Diffusion and Osmosis and the Cell Membrane Why

Diffusion and Osmosis and the Cell Membrane

• Why can you smell baked bread before you see it? • Spray demo

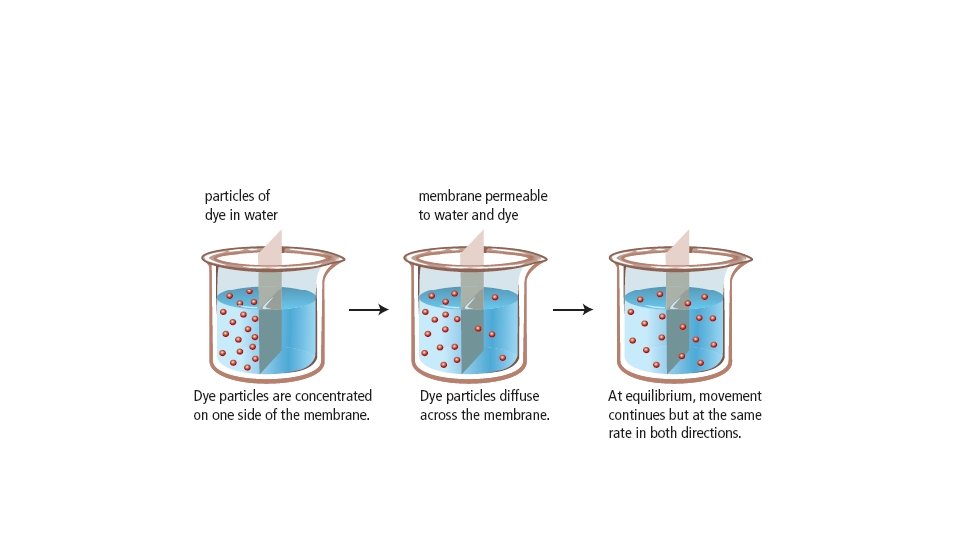
What is Diffusion? • Diffusion is the movement of particles from an area of higher concentration to an area of lower concentration. See pages 40 - 41 (c) Mc. Graw Hill Ryerson 2007

What is Concentration? • Concentration is the amount of particles in a given space. • The smell of fresh baked bread “spreading” throughout the room is an example of diffusion The diffusion of ink in water. See pages 40 - 41 (c) Mc. Graw Hill Ryerson 2007

Does diffusion work in solutions? • Demo

What factors would affect the rate of diffusion • Demo
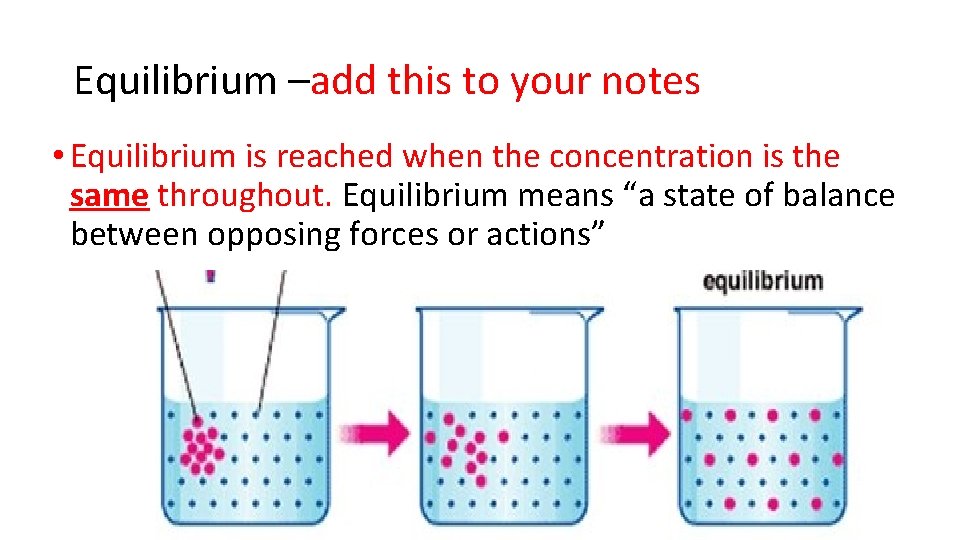
Equilibrium –add this to your notes • Equilibrium is reached when the concentration is the same throughout. Equilibrium means “a state of balance between opposing forces or actions” See page 42 (c) Mc. Graw Hill Ryerson 2007
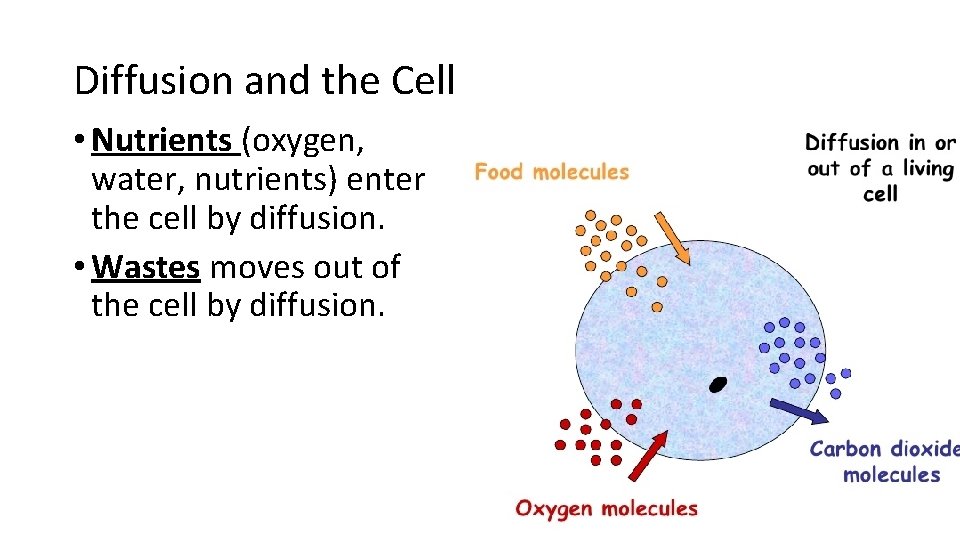
Diffusion and the Cell • Nutrients (oxygen, water, nutrients) enter the cell by diffusion. • Wastes moves out of the cell by diffusion.

