Diffusion continued Lecture 17 Ficks First Law Last















- Slides: 20

Diffusion (continued) Lecture 17

Fick’s First Law Last time we derived Fick’s First Law

Continuity Equation • Imagine a volume and a flux of a species into that volume, Jx, at X and a flux out, Jx+dx, at x+dx. • Suppose nx atom/sec at x and nx+dx atoms/sec pass through the plane at x+dx. • The fluxes at the two planes are Jx = nx/dx 2 -sec and Jx+dx = nx+dx/dx 2 -sec. • Conservation of mass dictates that the increase in the number of atoms, dn, within the volume is just what goes in minus what comes out. • Over an increment of time dt this will be dn = (Jx - Jx+dx)dt. The concentration change is or: At fixed t and x

Deriving Fick’s Second Law • Continuity equation relates any change in flux along the gradient to change in concentration with time. • Since: • we have:

Fick’s Second Law • The second law says that the change in concentration with time at some point on a conc. gradient depends on the second derivative of the concentration gradient. • The straight line (∂2 c/∂x 2) is the steady state case. It is also the situation towards which a system will evolve over time.

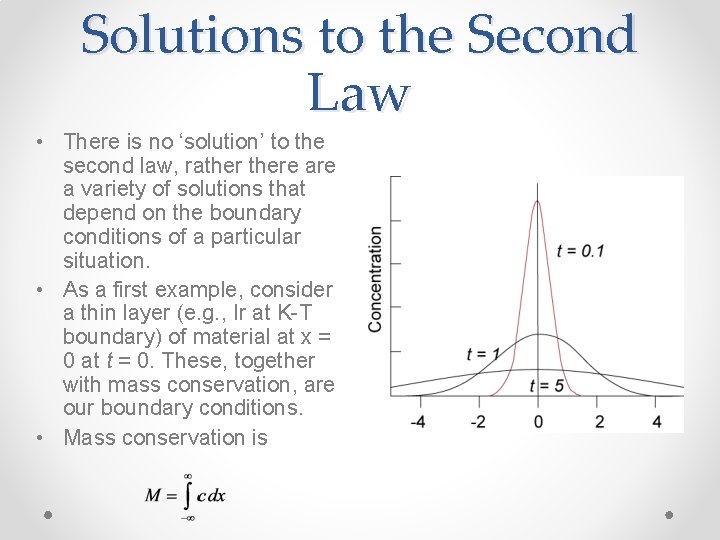
Solutions to the Second Law • There is no ‘solution’ to the second law, rathere a variety of solutions that depend on the boundary conditions of a particular situation. • As a first example, consider a thin layer (e. g. , Ir at K-T boundary) of material at x = 0 at t = 0. These, together with mass conservation, are our boundary conditions. • Mass conservation is

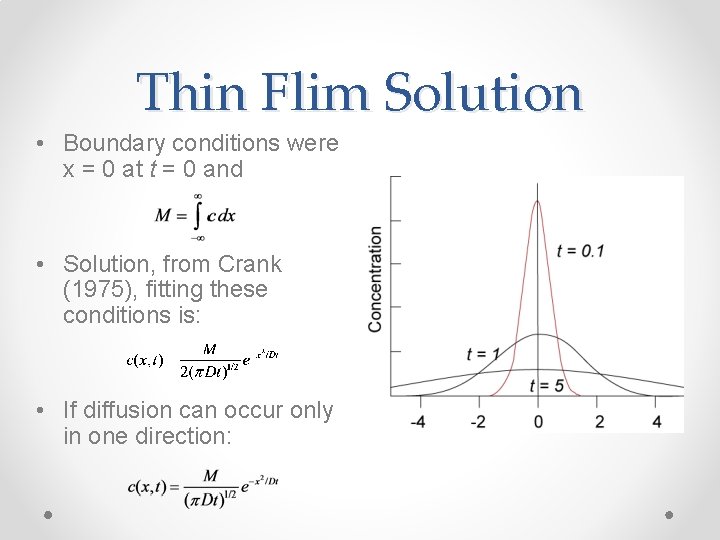
Thin Flim Solution • Boundary conditions were x = 0 at t = 0 and • Solution, from Crank (1975), fitting these conditions is: • If diffusion can occur only in one direction:

Adjacent Layers • Now suppose with have one layer with concentration co abutting a second with c=0. • Crank’s solution is to superimpose the solutions for an infinite number of thin flims, dξ, and to sum their contributions to the total concentration at Xp. • Solution is:

Adjacent Layers • The solution to: • is • where erf is the error function and has the approximate form: • also available as the ERF function in Excel.

