Difficult Cases Asthma vs COPD Matthew C Bell

Difficult Cases: Asthma vs. COPD Matthew C. Bell, M. D. Assistant Professor of Pediatrics and Medicine University of Arkansas for Medical Sciences Little Rock, AR William W. Busse, M. D. Professor of Medicine University of Wisconsin Hospital and Clinics Madison, WI

Matthew Bell - Disclosure Slide • Employment – University of Arkansas for Medical Sciences • Financial Interests – Nothing to Disclose • Research Interests – Nothing to Disclose • Gifts – Nothing to Disclose • Other Interests – Nothing to Disclose

William Busse - Disclosure Slide • Employment – University of Wisconsin • Financial Interests – Advisory Boards: Merck – Consultant: Novartis, Glaxo. Smith. Kline, Genentech • Data Monitoring Boards and Study Oversight Committees − Boston Scientific − Circassia − Icon • Research Interests – NHLBI, NIAID, • Organizational Interests – AAAAI, ATS, AAP, AAI, CIS • Gifts – Nothing to Disclose • Other Interests – Nothing to Disclose

• 68 year old Caucasian lady presents to your clinic with a chief complaint of “cough and shortness of breath” that became progressively more severe over the past 6 months.

• History of Present Illness – Daily cough with occasional wheeze – Not necessarily worse at any time of day – Cough is productive of whitish sputum – She is able to perform normal activities, but does get short of breath when she climbs more than 1 flight of stairs – Has tried over the counter cough medicines without benefit

• Past Medical History – Mild hypertension on hydrochlorothiazide – Was told she had asthma while in grade school, but was not treated • No problems through teenage years into adulthood – Had “hay fever” as a child, but outgrew this as well – Recently had normal dobutamine stress echocardiogram • Social History – History professor at local university – Current nonsmoker, smoked roughly 1 -1. 5 packs of cigarettes daily through college, graduate school, and the early part of her career equaling about 15 -20 pack years total – Lives with her husband in a suburban neighborhood. No pets. • Family History – Father was a longtime smoker and died of lung cancer at 73 – Mother has “exercise-induced asthma” and is still living at 87

• Physical Exam – Height: 177 cm (5’ 10”) – Weight: 93 kg (205 lbs. ) – HEENT: normal – CV: Normal S 1 S 2, no murmur or gallop – Chest: Clear to auscultation without any adventitious breath sounds – Extremities: no clubbing noted

Which of the following lung function tests would you order first? A. Peak expiratory flow B. Methacholine provocation C. Exercise challenge D. FEV 1/FVC ANSWER on next slide

Which of the following lung function tests would you order first? A. Peak expiratory flow B. Methacholine provocation C. Exercise challenge D. FEV 1/FVC
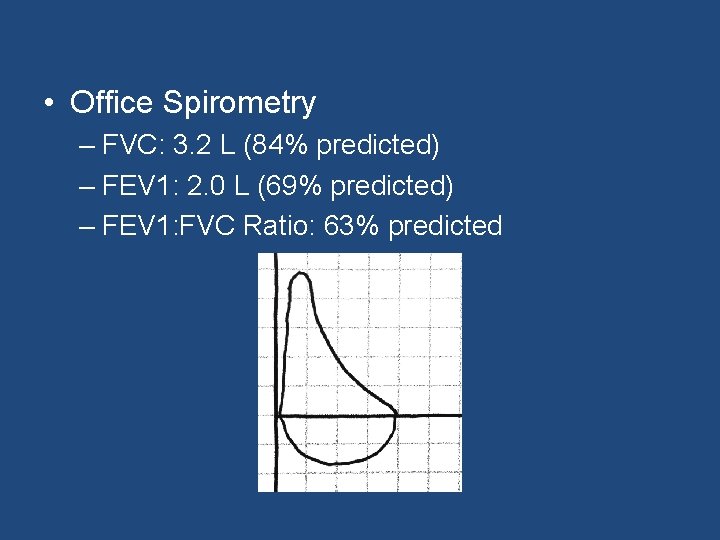
• Office Spirometry – FVC: 3. 2 L (84% predicted) – FEV 1: 2. 0 L (69% predicted) – FEV 1: FVC Ratio: 63% predicted

