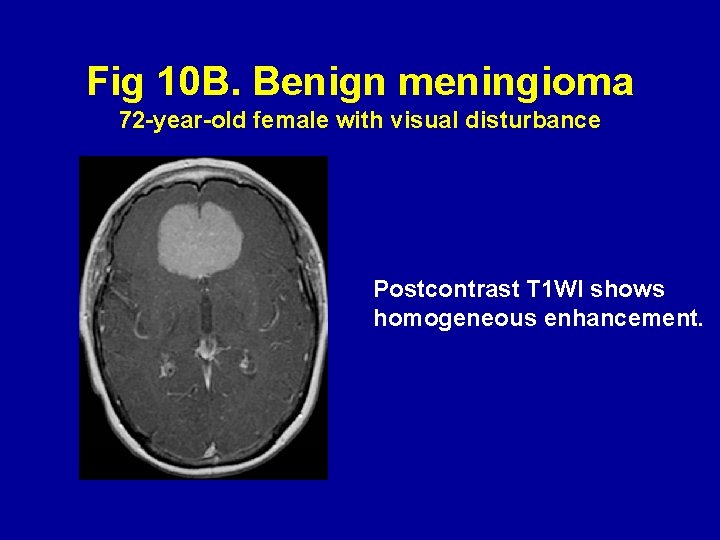
Fig 10 B. Benign meningioma 72 -year-old female with visual disturbance Postcontrast T 1 WI shows homogeneous enhancement.
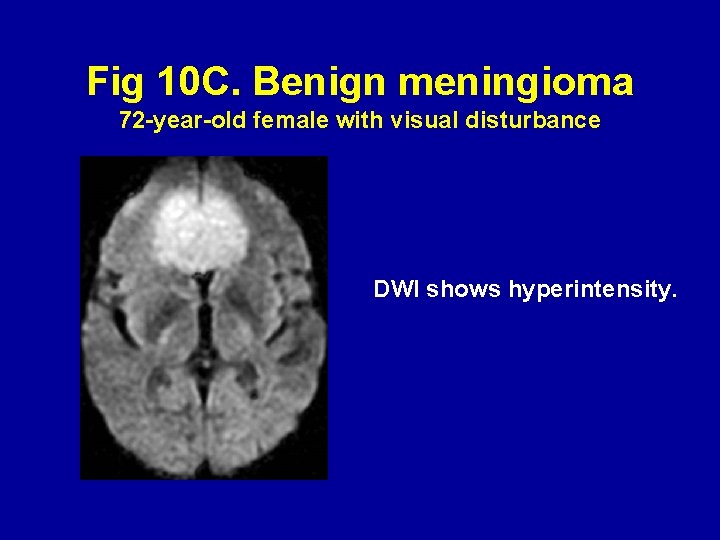
Fig 10 C. Benign meningioma 72 -year-old female with visual disturbance DWI shows hyperintensity.
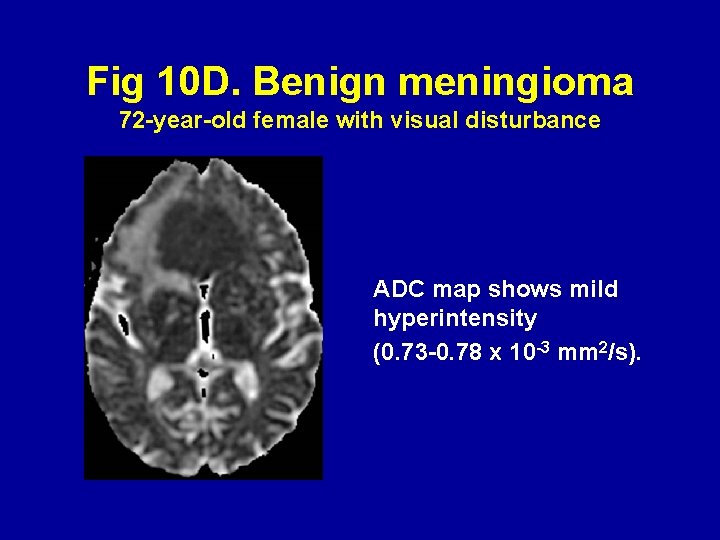
Fig 10 D. Benign meningioma 72 -year-old female with visual disturbance ADC map shows mild hyperintensity (0. 73 -0. 78 x 10 -3 mm 2/s).