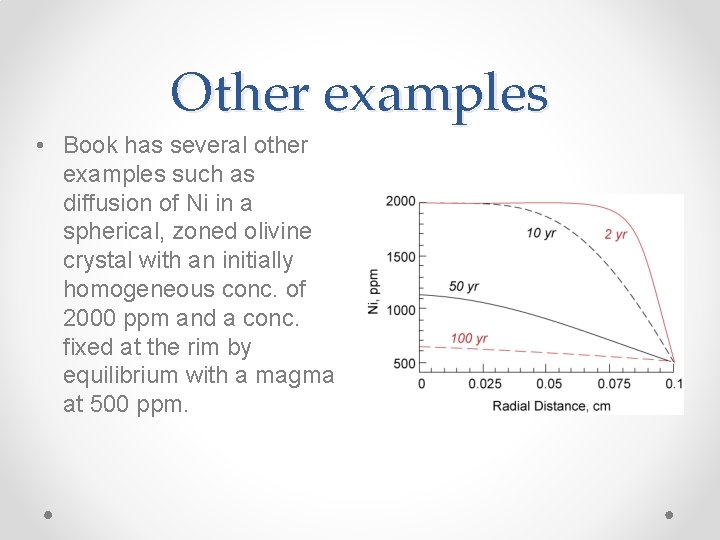
Other examples • Book has several other examples such as diffusion of Ni in a spherical, zoned olivine crystal with an initially homogeneous conc. of 2000 ppm and a conc. fixed at the rim by equilibrium with a magma at 500 ppm.

Diffusion in Multicomponents Systems • So far, we have considered only what is called tracer or self diffusion - diffusion of a species who concentration is so small it has not affect on the system. • By definition, diffusion is a process in which there is no net transport across the boundary of interest (otherwise it is advection). Mass & charge balance require diffusion in one direction to be balanced by diffusion in another. • More generally, we can consider o Chemical diffusion o Multicomponent chemical diffusion

Chemical Diffusion • Chemical diffusion refers to non-ideal situations where for one reason or another (e. g. , medium is inhomogeneous, or diffusing species affects behavior of medium) we replace concentration with the chemical potential gradient. • where L is the chemical or phenomenological coefficient

Multicomponent Diffusion • This is where the concentration of an species is great enough that its diffusion depends on the diffusion of another species, e. g. , Fe and Mg in ferromagnesian silicates (these elements tend to occupy the same site, so Mg can move in only if Fe moves out, etc). • Di, k is called the interdiffusion coefficient and computed as: o where n’s are the mole fractions. • The total solution is J = -DC (minus sign missing in book) • where J and C are (vertical) vectors of species and D is a matrix.

Multicomponent Chemical • Multicomponent chemical diffusion is a combination of the latter two, where we use interdiffusion coefficients and chemical potential. • Diffusion matrix is: J = -LC • Watson’s experiments dissolving quartz in basalt show an example where chemical diffusion must be used - note Na and K diffusing up a concentration gradient!

Mechanisms of Diffusion in Solids • Exchange: low probability • Interstitial: pretty much limited to He, etc. • Interstitiality: also low probability • Vacancy: most likely • Diffusion will then depend on both the probability of a jump and the probability of a vacancy. o Vacancies will increase with T: o where k is a rate constant and EH and activation energy for vacancy creation.

Diffusion Rates • Probability of atom making a jump to vacancy is • where ℵ is number of attempts and EB is the activation or barrier energy. • Combining, total diffusion rate will be product of probability. of a vacancy times probability of a jump: o where m and n are constants • At higher T, thermal vacancies dominate and • Bottom line: like other things in kinetics, we expect diffusion rates to increase exponentially with temperature.