Where does Diffusion occur in the Body? • Eg. Diffusion of oxygen and carbon dioxide occurs in the lungs. • Eg. Diffusion of calcium from food into cells occurs in the intestines. • Eg. Diffusion of water, salts, and waste products occurs in the kidneys.
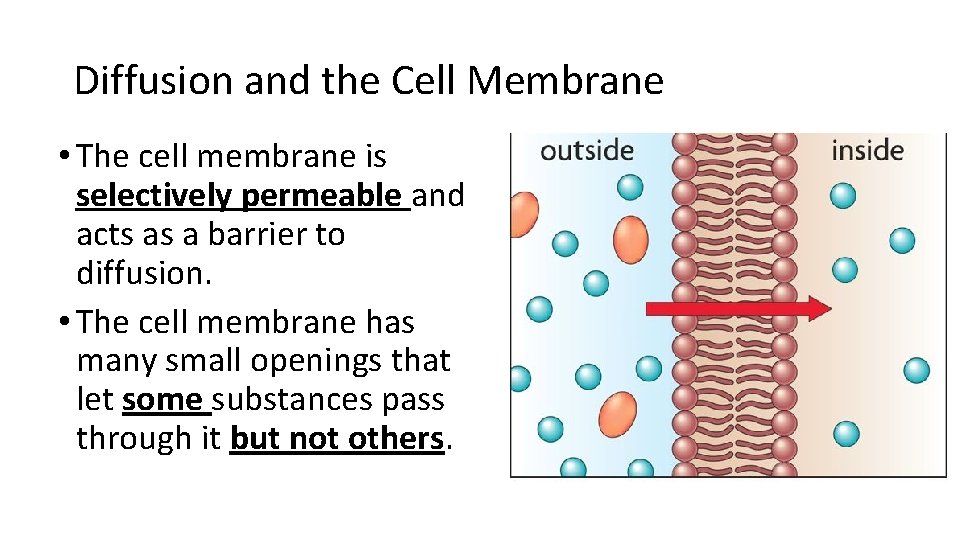
Diffusion and the Cell Membrane • The cell membrane is selectively permeable and acts as a barrier to diffusion. • The cell membrane has many small openings that let some substances pass through it but not others.