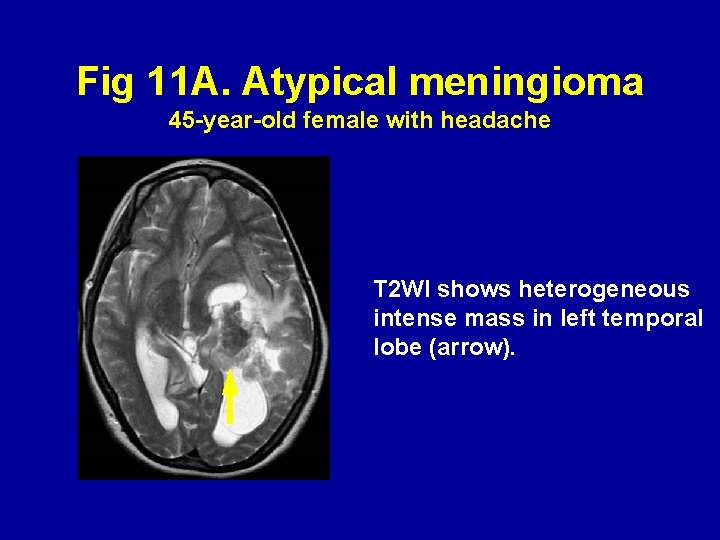
Fig 11 A. Atypical meningioma 45 -year-old female with headache T 2 WI shows heterogeneous intense mass in left temporal lobe (arrow).
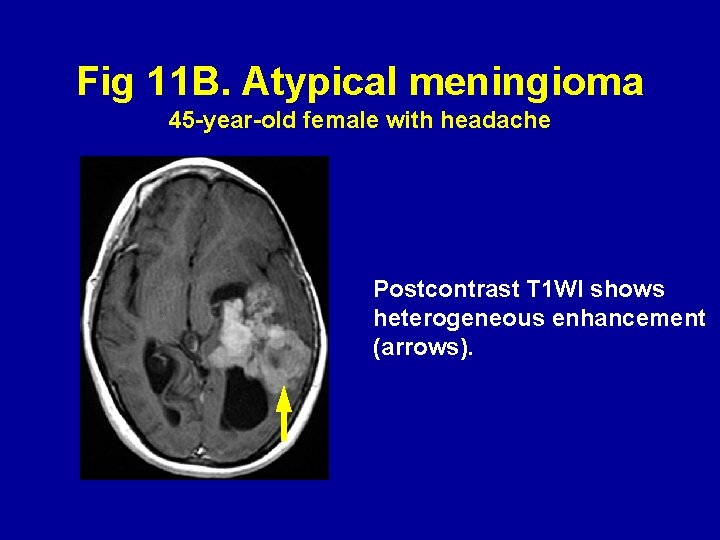
Fig 11 B. Atypical meningioma 45 -year-old female with headache Postcontrast T 1 WI shows heterogeneous enhancement (arrows).
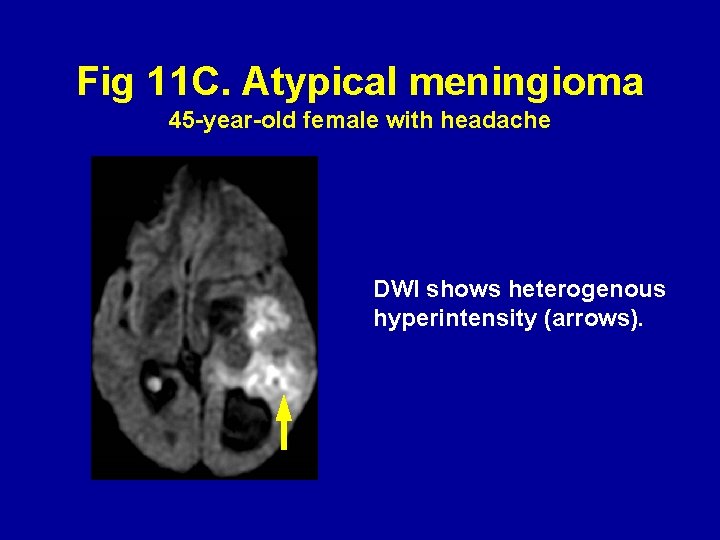
Fig 11 C. Atypical meningioma 45 -year-old female with headache DWI shows heterogenous hyperintensity (arrows).
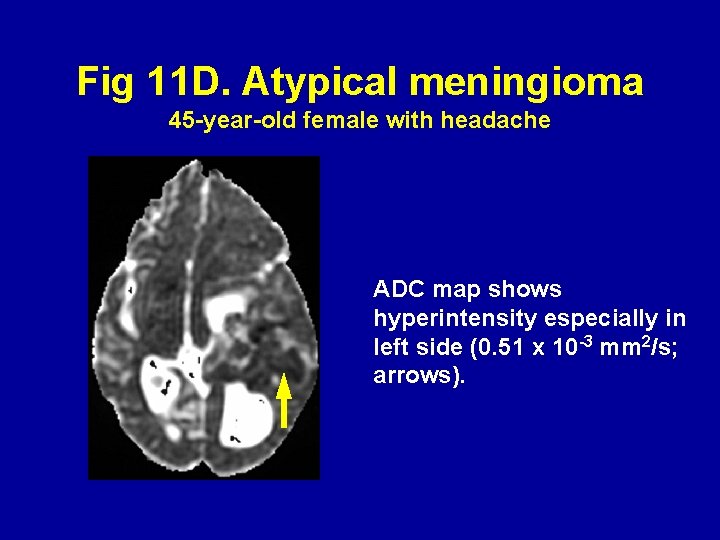
Fig 11 D. Atypical meningioma 45 -year-old female with headache ADC map shows hyperintensity especially in left side (0. 51 x 10 -3 mm 2/s; arrows).