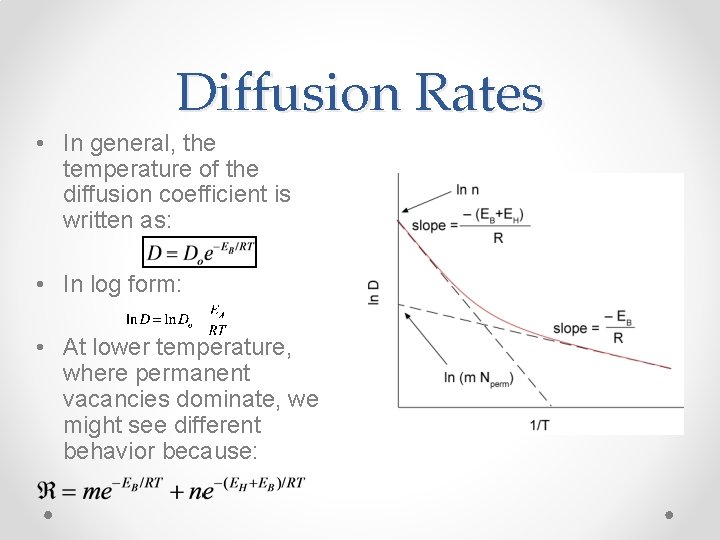
Diffusion Rates • In general, the temperature of the diffusion coefficient is written as: • In log form: • At lower temperature, where permanent vacancies dominate, we might see different behavior because:

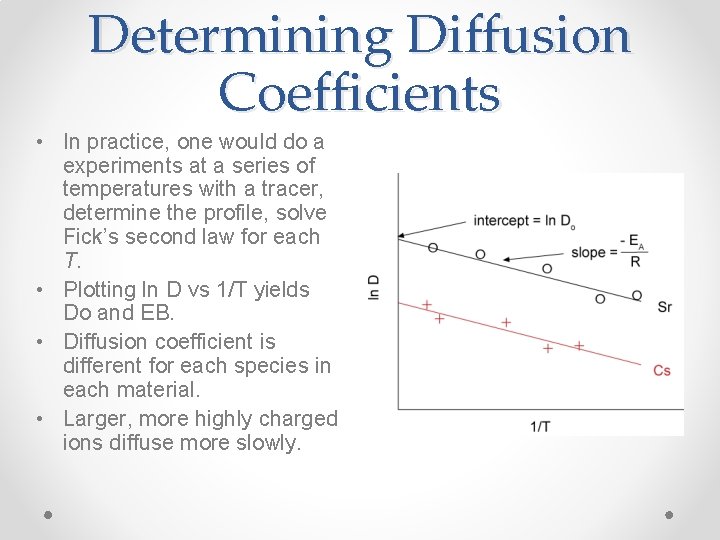
Determining Diffusion Coefficients • In practice, one would do a experiments at a series of temperatures with a tracer, determine the profile, solve Fick’s second law for each T. • Plotting ln D vs 1/T yields Do and EB. • Diffusion coefficient is different for each species in each material. • Larger, more highly charged ions diffuse more slowly.

Reactions at Surfaces Homogeneous reactions are those occurring within a single phase (e. g. , an aqueous solution). Heterogeneous reactions are those occurring between phases, e. g. , two solids or a solid and a liquid. Heterogeneous reactions necessarily occur across an interface.

Interfaces, Surfaces, and partial molar area • By definition, an interface is boundary between two condensed phases (solids and liquids). • A surface is the boundary between a condensed phase and a gas (or vacuum). o In practice, surface is often used in place of interface. • We previously defined partial molar parameters as the change in the parameter for an infinitesimal addition of a component, e. g. , vi = (∂V/∂n)T, P, nj. We define the partial molar area of phase ϕ as: • where n is moles of substance. • Unlike other molar quantities, partial molar area is not an intrinsic property of the phase, but depends on shape, size, roughness, etc. For a perfect sphere: