Diffusion osmosis and the cell membrane Draw what
Diffusion, osmosis, and the cell membrane

Draw what you think a cell membrane looks like at the cellular level-
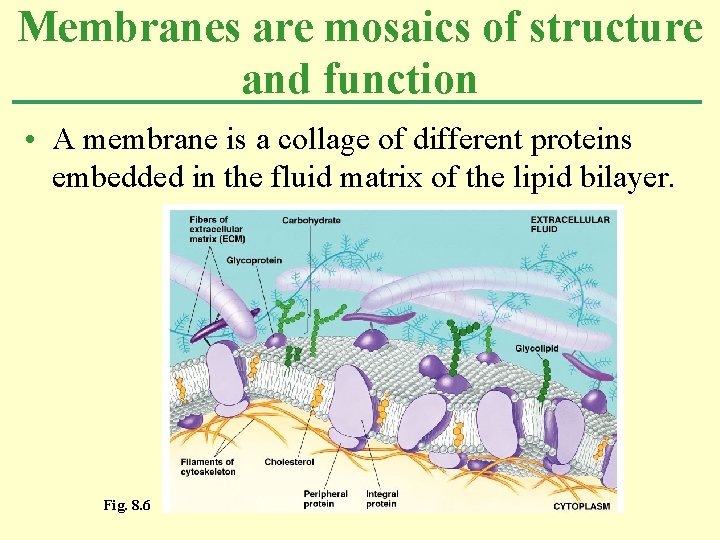
Membranes are mosaics of structure and function • A membrane is a collage of different proteins embedded in the fluid matrix of the lipid bilayer. Fig. 8. 6

What do you think membranes do?
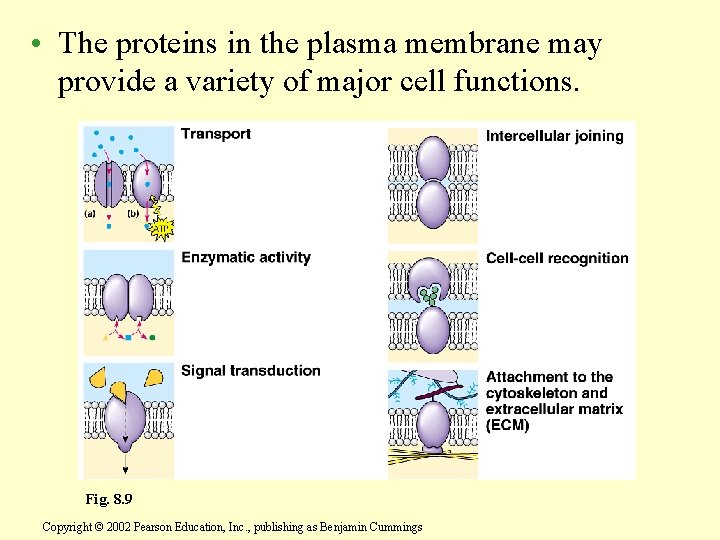
• The proteins in the plasma membrane may provide a variety of major cell functions. Fig. 8. 9 Copyright © 2002 Pearson Education, Inc. , publishing as Benjamin Cummings

What is diffusion? • Diffusion is a process where molecules move from greater molecule concentrations to areas of less molecule concentrations until an equal distribution of those molecules is reached.



Passive transport is diffusion across a membrane • Movements of individual molecules are random. • However, movement of a population of molecules may be directional. Copyright © 2002 Pearson Education, Inc. , publishing as Benjamin Cummings

What is the difference between permeable, semipermeable and impermeable membranes?
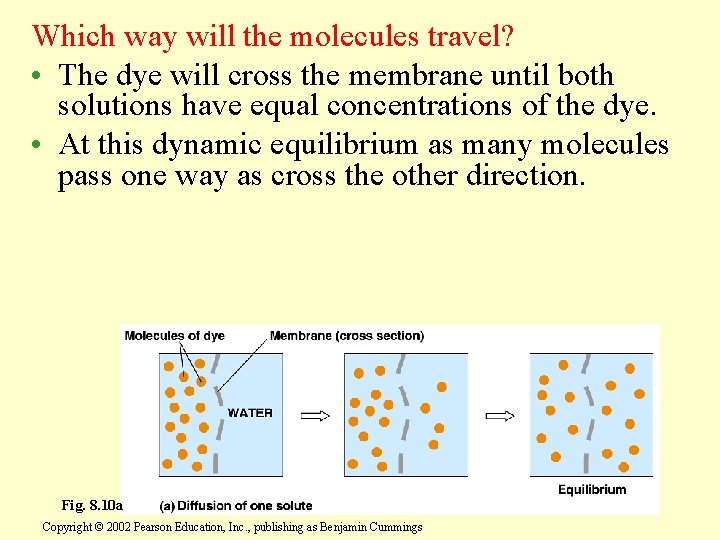
Which way will the molecules travel? • The dye will cross the membrane until both solutions have equal concentrations of the dye. • At this dynamic equilibrium as many molecules pass one way as cross the other direction. Fig. 8. 10 a Copyright © 2002 Pearson Education, Inc. , publishing as Benjamin Cummings