Malignant lymphomas (Fig. 12) Most lymphomas are iso- or hyperintense on T 2 WI and show homogeneous enhancement in immunocompetent patients. But in immunosuppressed patients a rim enhancement is a more common finding (18, 38 -40). The enhancing components of lymphomas are generally hyperintense on DWI (16, 18, 41). ADC of lymphomas is often lower than in high grade gliomas (16). As mentioned before, this corresponds to the hypercellularity and might help in differentiating between lymphomas and high grade gliomas (16).
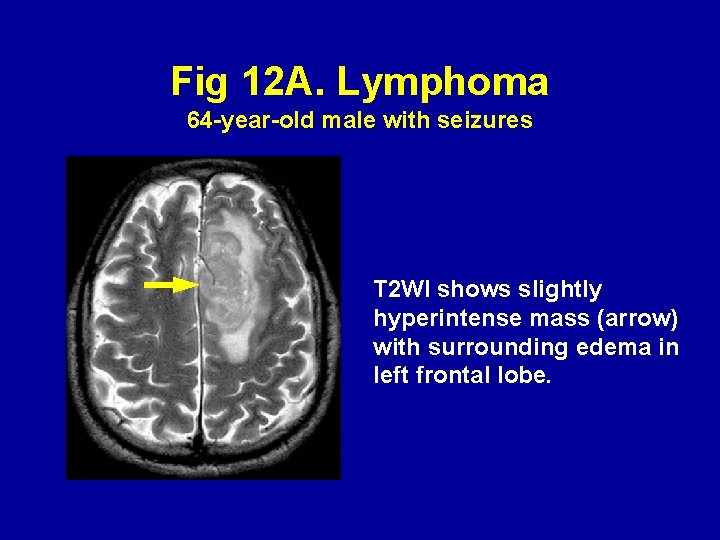
Fig 12 A. Lymphoma 64 -year-old male with seizures T 2 WI shows slightly hyperintense mass (arrow) with surrounding edema in left frontal lobe.
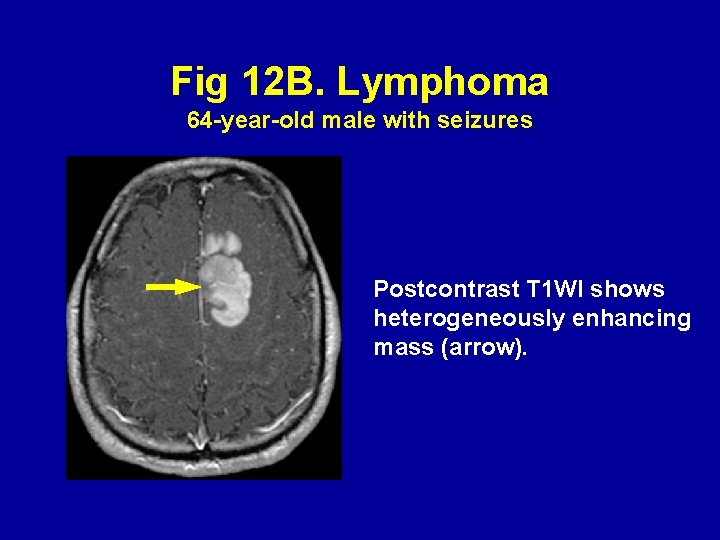
Fig 12 B. Lymphoma 64 -year-old male with seizures Postcontrast T 1 WI shows heterogeneously enhancing mass (arrow).
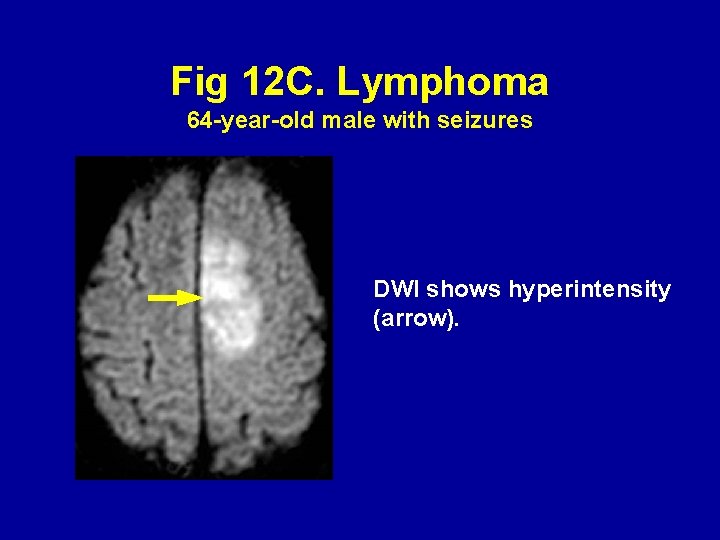
Fig 12 C. Lymphoma 64 -year-old male with seizures DWI shows hyperintensity (arrow).