Based upon the PFT values with an FEV 1/FVC of < 70%, you conclude that the patient has COPD. Based on the GOLD Grades, what is her level of severity? A. GOLD 1 (mild) B. GOLD 2 (moderate) C. GOLD 3 (severe) D. GOLD 4 (very severe) ANSWER on next slide

Based upon the PFT values with an FEV 1/FVC of < 70%, you conclude that the patient has COPD. Based on the GOLD Grades, what is her level of severity? A. GOLD 1 (mild) B. GOLD 2 (moderate) C. GOLD 3 (severe) D. GOLD 4 (very severe)

GOLD (Global Initiative for Chronic Obstructive Lung Disease) Grades in COPD Classification of Severity of Airflow Limitation in COPD (Based on Post. Bronchodilator FEV 1) In patients with FEV 1/FVC < 0. 70: GOLD 1 Mild FEV 1 ≥ 80% predicted GOLD 2 Moderate 50% ≤ FEV 1 < 80% predicted GOLD 3 Severe 30% ≤ FEV 1 < 50% predicted GOLD 4 Very Severe FEV 1 < 30% predicted Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated February 2013. www. goldcopd. org.

• Patient group A—low risk, less symptoms – GOLD 1– 2 (mild or moderate airflow limitation) and 0– 1 exacerbation per year and m. MRC grade 0– 1 or CAT score < 10 • Patient group B—low risk, more symptoms – GOLD 1– 2 (mild or moderate airflow limitation) and 0– 1 exacerbation per year and m. MRC grade > 2 or CAT score > 10 • Patient group C—high risk, less symptoms – GOLD 3– 4 (severe or very severe airflow limitation) and/or > 2 exacerbations per year and/or > 1 hospitalized exacerbation per year and m. MRC grade 0– 1 or CAT score < 10 • Patient group D—high risk, more symptoms – GOLD 3– 4 (severe or very severe airflow limitation) and/or > 2 exacerbations per year / > 1 hospitalized exacerbation per year and m. MRC grade > 2 or CAT score > 10

COPD - Assessment Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated February 2013. www. goldcopd. org.

Based upon the information you have available, which of the following would you select as your initial treatment? A. Albuterol, prn B. Ipratropium, prn C. Tiotropium daily D. LABA + ICS daily ANSWER on next slide

Based upon the information you have available, which of the following would you select as your initial treatment? A. Albuterol, prn B. Ipratropium, prn C. Tiotropium daily D. LABA + ICS daily

In patients who fall in Group B, symptoms but low risk for exacerbations, daily LABA or LAMA are the initial treatments of choice.

Patients in Group C have few symptoms, but a high risk of exacerbations. • ICS +/ LABA or LAMA are the initial treatments of choice
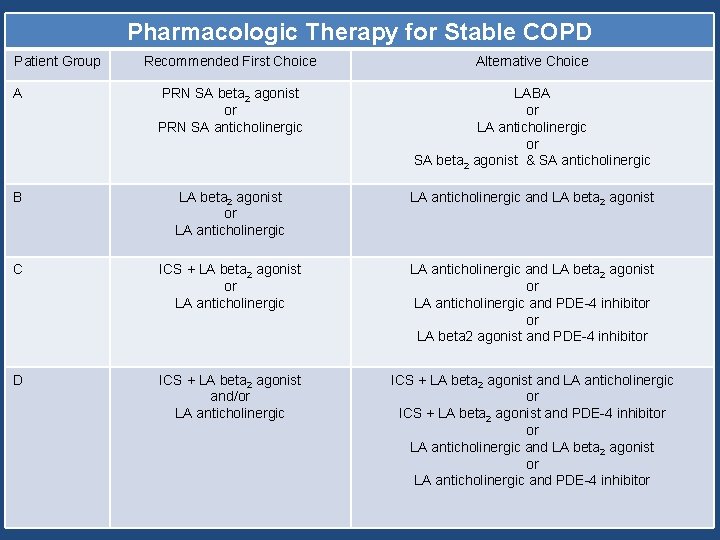
Pharmacologic Therapy for Stable COPD Patient Group Recommended First Choice Alternative Choice A PRN SA beta 2 agonist or PRN SA anticholinergic LABA or LA anticholinergic or SA beta 2 agonist & SA anticholinergic B LA beta 2 agonist or LA anticholinergic and LA beta 2 agonist C ICS + LA beta 2 agonist or LA anticholinergic and PDE-4 inhibitor or LA beta 2 agonist and PDE-4 inhibitor D ICS + LA beta 2 agonist and/or LA anticholinergic ICS + LA beta 2 agonist and LA anticholinergic or ICS + LA beta 2 agonist and PDE-4 inhibitor or LA anticholinergic and LA beta 2 agonist or LA anticholinergic and PDE-4 inhibitor
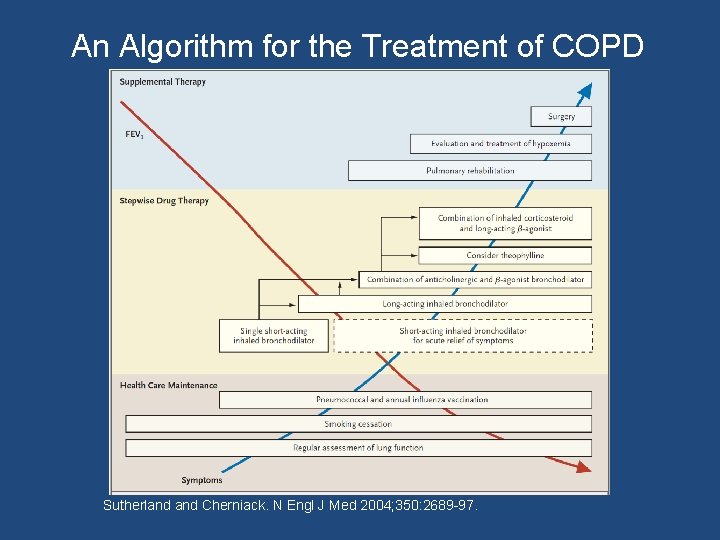
An Algorithm for the Treatment of COPD Sutherland Cherniack. N Engl J Med 2004; 350: 2689 -97.

The patient returns in a month after using tiotropium daily. She has noted some improvement in symptom control but still requires rescue use of albuterol with exercise and upon awakening at night, 2 to 3 times per week. Her physical examination is unchanged as are her spirometry values.

Which of the following tests would you consider next? A. Diffusing capacity (DLco) B. Lung volumes C. Exercise challenge D. Post-bronchodilator FEV 1 ANSWER on next slide

Which of the following tests would you consider next? A. Diffusing capacity (DLco) B. Lung volumes C. Exercise challenge D. Post-bronchodilator FEV 1

Diffusing capacity was 95% predicted • In COPD, emphysema, diffusing capacity is decreased because of parenchymal destruction • In asthma, DLco is normal, or, in some cases, elevated
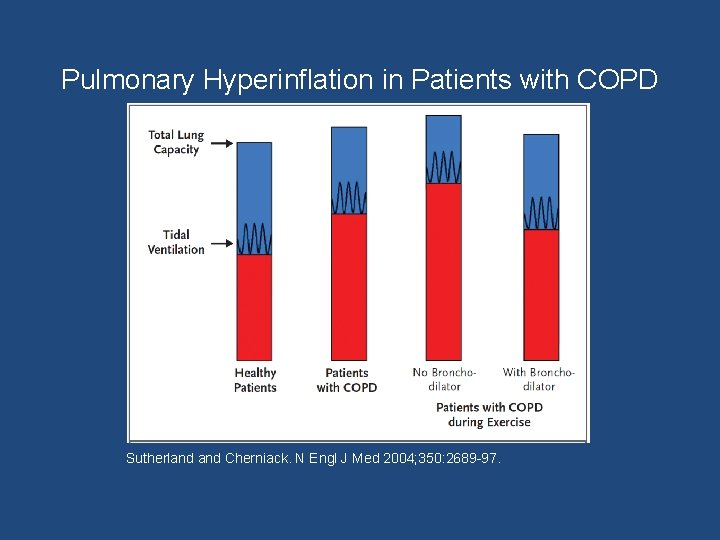
Pulmonary Hyperinflation in Patients with COPD Sutherland Cherniack. N Engl J Med 2004; 350: 2689 -97.
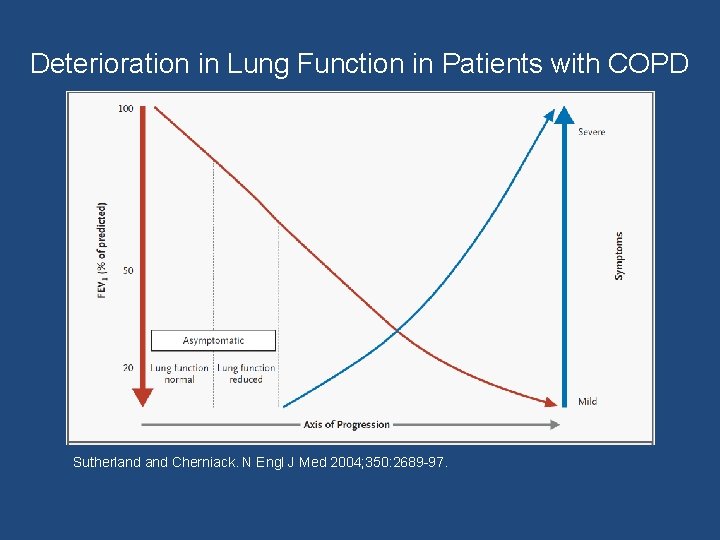
Deterioration in Lung Function in Patients with COPD Sutherland Cherniack. N Engl J Med 2004; 350: 2689 -97.

• Bronchodilator challenge – Spirometry after 4 puffs of albuterol • FVC: 3. 4 L • FEV 1: 2. 24 L (12% improvement and 240 ml) • FEV 1/FVC : 66%

Case Recap • • • 68 year old nonatopic Caucasian female Daily cough, occasional wheeze x 6 months Whitish sputum production No worsening at any certain time of day Dyspnea on exertion with 1 flight of stairs FEV 1=69% predicted, FEV 1/FVC=66% – 12% reversible after bronchodilator • No clinical improvement with LAMA • Methacholine PC 20 is 10 mg/m. L

Asthma or COPD?

COPD • Definition per the ATS – Chronic obstructive pulmonary disease (COPD) is a preventable and treatable disease state characterized by airflow limitation that is not fully reversible. – The airflow limitation is usually progressive and is associated with an abnormal inflammatory response of the lungs to noxious particles or gases, primarily caused by cigarette smoking. – Although COPD affects the lungs, it also produces significant systemic consequences
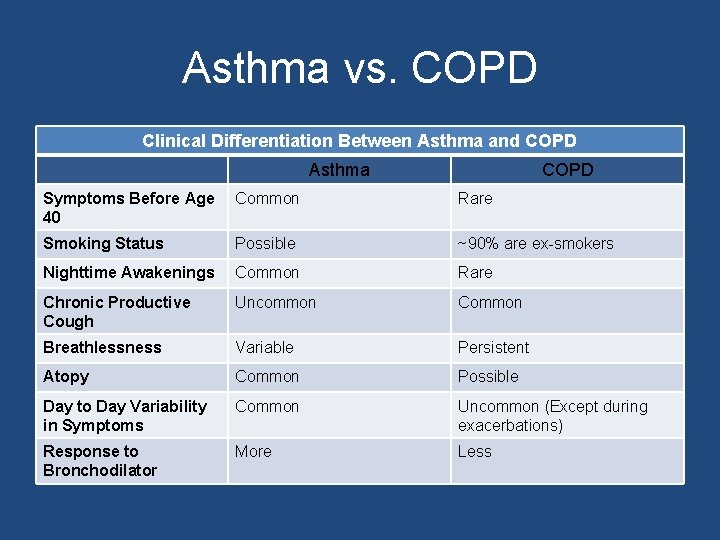
Asthma vs. COPD Clinical Differentiation Between Asthma and COPD Asthma COPD Symptoms Before Age 40 Common Rare Smoking Status Possible ~90% are ex-smokers Nighttime Awakenings Common Rare Chronic Productive Cough Uncommon Common Breathlessness Variable Persistent Atopy Common Possible Day to Day Variability in Symptoms Common Uncommon (Except during exacerbations) Response to Bronchodilator More Less
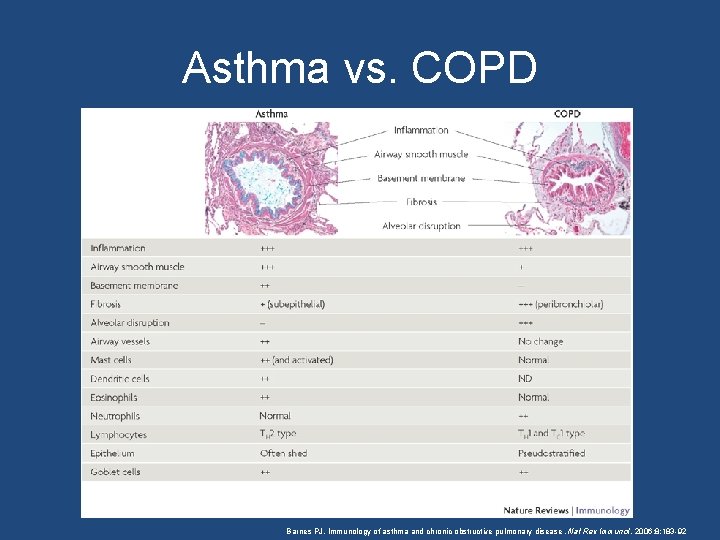
Asthma vs. COPD Barnes PJ. Immunology of asthma and chronic obstructive pulmonary disease. Nat Rev Immunol. 2006; 8: 183 -92