• In the absence of other forces, a substance will diffuse from where it is more concentrated to where it is less concentrated, down its concentration gradient. – This spontaneous process decreases free energy and increases entropy by creating a randomized mixture. • Each substance diffuses down its own concentration gradient, independent of the concentration gradients of other substances. Fig. 8. 10 b Copyright © 2002 Pearson Education, Inc. , publishing as Benjamin Cummings

• The diffusion of a substance across a biological membrane is passive transport because it requires no energy from the cell to make it happen. – The concentration gradient represents potential energy and drives diffusion. • However, because membranes are selectively permeable, the interactions of the molecules with the membrane play a role in the diffusion rate. • Diffusion of molecules with limited permeability through the lipid bilayer may be assisted by transport proteins.
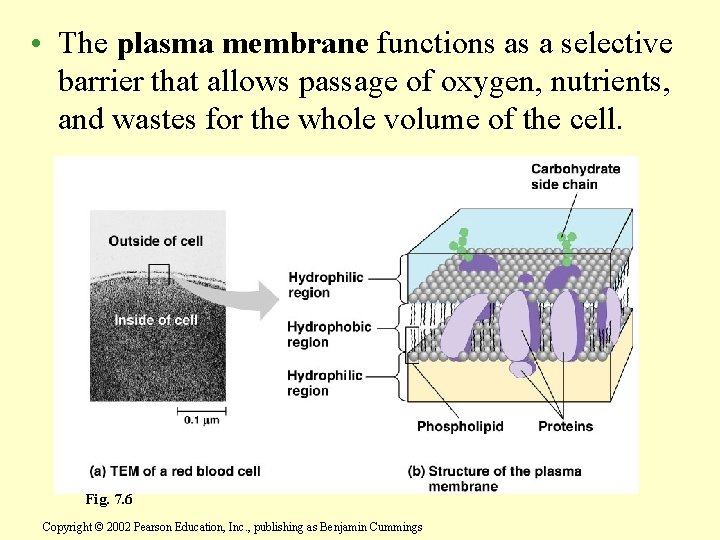
• The plasma membrane functions as a selective barrier that allows passage of oxygen, nutrients, and wastes for the whole volume of the cell. Fig. 7. 6 Copyright © 2002 Pearson Education, Inc. , publishing as Benjamin Cummings

Osmosis is defined as the diffusion of water across a selectively permeable membrane.

Practice problem- please write on notes


• Differences in the relative concentration of dissolved materials in two solutions can lead to the movement of ions from one to the other. – The solution with the higher concentration of solutes is hypertonic. – The solution with the lower concentration of solutes is hypotonic. – These are comparative terms. • Tap water is hypertonic compared to distilled water but hypotonic when compared to sea water. – Solutions with equal solute concentrations are isotonic.

Say what? • Let’s refer to the graph that you made for the potato cores. • What is causing the cores to lose their mass? Water is being lost from the potato core cells. • What is causing them to gain in mass? • Water is being gained by the potato cells. • What is the only variable that is being manipulated? • The solute concentration.

• Imagine that two sugar solutions differing in concentration are separated by a membrane that will allow water through, but not sugar. • The hypertonic solution has a lower water concentration than the hypotonic solution. – More of the water molecules in the hypertonic solution are bound up in hydration shells around the sugar molecules, leaving fewer unbound water molecules. • Draw a diagram that represents this scenario-
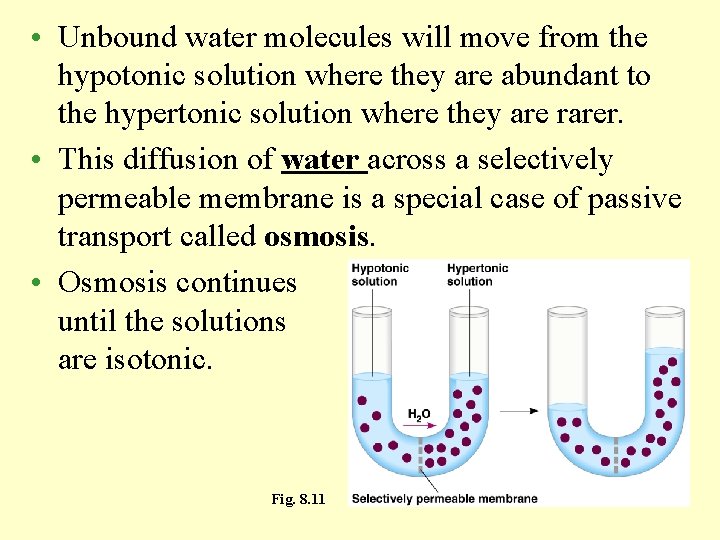
• Unbound water molecules will move from the hypotonic solution where they are abundant to the hypertonic solution where they are rarer. • This diffusion of water across a selectively permeable membrane is a special case of passive transport called osmosis. • Osmosis continues until the solutions are isotonic. Fig. 8. 11
Back to your graph • What is the solute concentration surrounding the potato cores relative to the solute concentration inside the potato cells? • they are the same • When the average percent change in mass of the potato cores increases how does the solute concentration compare between the outside solution and inside solution? • The solute concentration in the cells is greater than solute concentrrtion in the surrounding solution