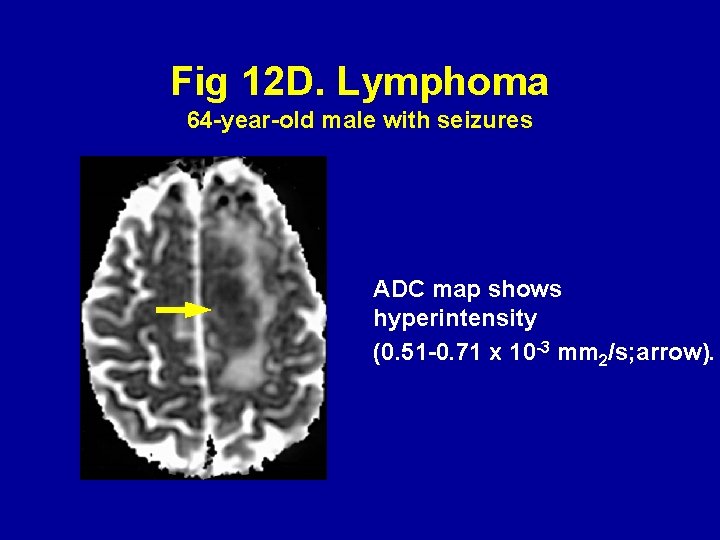
Fig 12 D. Lymphoma 64 -year-old male with seizures ADC map shows hyperintensity (0. 51 -0. 71 x 10 -3 mm 2/s; arrow).

Craniopharyngiomas (Fig. 13) Craniopharyngiomas typically show a combination of contrast enhancement, cyst formation and calcifications. Signal intensity on DWI depends on ADC and T 2 values (42).
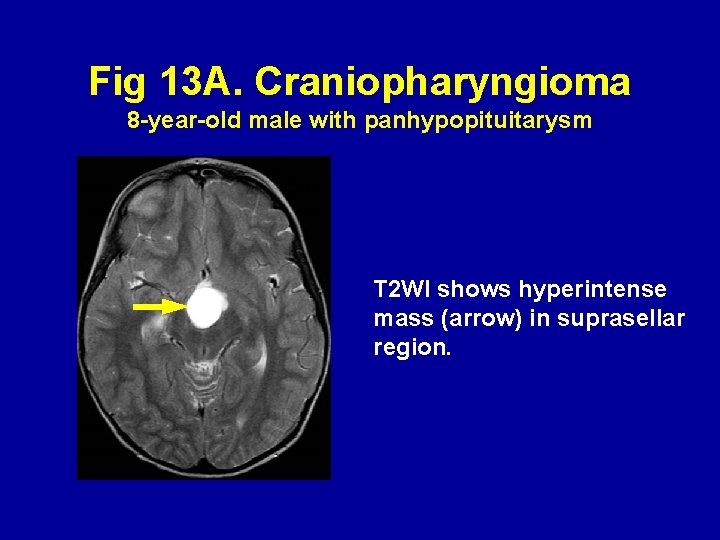
Fig 13 A. Craniopharyngioma 8 -year-old male with panhypopituitarysm T 2 WI shows hyperintense mass (arrow) in suprasellar region.
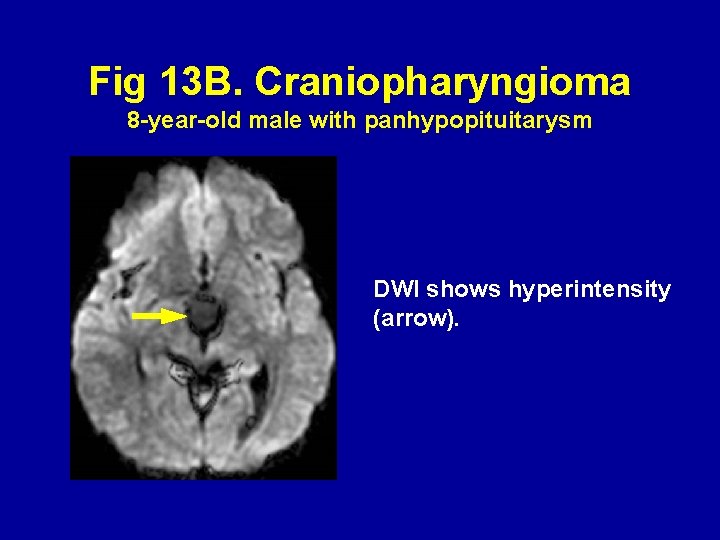
Fig 13 B. Craniopharyngioma 8 -year-old male with panhypopituitarysm DWI shows hyperintensity (arrow).