The Overlap Syndrome • As many as 50% of older adults with obstructive airway disease have overlapping diagnoses of asthma and COPD – This percentage increases linearly with age • These patients are often excluded from clinical studies for asthma (because of smoking history) and for COPD (because of at least a partial reversibility of airflow obstruction) – Leading to questions regarding whether recommended treatment regimens are applicable to individual patients with symptom overlap Gibson PG & Simpson JL. The overlap syndrome of asthma and COPD: what are its features and how important is it? . Thorax 2009; 64: 728– 735.

Worldwide, what is the frequency of asthma patients who are current smokers? A. 5% B. 10% C. 25% D. 40% ANSWER on next slide
Worldwide, what is the frequency of asthma patients who are current smokers? A. 5% B. 10% C. 25% D. 40%
• 25% of patients with asthma are current smokers • An additional 15 -20% are former smokers • In the U. S. , particularly high rates of asthma patients who smoke present to emergency departments with acute asthma symptoms Thomson NC, Chaudhuri R, Livingston E. Asthma and cigarette smoking. Eur Respir J. 2004; 24(5): 822 -23.
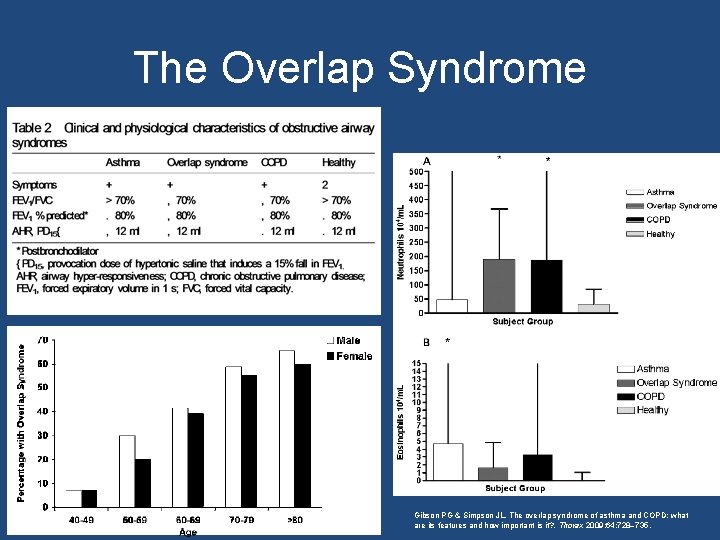
The Overlap Syndrome Gibson PG & Simpson JL. The overlap syndrome of asthma and COPD: what are its features and how important is it? . Thorax 2009; 64: 728– 735.

