Diffusion Mass transfer in the absence of fluid

Diffusion Mass transfer in the absence of fluid motion http: //theonlinephotographer. typepad. com/the_online_photographer/2011/07/fixer-doesnt-sink. html Clemson Hydro
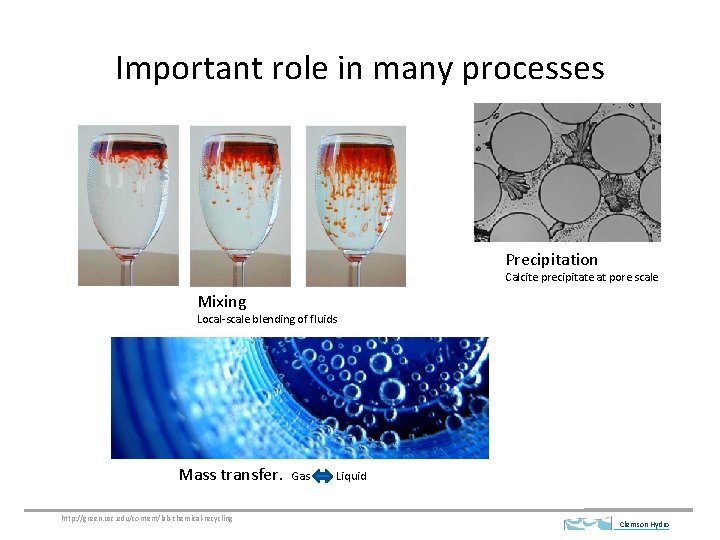
Important role in many processes Precipitation Calcite precipitate at pore scale Mixing Local-scale blending of fluids Mass transfer. http: //green. usc. edu/content/lab-chemical-recycling Gas Liquid Clemson Hydro
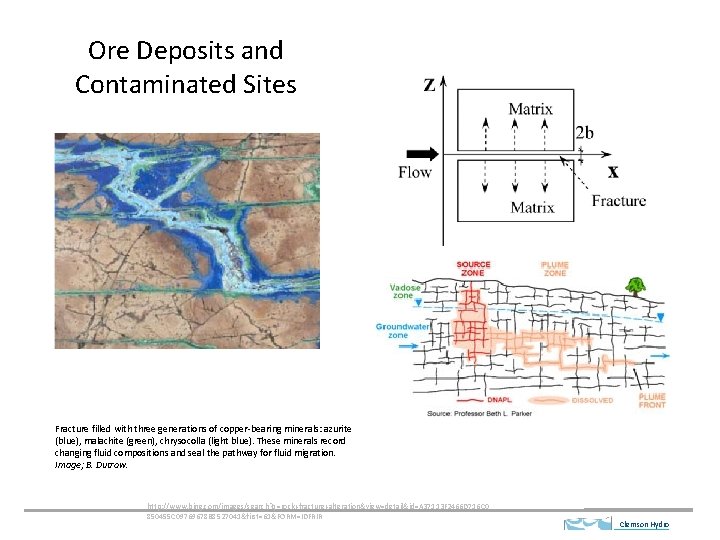
Ore Deposits and Contaminated Sites Fracture filled with three generations of copper-bearing minerals: azurite (blue), malachite (green), chrysocolla (light blue). These minerals record changing fluid compositions and seal the pathway for fluid migration. Image; B. Dutrow. http: //www. bing. com/images/search? q=rock+fracture+alteration&view=detail&id=A 37113 F 2466 D 716 C 0 850455 C 09769678 B 8527041&first=61&FORM=IDFRIR Clemson Hydro