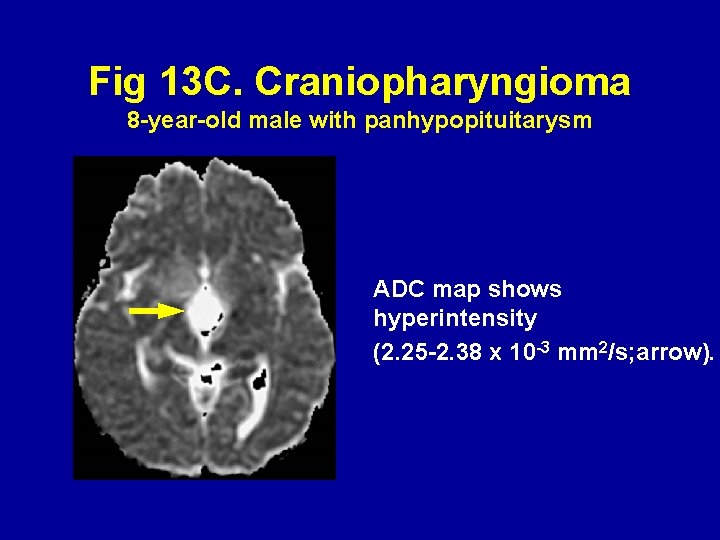
Fig 13 C. Craniopharyngioma 8 -year-old male with panhypopituitarysm ADC map shows hyperintensity (2. 25 -2. 38 x 10 -3 mm 2/s; arrow).

Metastases (Fig 14) The signal intensity of solid components of metastases on DWI is variable and depends on their T 2 and ADC (2, 6, 11, 12, 18, 19, 30, 43 -51). DWI findings of metastasis are similar to that of gliomas, probably reflecting the cellularity of the primary tumor (6, 11, 18, 30). Common signal intensity of necrotic/cystic components of metastases may relate to an increase in free water, showing hyperintensity on DWI with increased ADC. However, in the presence of extracellular methemoglobin and/or increased viscosity, DWI can show hyperintensity with decreased ADC (18, 43 -51).
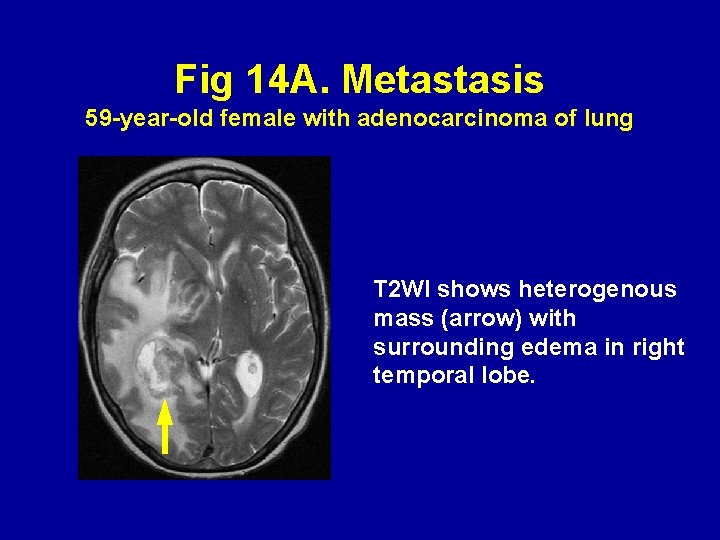
Fig 14 A. Metastasis 59 -year-old female with adenocarcinoma of lung T 2 WI shows heterogenous mass (arrow) with surrounding edema in right temporal lobe.
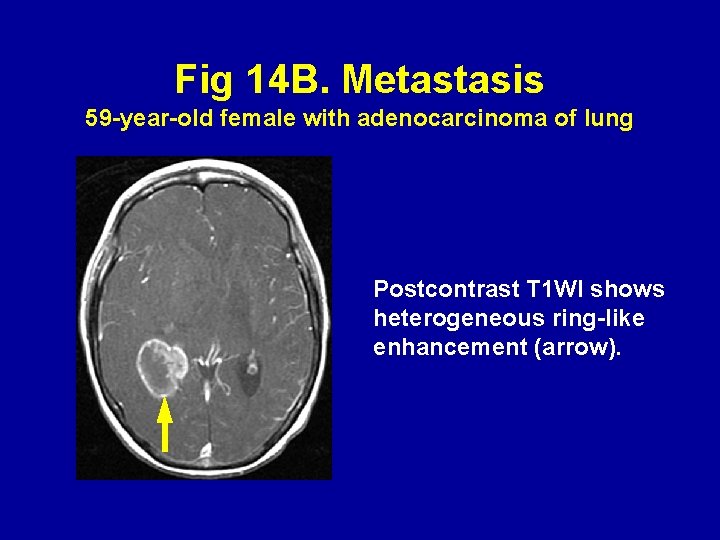
Fig 14 B. Metastasis 59 -year-old female with adenocarcinoma of lung Postcontrast T 1 WI shows heterogeneous ring-like enhancement (arrow).