Osmosis • Osmosis is a special kind of diffusion that involves movement of only water through a selectively permeable membrane. • Osmosis is the Diffusion of Water: Water moves from area of high low concentration •

Osmosis and the Cell • Cells contain and need water to survive. • Osmosis is how the cell gains and loses its needed water. • Osmosis occurs in ALL cells. Explain how placing this wilted flower in water will cause the flower to “straighten up”. See pages 43 - 44 (c) Mc. Graw Hill Ryerson 2007

For Osmosis to Occur: There must be: 1. Two solutions of different concentrations 2. A selectively permeable membrane

Osmosis • What would happen when concentration of water outside a cell is higher than inside?
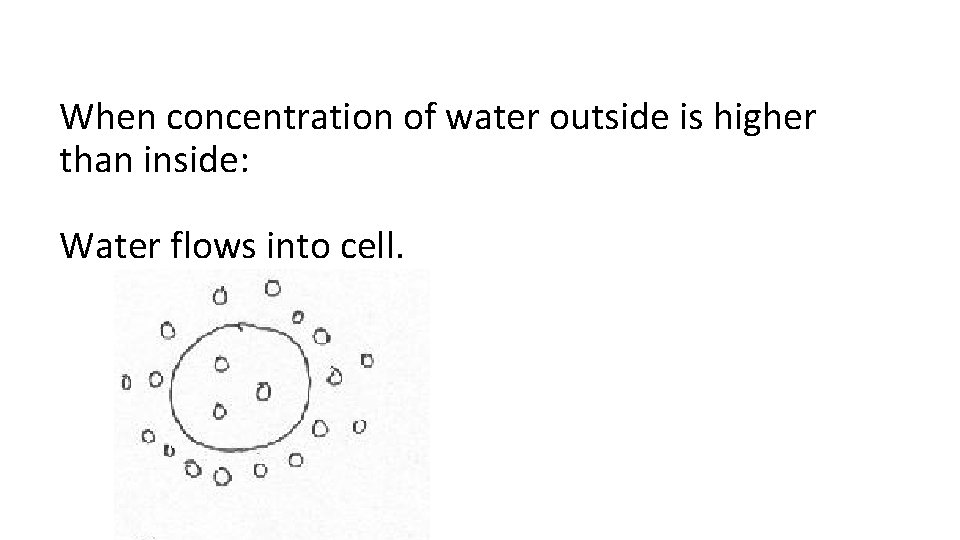
When concentration of water outside is higher than inside: Water flows into cell.

When concentration of water is higher inside: Water flows out of cell. Cell shrinks.
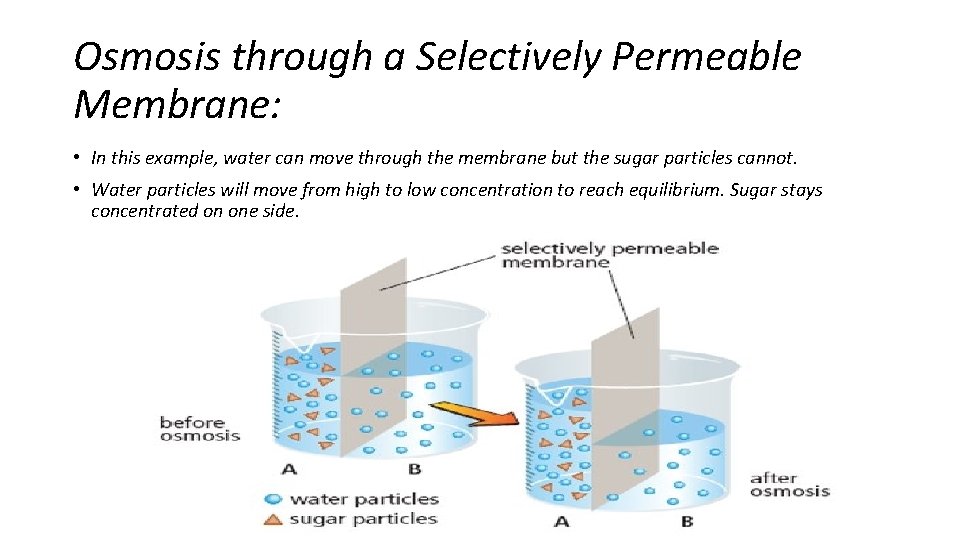
Osmosis through a Selectively Permeable Membrane: • In this example, water can move through the membrane but the sugar particles cannot. • Water particles will move from high to low concentration to reach equilibrium. Sugar stays concentrated on one side.

