Diffusion Osmosis Lab Review the Day Before Water

Diffusion Osmosis Lab *Review the Day Before *
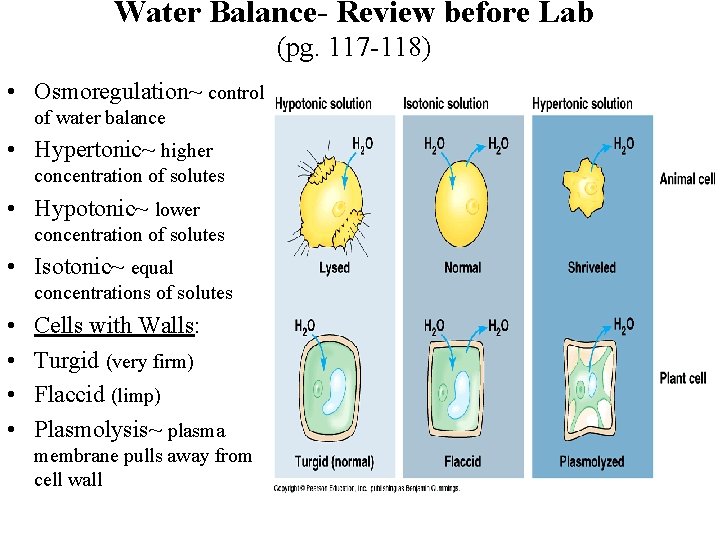
Water Balance- Review before Lab (pg. 117 -118) • Osmoregulation~ control of water balance • Hypertonic~ higher concentration of solutes • Hypotonic~ lower concentration of solutes • Isotonic~ equal concentrations of solutes • • Cells with Walls: Turgid (very firm) Flaccid (limp) Plasmolysis~ plasma membrane pulls away from cell wall

Help with Data Collection & Analysis • Mass Difference: Final – initial (absolute diff. ) • % Change in Mass: Final – initial x 100 initial **Why do we use the % change in mass instead of simply the straight difference? • Plot your data on the graph. • Determine the molar solute concentration of the potato cores. How? ? ? *Hint: Where does your line cross “ 0”

Calculating Solute Potential • Variables involved: i, C, R, T i = ionization constant: Na. Cl = 2. 0 (Na+ & Cl-) **for sucrose it will be 1. 0 (it doesn’t ionize) C = Molar concentration of your potato (determine from your graph) R= rate constant: 0. 0821 L · atm (bar) mol · K T = Temperature: K

Calculating the Solute Potential ( s) • s = - i. CRT • Sample Calc. A 1. 0 M sugar solution @ 22° C under standard atmospheric conditions: s = -(1)(1. 0 mol)(0. 0821 L · bar )(295 K) L s = -24. 22 bars mol · K
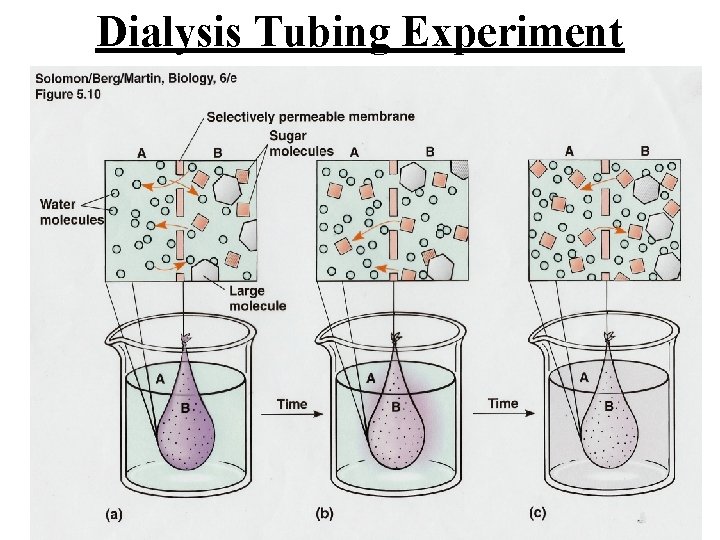
Dialysis Tubing Experiment
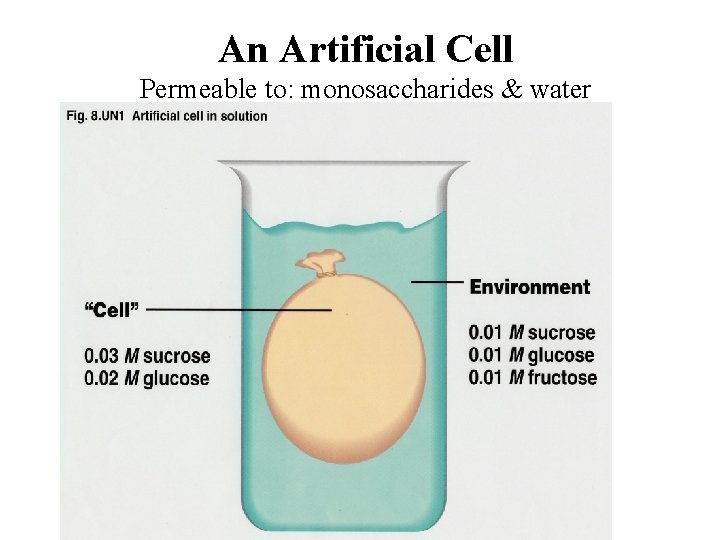
An Artificial Cell Permeable to: monosaccharides & water Impermeable to: Disaccharides

Potato Cores • Review Molarity and how to make solutions before your group begins

What to have Ready for Lab- Day 1 • Bring in a Large Potato if I tell you to (one per lab table) • Have the Pre-lab finished w/quiz • Write a statement of purpose or reason for doing this lab in your journal. Take notes in journal. • Materials list - review the handout • Hypothesis: • Data Table/ Graphs – review the lab handout the day before so you’ll know what you’re collecting & analyzing before you begin!

Key Sections for Formal Lab • Title • Intro/Background: Defining water potential Importance & Sig. Of the lab • Hypothesis & reason for your prediction • ID Experimental Variables • Materials (diagram & visual of set up optional) • Procedure • Data: tables charts & graphs • Analysis- be thorough • Conclusion & Evaluation: error & improvements

Pre- Lab Questions 1) Suppose you have an artificial cell that was permeable to monosaccharides and impermeable to disaccharides. What would happen to the cell if it had 0. 80 M maltose and 0. 85 M fructose in it and was placed in a solution containing 0. 45 M glucose, 0. 65 M fructose, and 0. 40 M sucrose. a) Which direction would the water flow? b) Which area has a higher water potential? c) What would happen to the concentration of the maltose inside the cell (increase, decrease, remain the same)? 2) What is the ionization constant (i) for sucrose?
- Slides: 11