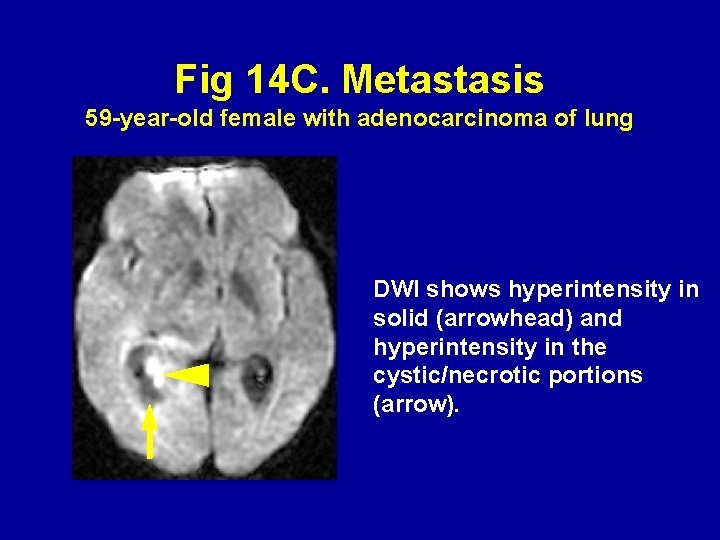
Fig 14 C. Metastasis 59 -year-old female with adenocarcinoma of lung DWI shows hyperintensity in solid (arrowhead) and hyperintensity in the cystic/necrotic portions (arrow).
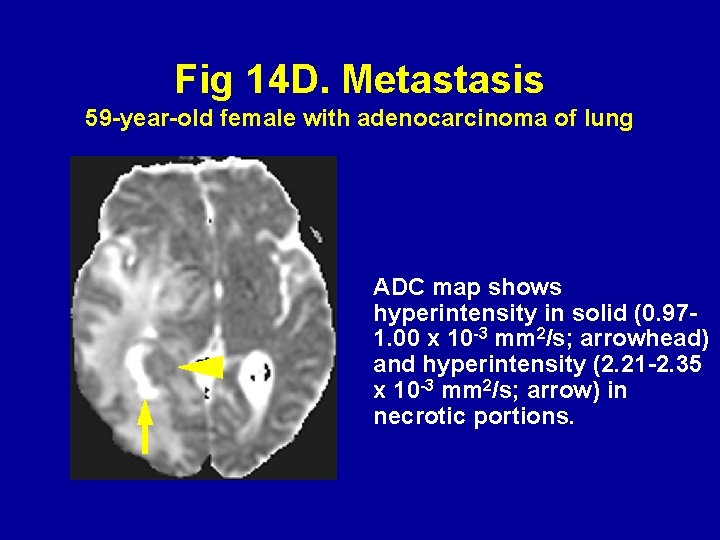
Fig 14 D. Metastasis 59 -year-old female with adenocarcinoma of lung ADC map shows hyperintensity in solid (0. 971. 00 x 10 -3 mm 2/s; arrowhead) and hyperintensity (2. 21 -2. 35 x 10 -3 mm 2/s; arrow) in necrotic portions.

Conclusion DW imaging can provide additional information about tumor cellularity and help in the characterization and grading of tumors. The ability of specific tumor differentiation and determination of tumor infiltration is difficult in most situations.

References (1/5) 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Le. Bihan D, Breton E, Lallemand D, Grenier P, Cabanis E, Laval-Jeantet M. MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurologic disorders. Radiology 1986 161: 401 -7 Hajnal JV, Doran M, Hall AS, et al. MR imaging of anisotropically restricted diffusion of water in the nervous system: technical, anatomic, and pathologic considerations. J Comput Assist Tomogr 1991 15: 1 -18 Tien RD, Felsberg GJ, Friedman H, Brown M, Mac. Fall J. MR imaging of high-grade cerebral gliomas: value of diffusion-weighted echoplanar pulse sequences. AJR Am J Roentgenol 1994; 162: 671 -677 Eis M, Els T, Hoehn-Berlage M, Hossmann KA. Quantitative diffusion MR imaging of cerebral tumor and edema. Acta Neurochir Suppl (Wien) 1994; 60: 344 -6 Brunberg JA, Chenevert TL, Mc. Keever PE, et al. In vivo MR determination of water diffusion coefficients and diffusion anisotropy: correlation with structural alteration in gliomas of the cerebral hemispheres. AJNR Am J Neuroradiol 1995; 16: 361 -71 Krabbe K, Gideon P, Wagn P, Hansen U, Thomsen C, Madsen F. MR diffusion imaging of human intracranial tumours. Neuroradiology 1997; 39: 483 -9 Gupta RK, Sinha U, Cloughesy TF, Alger JR. Inverse correlation between choline magnetic resonance spectroscopy signal intensity and the apparent diffusion coefficient in human glioma. Magn Reson Med 1999; 41: 2 -7 Sugahara T, Korogi Y, Kochi M, et al. Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 1999; 9: 53 -60 Gupta RK, Cloughesy TF, Sinha U, et al. Relationships between choline magnetic resonance spectroscopy, apparent diffusion coefficient and quantitative histopathology in human glioma. J Neurooncol 2000; 50: 215 -226 Castillo M, Smith JK, Kwock L, Wilber K. Apparent diffusion coefficients in the evaluation of high-grade cerebral gliomas. AJNR Am J Neuroradiol 2001; 22: 60 -64

References (2/5) 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. Stadnik TW, Chaskis C, Michotte A, et al. Diffusion-weighted MR imaging of intracerebral masses: comparison with conventional MR imaging and histologic findings. AJNR Am J Neuroradiol 2001; 22: 969976 Kono K, Inoue Y, Nakayama K, et al. The role of diffusion-weighted imaging in patients with brain tumors. AJNR Am J Neuroradiol 2001; 22: 1081 -1088 Gauvain KM, Mc. Kinstry RC, Mukherjee P, et al. Evaluating pediatric brain tumor cellularity with diffusiontensor imaging. AJR Am J Roentgenol 2001; 177: 449 -54 Sinha S, Bastin ME, Whittle IR, Wardlaw JM. Diffusion tensor MR imaging of high-grade cerebral gliomas. AJNR Am J Neuroradiol 2002; 23: 520 -7 Bastin ME, Sinha S, Whittle IR, Wardlaw JM. Measurements of water diffusion and T 1 values in peritumoural