Conclusion • The line between asthma and COPD can be blurred, especially in older patients – This “blurring” can occur both clinically and histopathologically • Using all available resources to ensure a proper diagnosis allows for appropriate, guidelines-based care • Recognition of an “Overlap Syndrome” may lead to new treatment studies and thus new treatment recommendations

Comments William W. Busse, M. D.
Chronic Obstructive Pulmonary Diseases Asthma Overlap COPD

How frequent is an overlap syndrome seen?

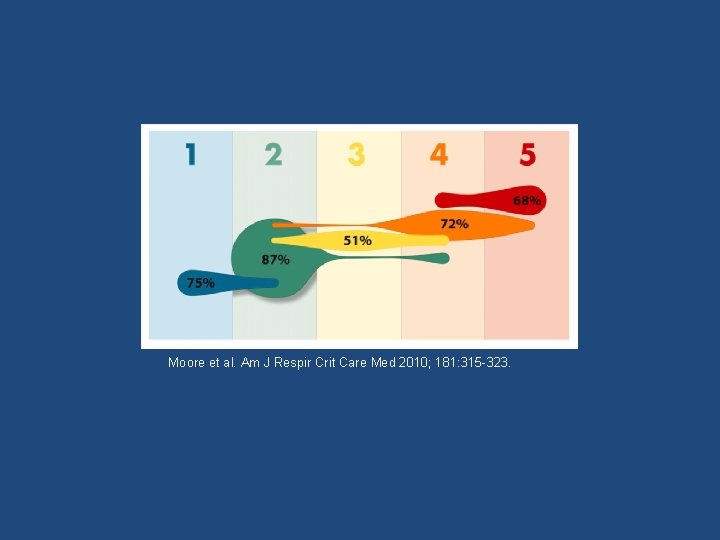
Cluster Analysis to Identify Phenotypes • Perform a multivariate cluster analysis to identify asthma groups who share similar phenotype profiles (defined by the factor scores identified in the variable reduction step) Moore et al. AJRCCM 2010; 181: 315 -323.
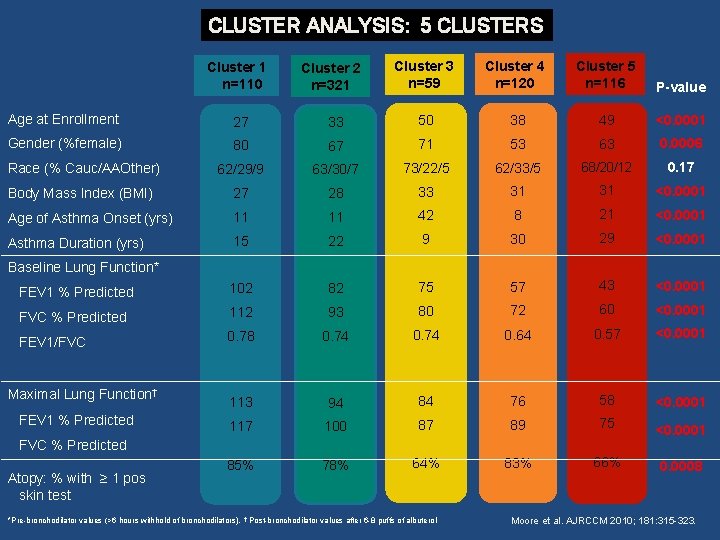
CLUSTER ANALYSIS: 5 CLUSTERS Cluster 1 n=110 Cluster 2 n=321 Cluster 3 n=59 Cluster 4 n=120 Cluster 5 n=116 P-value Age at Enrollment 27 33 50 38 49 <0. 0001 Gender (%female) 80 67 71 53 63 0. 0006 Race (% Cauc/AAOther) 62/29/9 63/30/7 73/22/5 62/33/5 68/20/12 0. 17 Body Mass Index (BMI) 27 28 33 31 31 <0. 0001 Age of Asthma Onset (yrs) 11 11 42 8 21 <0. 0001 Asthma Duration (yrs) 15 22 9 30 29 <0. 0001 FEV 1 % Predicted 102 82 75 57 43 <0. 0001 FVC % Predicted 112 93 80 72 60 <0. 0001 FEV 1/FVC 0. 