Diffusion Sampler Clemson Hydro
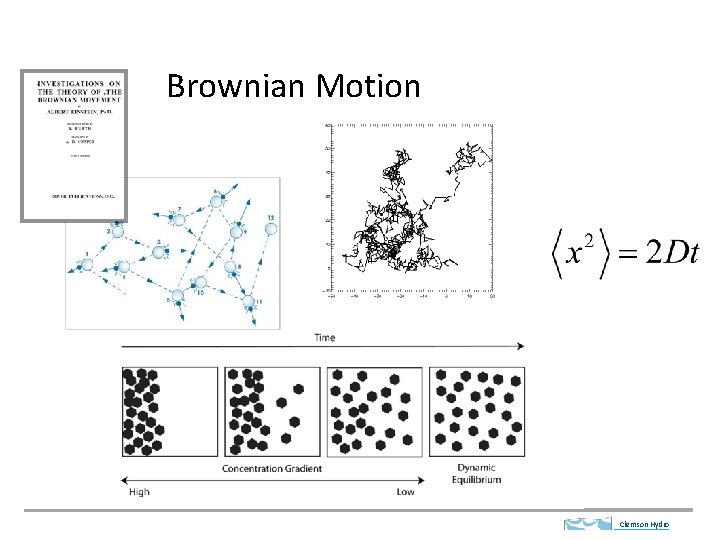
Brownian Motion Clemson Hydro

Chemical potential Ideal solutions Clemson Hydro

Concept for Mass Flux • Flux from high to low chemical potential • Flux magnitude proportional to gradient or potential difference Clemson Hydro

Diffusion through liquid Conceptual v Stokes law: Assume (Einstein, 1905): Equate and rearrange: From previous page: Mass flux: Cussler [1997, p. 116] Clemson Hydro
![Mass Flux Fick’s Law C x D: Diffusivity[L 2/T] Mass Transfer Law k: mass Mass Flux Fick’s Law C x D: Diffusivity[L 2/T] Mass Transfer Law k: mass](http://slidetodoc.com/presentation_image_h2/4754f2b76c2992d92197093cfcad3dd6/image-9.jpg)