Why does this happen? •

Why do we get Wrinkly Fingers? • Water is absorbed through skin

Homework • Diffusion Osmosis WB • Finish Microscope labs
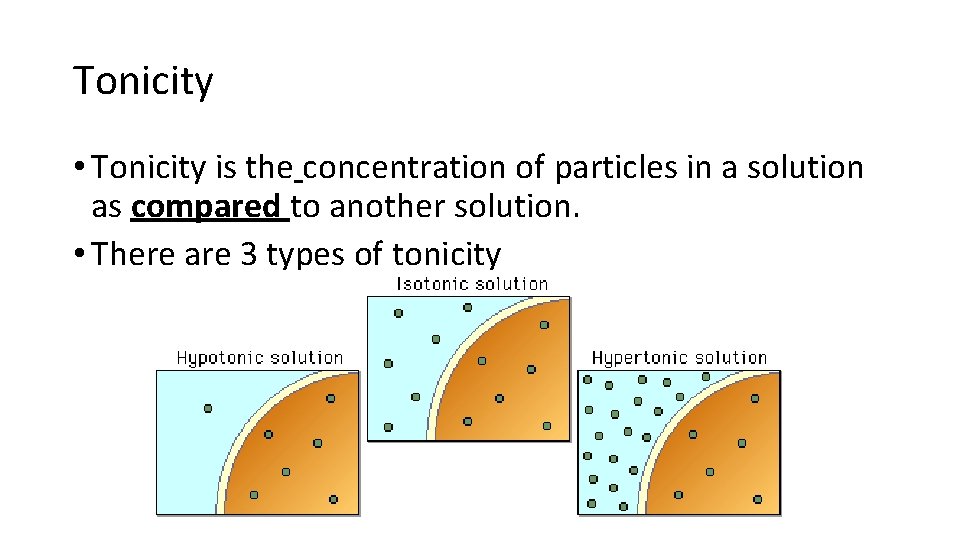
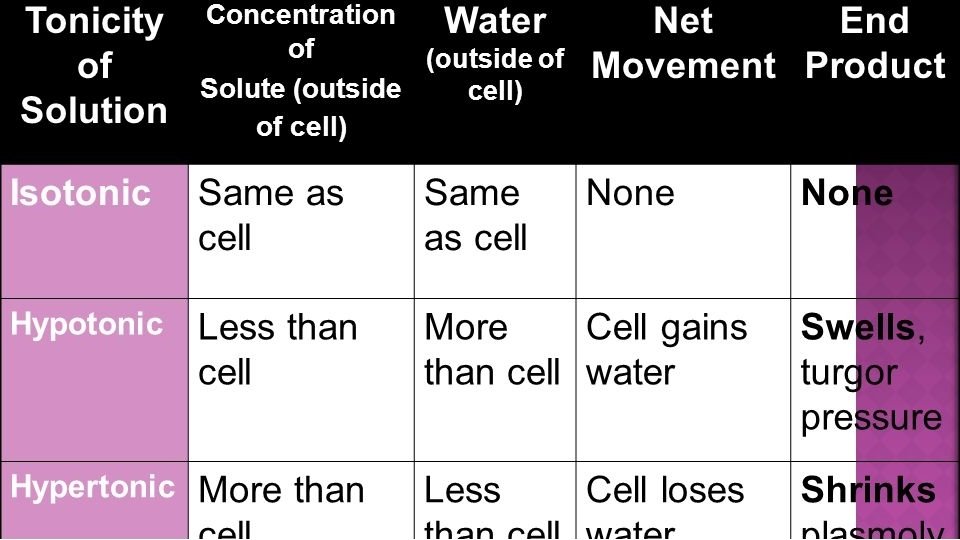
Tonicity • Tonicity is the concentration of particles in a solution as compared to another solution. • There are 3 types of tonicity
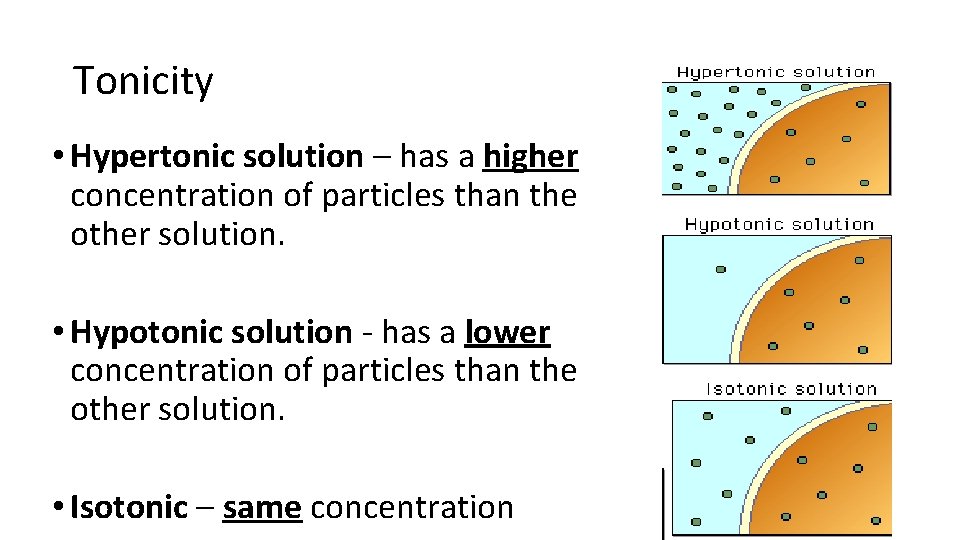
Tonicity • Hypertonic solution – has a higher concentration of particles than the other solution. • Hypotonic solution - has a lower concentration of particles than the other solution. • Isotonic – same concentration