78 0. 74 0. 64 0. 57 <0. 0001 113 94 84 76 58 <0. 0001 117 100 87 89 75 <0. 0001 85% 78% 64% 83% 66% 0. 0008 Baseline Lung Function* Maximal Lung Function† FEV 1 % Predicted FVC % Predicted Atopy: % with ≥ 1 pos skin test *Pre-bronchodilator values (>6 hours withhold of bronchodilators). † Post-bronchodilator values after 6 -8 puffs of albuterol Moore et al. AJRCCM 2010; 181: 315 -323.
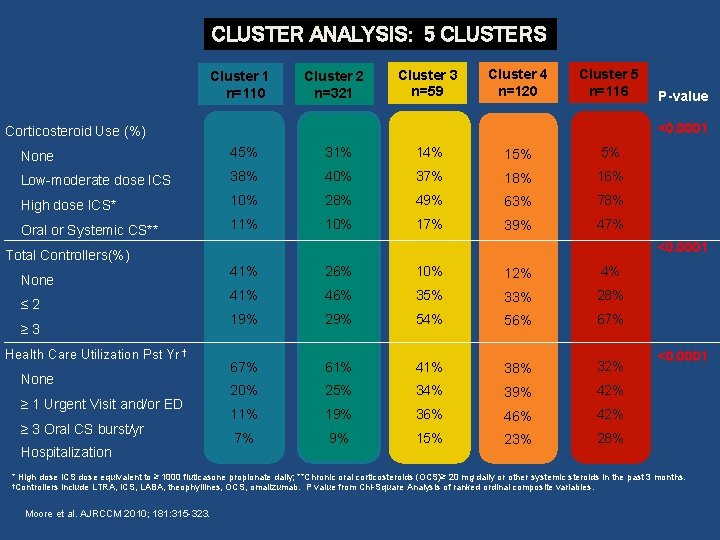
CLUSTER ANALYSIS: 5 CLUSTERS Cluster 1 n=110 Cluster 2 n=321 Cluster 3 n=59 Cluster 4 n=120 Cluster 5 n=116 <0. 0001 Corticosteroid Use (%) None 45% 31% 14% 15% 5% Low-moderate dose ICS 38% 40% 37% 18% 16% High dose ICS* 10% 28% 49% 63% 78% Oral or Systemic CS** 11% 10% 17% 39% 47% <0. 0001 Total Controllers(%) None ≤ 2 ≥ 3 Health Care Utilization Pst Yr † None ≥ 1 Urgent Visit and/or ED ≥ 3 Oral CS burst/yr Hospitalization P-value 41% 26% 10% 12% 4% 41% 46% 35% 33% 28% 19% 29% 54% 56% 67% 61% 41% 38% 32% 20% 25% 34% 39% 42% 11% 19% 36% 42% 7% 9% 15% 23% 28% <0. 0001 * High dose ICS dose equivalent to ≥ 1000 fluticasone propionate daily; **Chronic oral corticosteroids (OCS)≥ 20 mg daily or other systemic steroids in the past 3 months. †Controllers include LTRA, ICS, LABA, theophyllines, OCS, omalizumab. P value from Chi-Square Analysis of ranked ordinal composite variables. Moore et al. AJRCCM 2010; 181: 315 -323.