Mass Flux Fick’s Law C x D: Diffusivity[L 2/T] Mass Transfer Law k: mass transfer coefficient [L/T] k~D/L C 1: reference concentration [M/L 3] Clemson Hydro

Conservation of Mass Diffusion Equation c=C Storage No Advective Flux Diffusive Flux (Fick’s Law) Source S=0 (for now) Governing Clemson Hydro

Simulation of Diffusion Governing Boundary Conditions Dirichlet, Specify Conc Neumann, Specify flux Cauchy, flux proportional to gradient Clemson Hydro
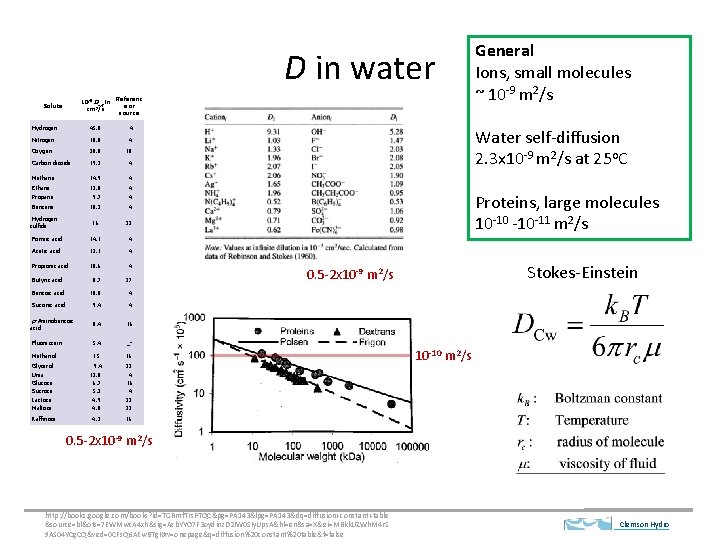
D in water 10 -6 Daq in cm 2/s Solute Referenc e or source Hydrogen 45. 0 4 Nitrogen 18. 8 4 Oxygen 20. 0 10 Carbon dioxide 19. 2 4 Methane 14. 9 4 Ethane Propane 12. 0 9. 7 4 4 Benzene 10. 2 4 Hydrogen sulfide 16 22 Formic acid 14. 1 4 Acetic acid 12. 1 4 Propionic acid 10. 6 4 8. 7 27 Benzoic acid 10. 0 4 Succinic acid 9. 4 4 8. 4 16 Fluorescein 5. 4 _a Methanol 15 16 Glycerol Urea Glucose Sucrose Lactose Maltose 9. 