Definitions
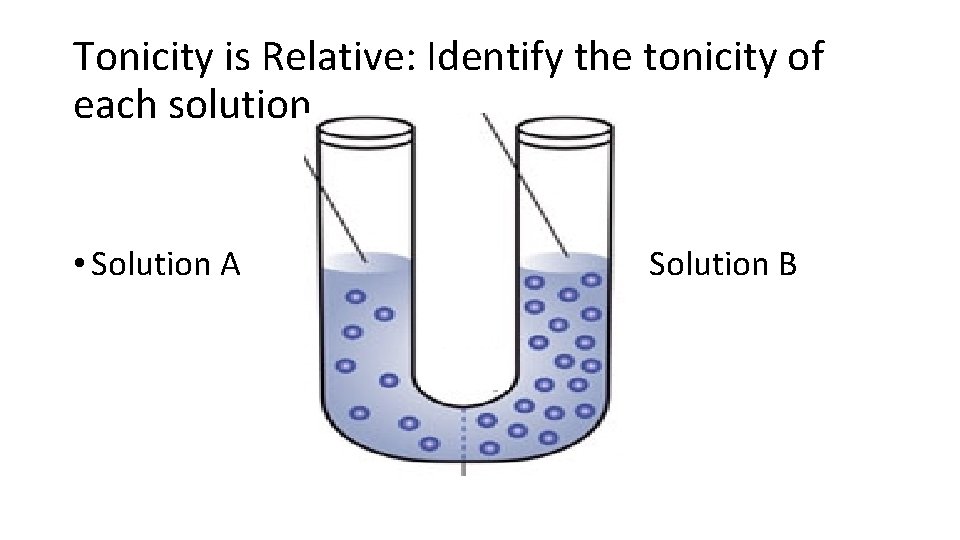
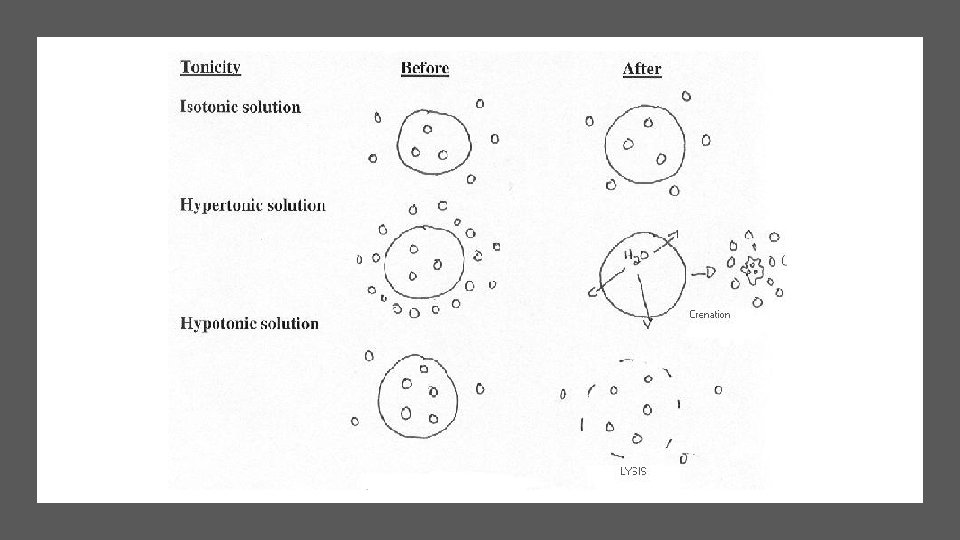
Tonicity is Relative: Identify the tonicity of each solution • Solution A Solution B
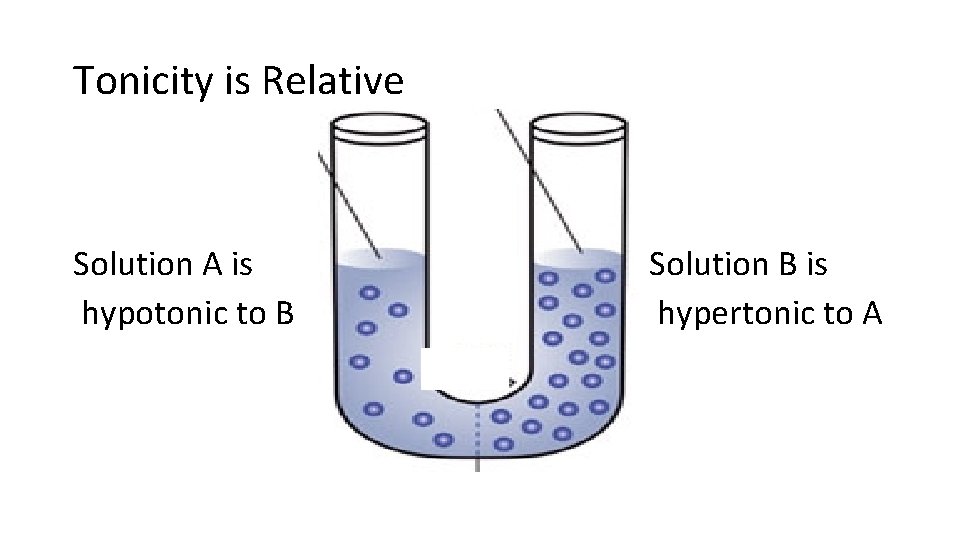
Tonicity is Relative Solution A is hypotonic to B Solution B is hypertonic to A