oedematous brain. Neuroreport 2002; 13: 1335 -40 Guo AC, Cummings TJ, Dash RC, Provenzale JM. Lymphomas and high-grade astrocytomas: comparison of water diffusibility and histologic characteristics. Radiology 2002; 224: 177 -83 Yang D, Korogi Y, Sugahara T, et al. Cerebral gliomas: prospective comparison of multivoxel 2 D chemical -shift imaging proton MR spectroscopy, echoplanar perfusion and diffusion-weighted MRI. Neuroradiology 2002; 44: 656 -66 Stadnik TW, Demaerel P, Luypaert RR, et al. Imaging Tutorial: Differential Diagnosis of Bright Lesions on Diffusion-weighted MR Images. Radiographics 2003; 23: E 7 -7 Bulakbasi N, Kocaoglu M, Ors F, Tayfun C, Ucoz T. Combination of Single-Voxel Proton MR Spectroscopy and Apparent Diffusion Coefficient Calculation in the Evaluation of Common Brain Tumors. AJNR Am J Neuroradiol 2003; 24: 225 -33 Chenevert TL, Mc. Keever PE, Ross BD. Monitoring early response of experimental brain tumors to therapy using diffusion magnetic resonance imaging. Clin Cancer Res 1997; 3: 1457 -66

References (3/5) 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. Chenevert TL, Stegman LD, Taylor JM, et al. Diffusion magnetic resonance imaging: an early surrogate marker of therapeutic efficacy in brain tumors. J Natl Cancer Inst 2000; 92: 2029 -36 Mardor Y, Roth Y, Lidar Z, et al. Monitoring response to convection-enhanced taxol delivery in brain tumor patients using diffusion-weighted magnetic resonance imaging. Cancer Res 2001; 61: 4971 -3 Asao C, Korogi Y, Kitajima M Nishimura R, Baba Y, Kochi M, Yamashita Y. Diffusion-Weighted MR imaging of radiation-induced brain injury: The usefulness for differentiation from tumor recurrence. American Society of Neuroradiology 41 st Annual meeting 2003 proceddings, abstract 19, page 16. Tsuruda JS, Chew WM, Moseley ME, Norman D. Diffusion-weighted MR imaging of the brain: value of differentiating between extraaxial cysts and epidermoid tumors. AJNR Am J Neuroradiol 1990; 11: 925 -31 Tsuruda JS, Chew WM, Moseley ME, Norman D. Diffusion-weighted MR imaging of extraaxial tumors. Magn Reson Med 1991; 19: 316 -20 Maeda M, Kawamura Y, Tamagawa Y, Intravoxel incoherent motion (IVIM) MRI in intracranial, extraaxial tumors and cysts. J Comput Assist Tomogr 1992; 16: 514 -8 Laing AD, Mitchell PJ, Wallace D. Diffusion-weighted magnetic resonance imaging of intracranial epidermoid tumours. Australas Radiol 1999; 43: 16 -9 Dechambre S, Duprez T, Lecouvet F, Raftopoulos C, Gosnard G. Diffusion-weighted MRI postoperative assessment of an epidermoid tumour in the cerebellopontine angle. Neuroradiology 1999; 41: 829 -31 Chen S, Ikawa F, Kurisu K, Arita K, Takaba J, Kanou Y. Quantitative MR evaluation of intracranial epidermoid tumors by fast fluid-attenuated inversion recovery imaging and echo-planar diffusionweighted imaging. AJNR Am J Neuroradiol 2001; 22: 1089 -96 Bergui M, Zhong J, Bradac GB, Sales S. Diffusion-weighted images of intracranial cyst-like lesions. Neuroradiology 2001; 43: 824 -9