4 13. 8 6. 7 5. 2 4. 9 4. 8 22 4 16 4 22 22 Raffinose 4. 3 16 Butyric acid p-Aminobenzoic acid General Ions, small molecules ~ 10 -9 m 2/s Water self-diffusion 2. 3 x 10 -9 m 2/s at 25 o. C Proteins, large molecules 10 -10 -10 -11 m 2/s Stokes-Einstein 0. 5 -2 x 10 -9 m 2/s 10 -10 m 2/s 0. 5 -2 x 10 -9 m 2/s http: //books. google. com/books? id=TGRmf. Trs. PTQC&pg=PA 143&lpg=PA 143&dq=diffusion+constant+table &source=bl&ots=7 EWMwc. A 4 xh&sig=Azb. YYO 7 F 3 oydinz. D 2 IW 0 SIy. Ups. A&hl=en&sa=X&ei=MBkk. UZWh. M 4 r. S 9 AS 04 YCg. CQ&ved=0 CFs. Q 6 AEw. BTg. K#v=onepage&q=diffusion%20 constant%20 table&f=false Clemson Hydro

What is affected by diffusion? Stokes-Einstein Lithium in Water rc: 0. 167 x 10 -9 m* 0. 076 x 10 -9 m, T: 293 K m: 0. 001 Pa s=0. 