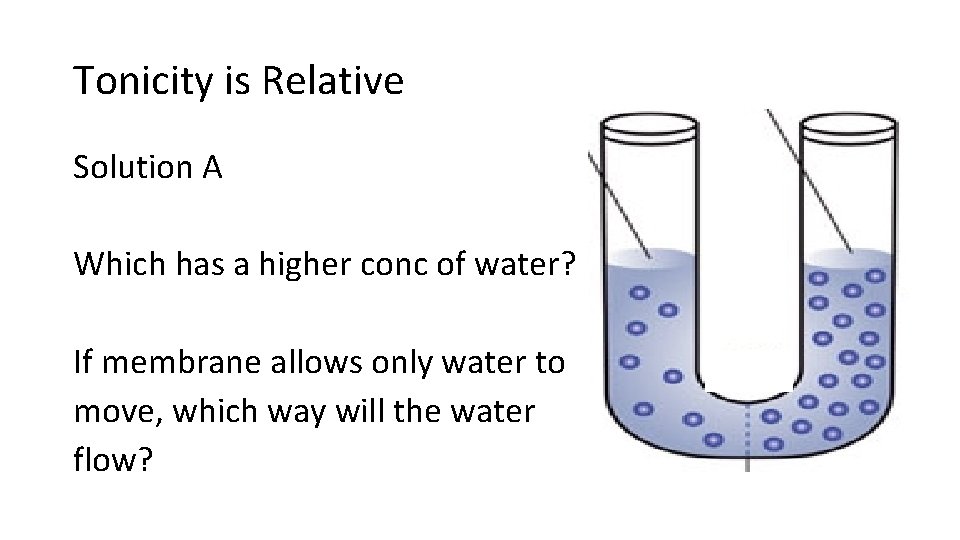
Tonicity is Relative Solution A Which has a higher conc of water? If membrane allows only water to move, which way will the water flow?
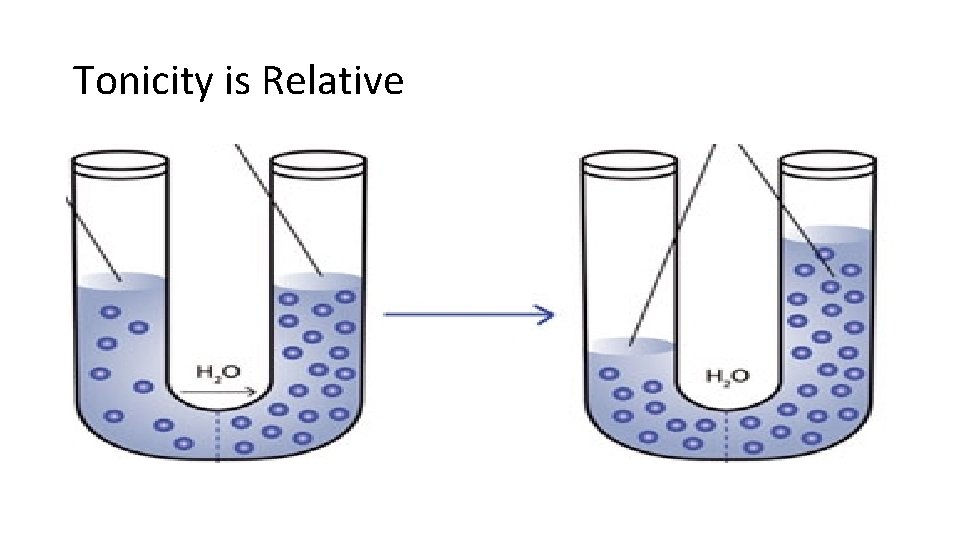
Tonicity is Relative