• When the average percent change in mass of the potato cores decreases how does the solute concentration compare between the outside solution and inside solution? • The solute concentration in the cells is less than the solute concentration in the surrounding solution


Cell survival depends on balancing water uptake and loss • An animal cell immersed in an isotonic environment experiences no net movement of water across its plasma membrane. – Water flows across the membrane, but at the same rate in both directions. – The volume of the cell is stable.
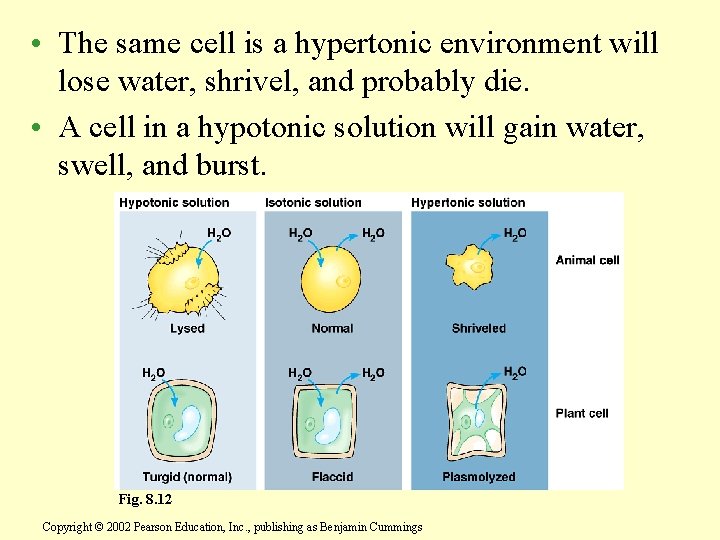
• The same cell is a hypertonic environment will lose water, shrivel, and probably die. • A cell in a hypotonic solution will gain water, swell, and burst. Fig. 8. 12 Copyright © 2002 Pearson Education, Inc. , publishing as Benjamin Cummings
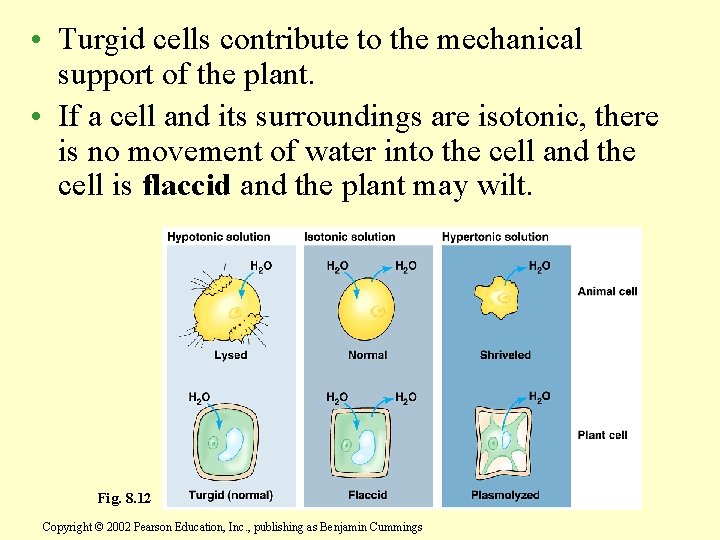
• Turgid cells contribute to the mechanical support of the plant. • If a cell and its surroundings are isotonic, there is no movement of water into the cell and the cell is flaccid and the plant may wilt. Fig. 8. 12 Copyright © 2002 Pearson Education, Inc. , publishing as Benjamin Cummings

• You. Tube - Statkraft - Osmotic Power Plant
- Slides: 28