001 kg/m s Sizes of small things *http: //www. periodictable. com/Properties/A/Atomic. Radius. v. wt. html http: //en. wikipedia. org/wiki/Bacteria#mediaviewer/File: Relative_scale. svg Check with example Prokaryote in Water rc: 1000 x 10 -9 m T: 293 K m: 0. 001 Pa s=0. 001 kg/m s Clemson Hydro
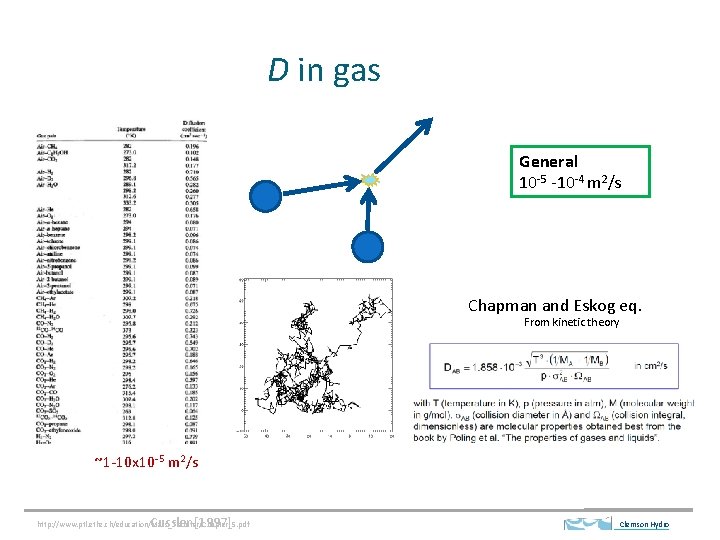
D in gas General 10 -5 -10 -4 m 2/s Chapman and Eskog eq. From kinetic theory ~1 -10 x 10 -5 m 2/s Cussler [1997] http: //www. ptl. ethz. ch/education/Mass_Transfer/Chapter_5. pdf Clemson Hydro
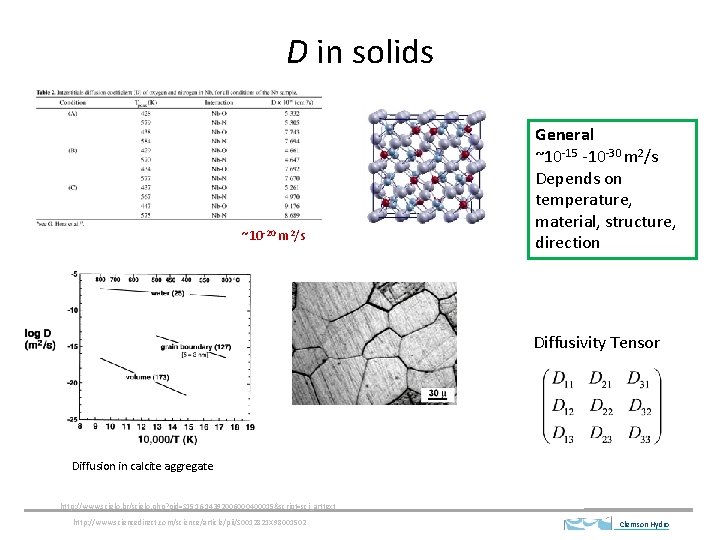
D in solids ~10 -20 m 2/s General ~10 -15 -10 -30 m 2/s Depends on temperature, material, structure, direction Diffusivity Tensor Diffusion in calcite aggregate http: //www. scielo. br/scielo. php? pid=S 1516 -14392006000400015&script=sci_arttext http: //www. sciencedirect. com/science/article/pii/S 0012821 X 98001502 Clemson Hydro