Osmosis and the Cell If cells are hypertonic to surroundings, more water is moving into cells than is moving out, Cells may swell or even burst. See page 45 (c) Mc. Graw Hill Ryerson 2007

Practice • Diffusion and Osmosis WS • Start Gummy

Tonicity and Osmosis • If a cell has a Low concentration of water compared to its surroundings, It must have a high concentration of particles inside, making the cell hypertonic. • Water moves into the cell.

Tonicity and Osmosis • If a cell has a High Water concentration compared to its surroundings, cell has low particle concentration inside the cell and is hypotonic to surroundings. • Water moves out of cell
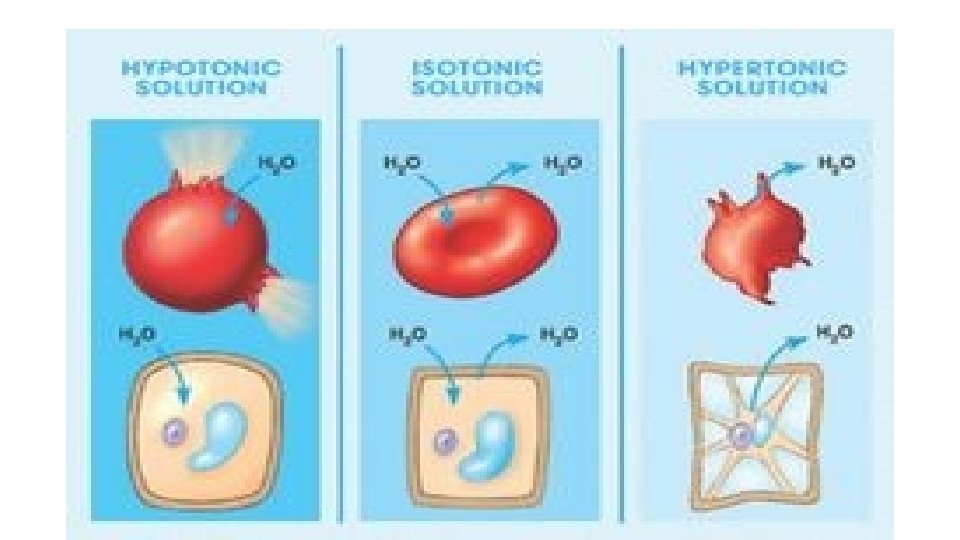
Osmosis and the Cell
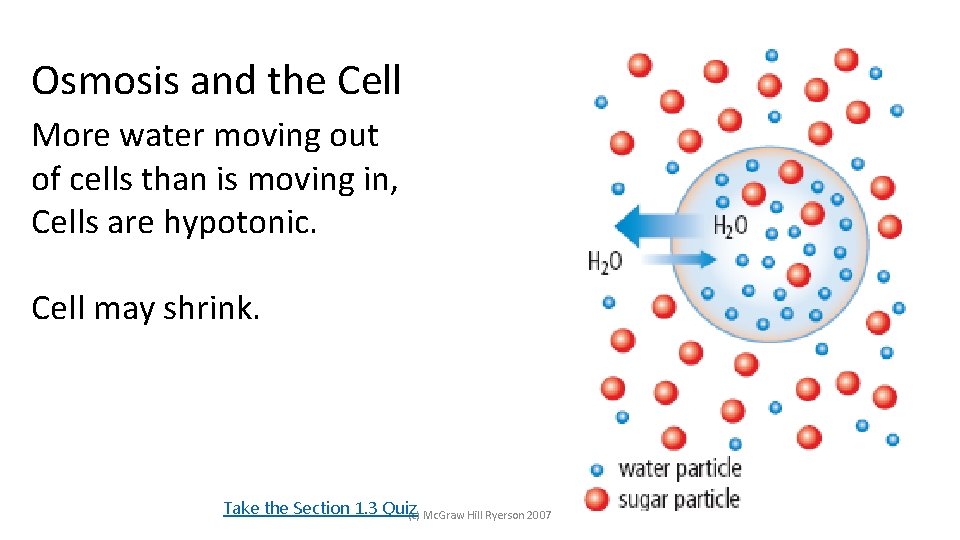
Osmosis and the Cell More water moving out of cells than is moving in, Cells are hypotonic. Cell may shrink. Take the Section 1. 3 Quiz (c) Mc. Graw Hill Ryerson 2007 See page 45