References (4/5) 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. Annet L, Duprez T, Grandin C, Dooms G, Collard A, Cosnard G. Apparent diffusion coefficient measurements within intracranial epidermoid cysts in six patients. Neuroradiology 2002; 44: 326 -8 Kotsenas AL, Roth TC, Manness WK, Faerber EN. Abnormal diffusion-weighted MRI in medulloblastoma: does it reflect small cell histology? Pediatr Radiol 1999; 29: 524 -6 Klisch J, Husstedt H, Hennings S, von Velthoven V, Pagenstecher A, Schumacher M. Supratentorial primitive neuroectodermal tumours: diffusion-weighted MRI. Neuroradiology 2000; 42: 393 -8 Wilke M, Eidenschink A, Muller-Weihrich S, Auer DP. MR diffusion imaging and 1 H spectroscopy in a child with medulloblastoma. A case report. Acta Radiol 2001; 42: 39 -42 Erdem E, Zimmerman RA, Haselgrove JC, Bilaniuk LT, Hunter JV. Diffusion-weighted imaging and fluid attenuated inversion recovery imaging in the evaluation of primitive neuroectodermal tumors. Neuroradiology 2001; 43: 927 -33 Filippi CG, Edgar MA, Ulug AM, Prowda JC, Heier LA, Zimmerman RD. Appearance of meningiomas on diffusion-weighted images: correlating diffusion constants with histopathologic findings. AJNR Am J Neuroradiol 2001; 22: 65 -72 Bitzer M, Klose U, Geist-Barth B, Alterations in diffusion and perfusion in the pathogenesis of peritumoral brain edema in meningiomas. Eur Radiol 2002; 12: 2062 -76 Jack CR Jr, Reese DF, Scheithauer BW. Radiographic findings in 32 cases of primary CNS lymphoma. AJR Am J Roentgenol 1986; 146: 271 -6 Hochberg FH, Miller DC. Primary central nervous system lymphoma. J Neurosurg 1988; 68: 835 -53 Poon T, Matoso I, Tchertkoff V, Weitzner I Jr, Gade M. CT features of primary cerebral lymphoma in AIDS and non-AIDS patients. J Comput Assist Tomogr 1989; 13: 6 -9

References (5/5) 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. Chang L, Ernst T. MR spectroscopy and diffusion-weighted MR imaging in focal brain lesions in AIDS. Neuroimaging Clin N Am 1997; 7: 409 -426 Sener RN, Dzelzite S, Migals A. Huge craniopharyngioma: diffusion MRI and contrast-enhanced FLAIR imaging. Comput Med Imaging Graph 2002; 26: 199 -203 Noguchi K, Watanabe N, Nagayoshi T, et al. Role of diffusion-weighted echo-planar MRI in distinguishing between brain abscess and tumour: a preliminary report. Neuroradiology 1999; 41: 171 -4 Park SH, Chang KH, Song IC, Kim YJ, Kim SH, Han MH. Diffusion-weighted MRI in cystic or necrotic intracranial lesions. Neuroradiology 2000; 42: 716 -21 Holtas S, Geijer B, Stromblad LG, Maly-Sundgren P, Burtscher IM. A ring-enhancing metastasis with central high signal on diffusion-weighted imaging and low apparent diffusion coefficients. Neuroradiology 2000; 42: 824 -7 Tung GA, Evangelista P, Rogg JM, Duncan JA 3 rd. Diffusion-weighted MR imaging of rim-enhancing brain masses: is markedly decreased water diffusion specific for brain abscess? AJR Am J Roentgenol 2001; 177: 709 -12 Hartmann M, Jansen O, Heiland S, Sommer C, Munkel K, Sartor K. Restricted diffusion within ring enhancement is not pathognomonic for brain abscess. AJNR Am J Neuroradiol 2001; 22: 1738 -42 Geijer B, Holtas S. Diffusion-weighted imaging of brain metastases: their potential to be misinterpreted as focal ischaemic lesions. Neuroradiology 2002; 44: 568 -73 Chang SC, Lai PH, Chen WL, et al. Diffusion-weighted MRI features of brain abscess and cystic or necrotic brain tumors: comparison with conventional MRI. Clin Imaging 2002; 26: 227 -36 Lai PH, Ho JT, Chen WL, Hsu SS, Wang JS, Pan HB, Yang CF. Brain abscess and necrotic brain tumor: discrimination with proton MR spectroscopy and diffusion-weighted imaging. AJNR Am J Neuroradiol 2002; 23: 1369 -77 Guzman R, Barth A, Lovblad KO, et al. Use of diffusion-weighted magnetic resonance imaging in differentiating purulent brain processes from cystic brain tumors. J Neurosurg 2002; 97: 1101 -7

Please refer to this book for detail.

Thank you
- Slides: 75