D in Porous Media Saturated Conditions Partially Saturated Millington Quirk Tortuosity Buckingham Penman http: //www. tue. nl/onderzoek/instituten-groepen-scholen/top-research-groups/eindhoven-multiscaleinstitute/events/past-events/14 -01 -2013 -porous-media/ [Logan, 2012] Clemson Hydro
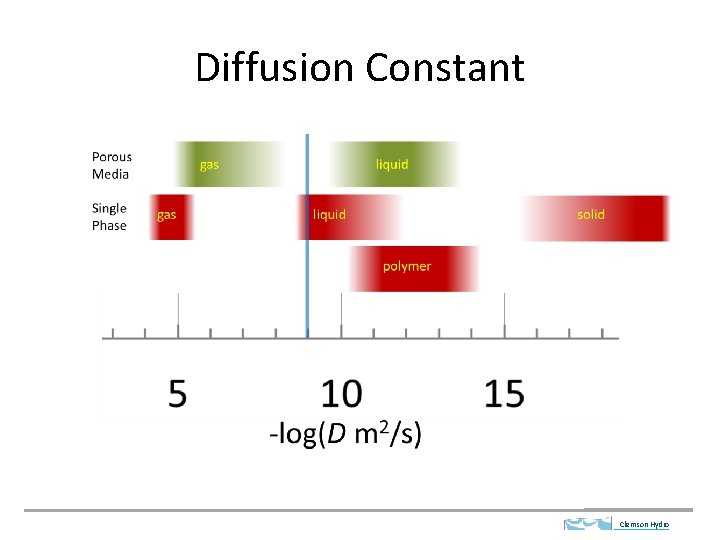
Diffusion Constant Clemson Hydro
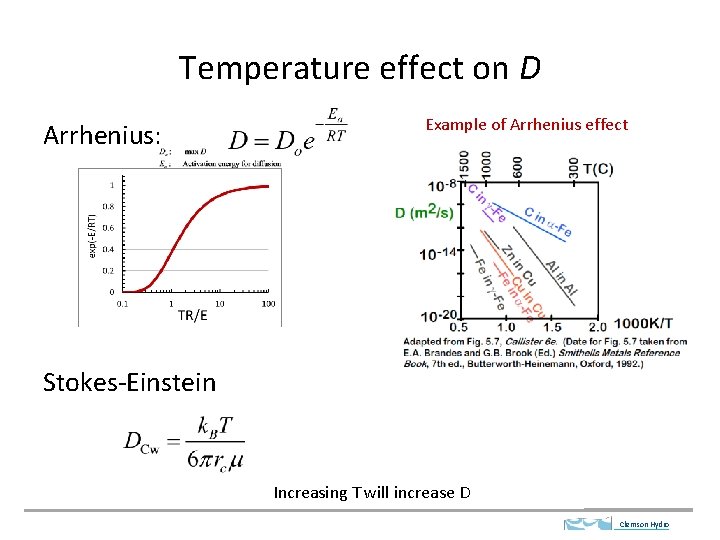
Temperature effect on D Arrhenius: Example of Arrhenius effect Stokes-Einstein Increasing T will increase D Clemson Hydro

Scaling • Sherwood number mass transfer vel. / diffusion velocity • Stanton Number mass transfer vel. /flow velocity • Schmidt Number diffusion momentum/diffusion of mass • Peclet Number flow velocity/diffusion velocity Clemson Hydro
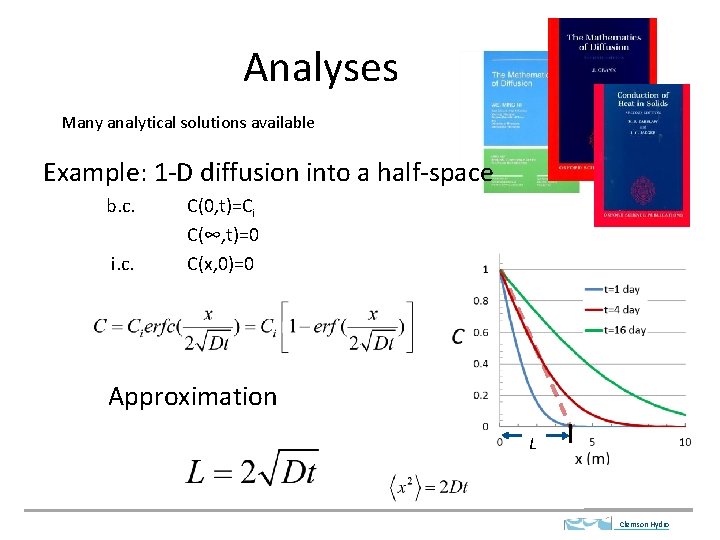
Analyses Many analytical solutions available Example: 1 -D diffusion into a half-space b. c. i. c. C(0, t)=Ci C(∞, t)=0 C(x, 0)=0 Approximation L Clemson Hydro
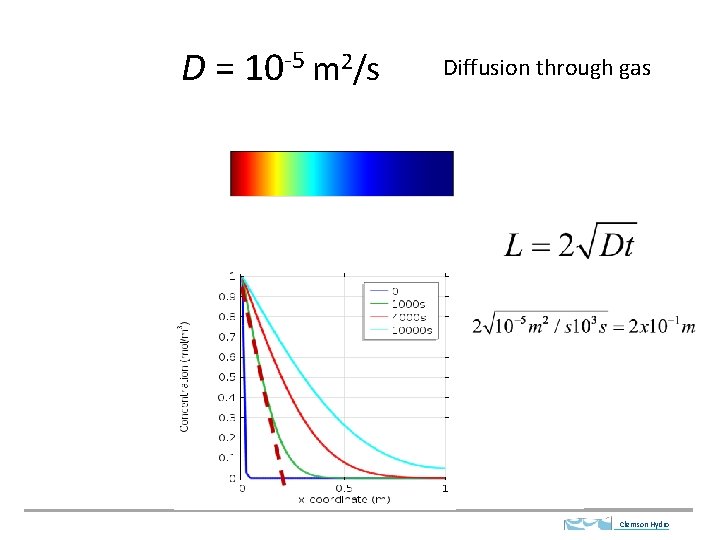
D = 10 -5 m 2/s Diffusion through gas Clemson Hydro
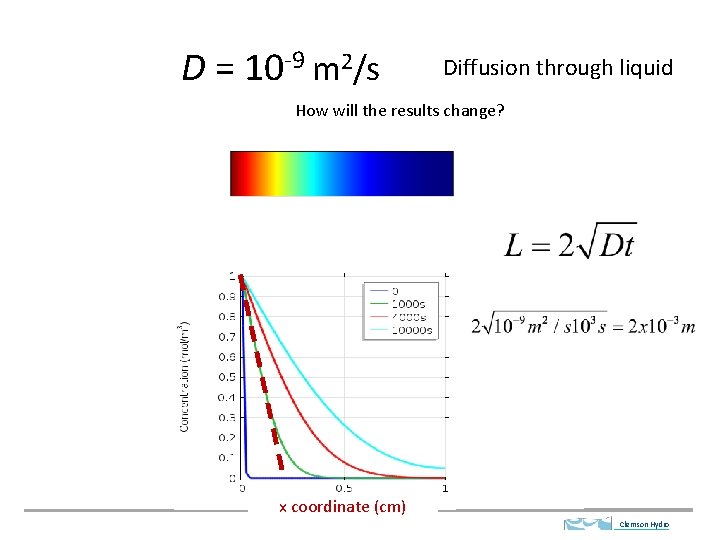
D = 10 -9 m 2/s Diffusion through liquid How will the results change? x coordinate (cm) Clemson Hydro
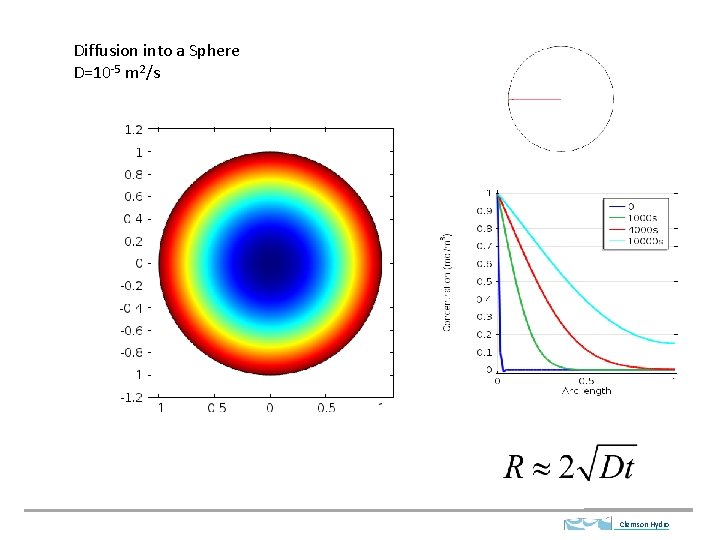
Diffusion into a Sphere D=10 -5 m 2/s Clemson Hydro
Effect of boundary Clemson Hydro
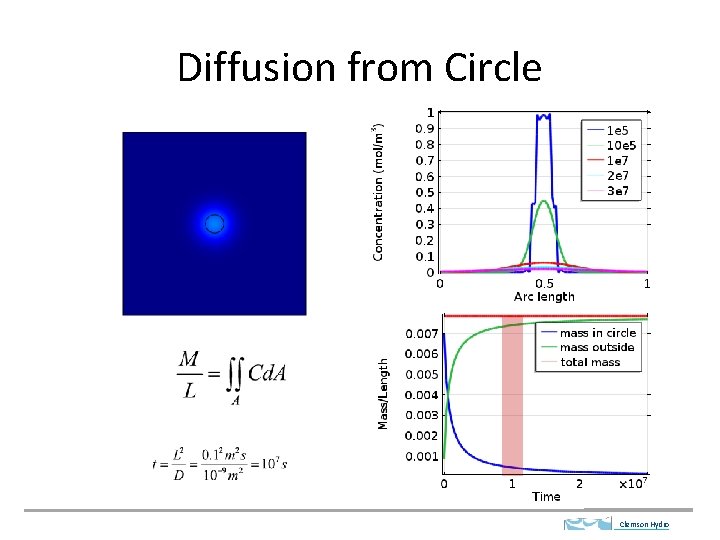
Diffusion from Circle Clemson Hydro
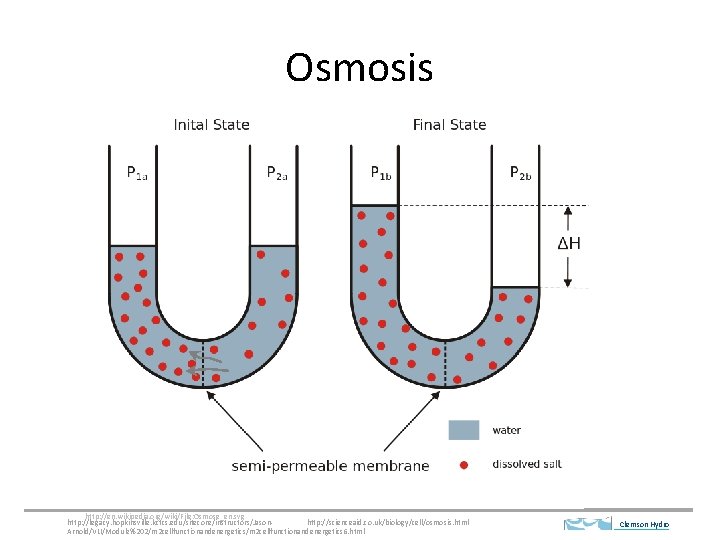
Osmosis http: //en. wikipedia. org/wiki/File: Osmose_en. svg http: //legacy. hopkinsville. kctcs. edu/sitecore/instructors/Jasonhttp: //scienceaid. co. uk/biology/cell/osmosis. html Arnold/VLI/Module%202/m 2 cellfunctionandenergetics 6. html Clemson Hydro
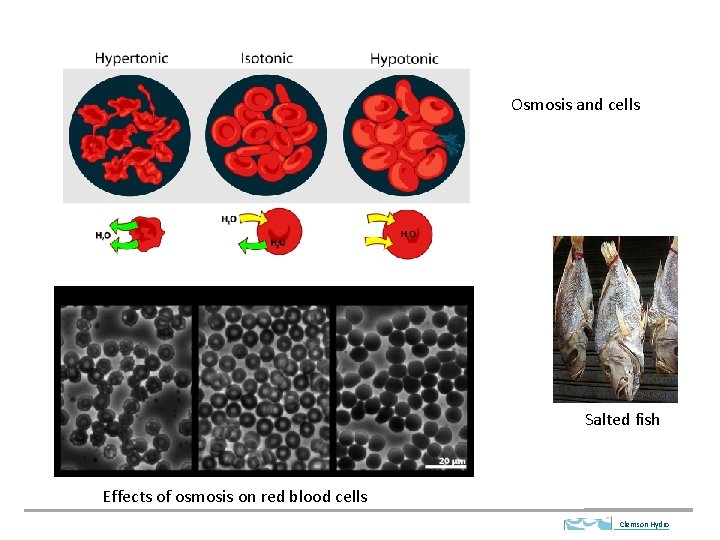
Osmosis and cells Salted fish Effects of osmosis on red blood cells Clemson Hydro

Osmosis and plants Amaryllis blooming sequence Clemson Hydro
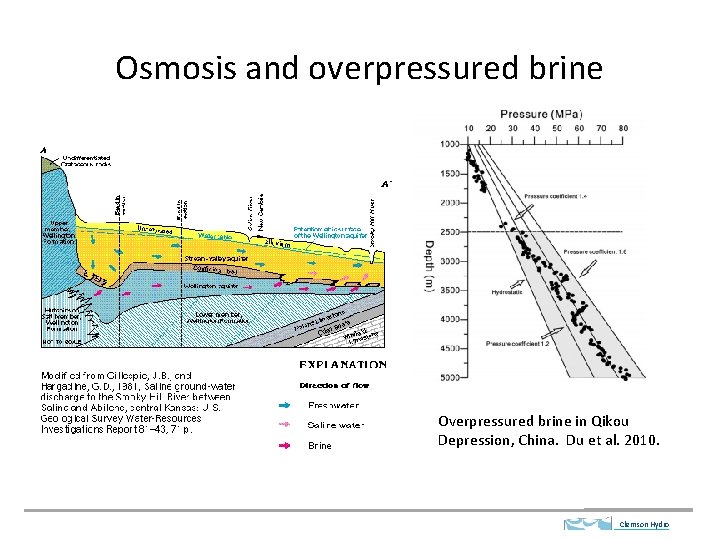
Osmosis and overpressured brine Overpressured brine in Qikou Depression, China. Du et al. 2010. Clemson Hydro
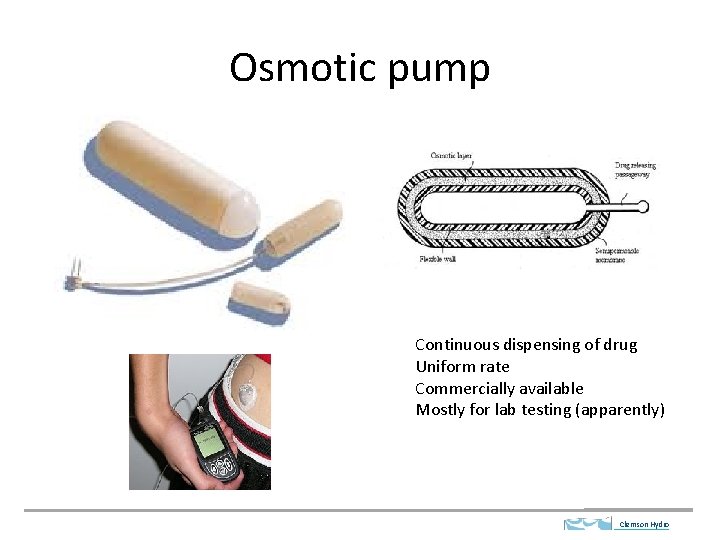
Osmotic pump Continuous dispensing of drug Uniform rate Commercially available Mostly for lab testing (apparently) Clemson Hydro

Osmotic power plant, Norway Experimental right now Commercial plant in 2015 http: //www. nordicenergysolutions. org/innovation/demonstration-pilot/ocean-energy/osmotic-powerpilot/? searchterm=osmotic Clemson Hydro

Clemson Hydro
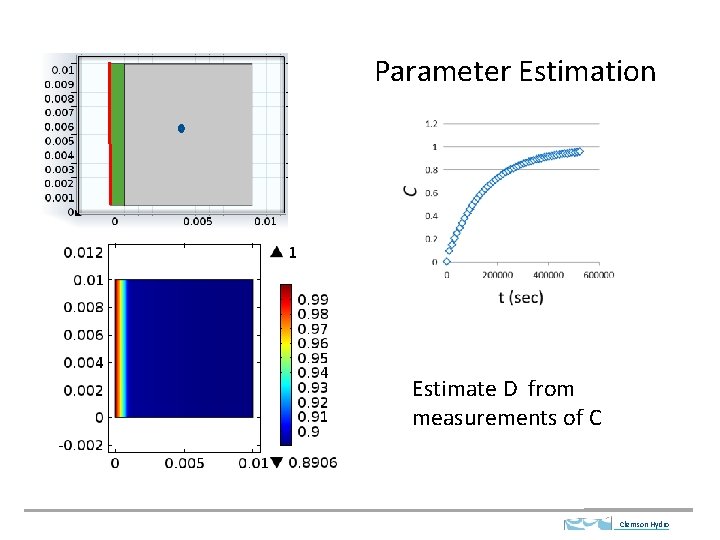
Parameter Estimation Estimate D from measurements of C Clemson Hydro
- Slides: 33