Osmosis and the Cell Equal movement of water in and out of cells. Cell is isotonic. See page 45 (c) Mc. Graw Hill Ryerson 2007

Amoeba Sisters Video on Osmosis • https: //www. youtube. com/watch? v=Ia. Z 8 Mt. F 3 C 6 M

Osmosis Gummy Bear Experiment • Gummy Bear Song • https: //www. youtube. com/watch? v=ast. ISOtt. CQ 0

Osmosis and the cell • Osmosis is the movement (diffusion) of water from High Low Concentration of water) • If a cell has a Low concentration of water compared to its surroundings, • Water will move into the cell
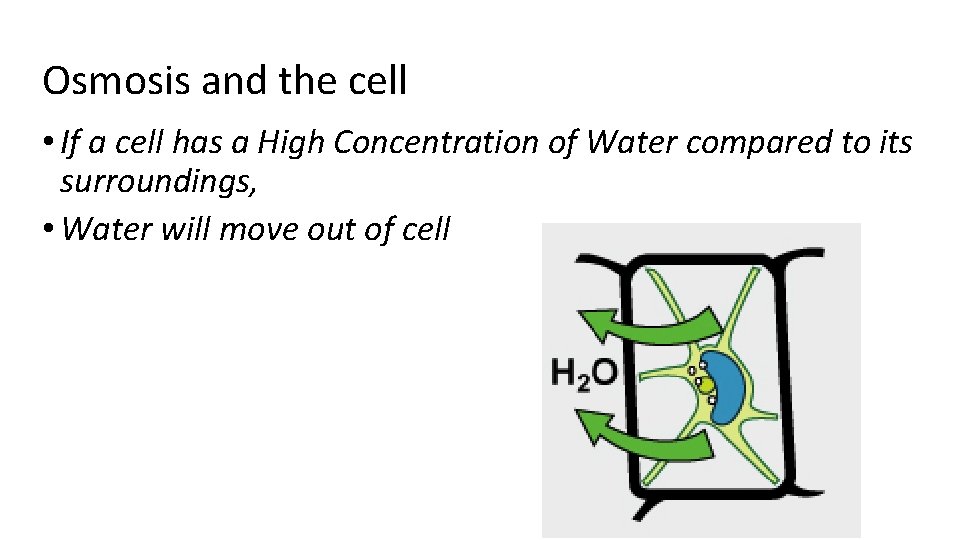
Osmosis and the cell • If a cell has a High Concentration of Water compared to its surroundings, • Water will move out of cell
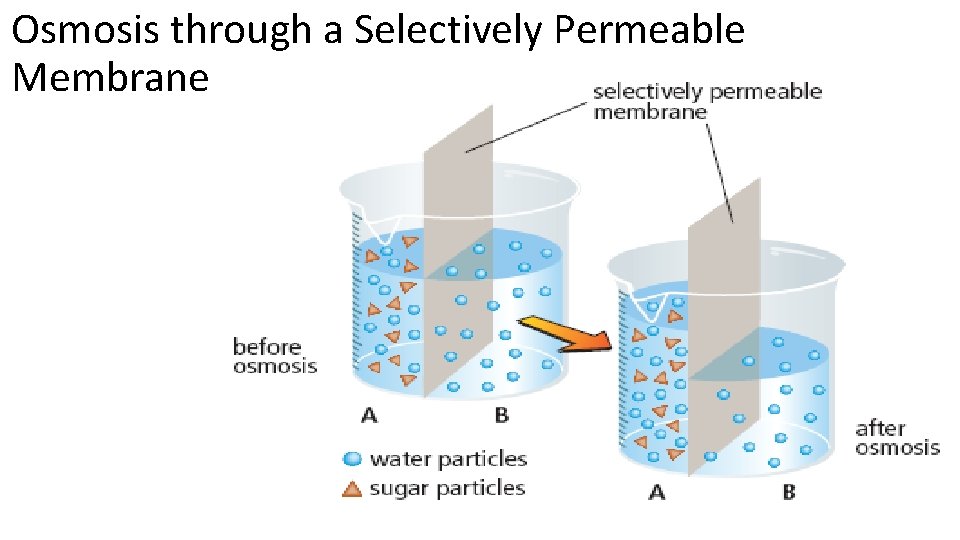
Osmosis through a Selectively Permeable Membrane
- Slides: 42