SARP Cluster Analysis • Cluster 5 in this analysis represents an “overlap” group – FEV 1 at baseline about 45% predicted • Lower baseline FEV 1 suggests COPD – Other factors point to a clear diagnosis of asthma • Younger age at onset (21 years) • Significant bronchodilator reversibility (~15% improvement in FEV 1 after bronchodilator) • Normal DLco
Moore et al. Am J Respir Crit Care Med 2010; 181: 315 -323.

What factors underlie irreversible airflow obstruction in asthma?
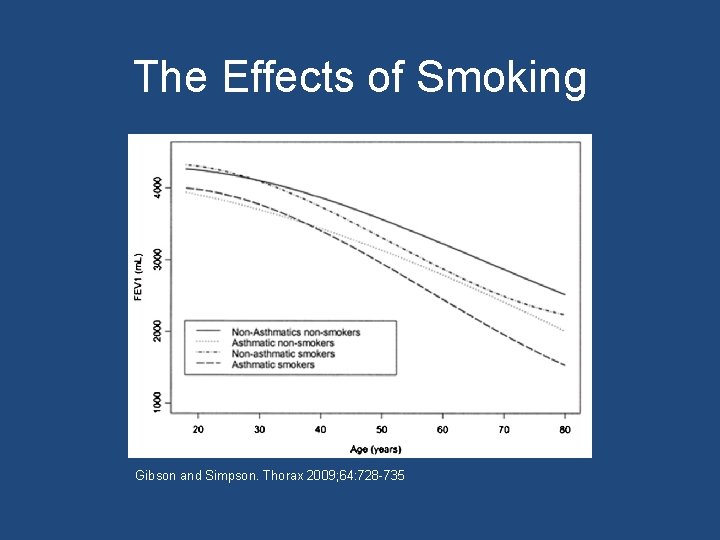
The Effects of Smoking Gibson and Simpson. Thorax 2009; 64: 728 -735

Bai et al. Severe exacerbations predict excess lung function decline in asthma. Eur Respir J 2007; 30: 452 -456. • Evaluated the effect of severe exacerbations on the progression of airway obstruction in 93 non-smoking asthma patients prior to treatment with inhaled corticosteroids.
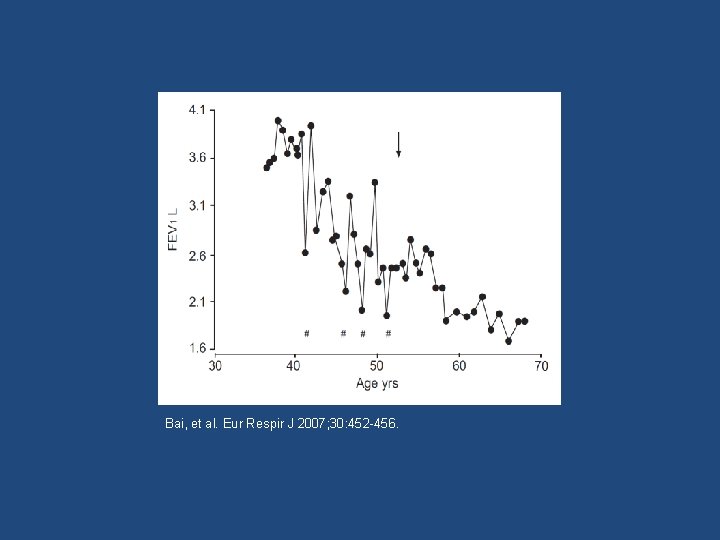
Bai, et al. Eur Respir J 2007; 30: 452 -456.
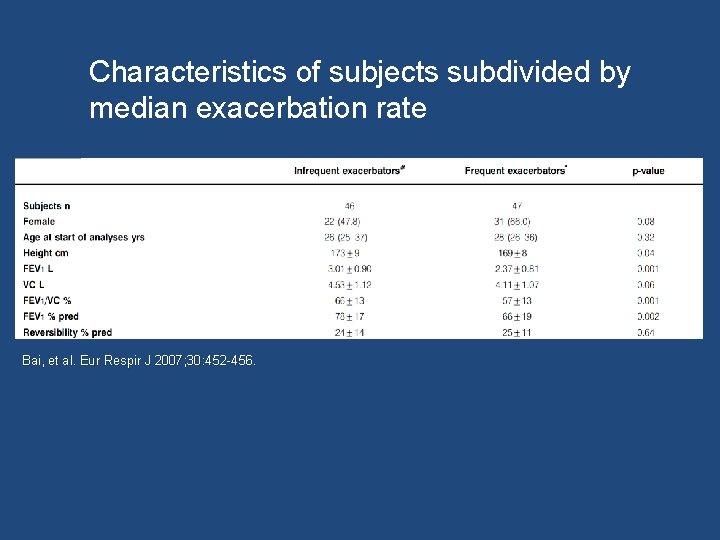
Characteristics of subjects subdivided by median exacerbation rate Bai, et al. Eur Respir J 2007; 30: 452 -456.
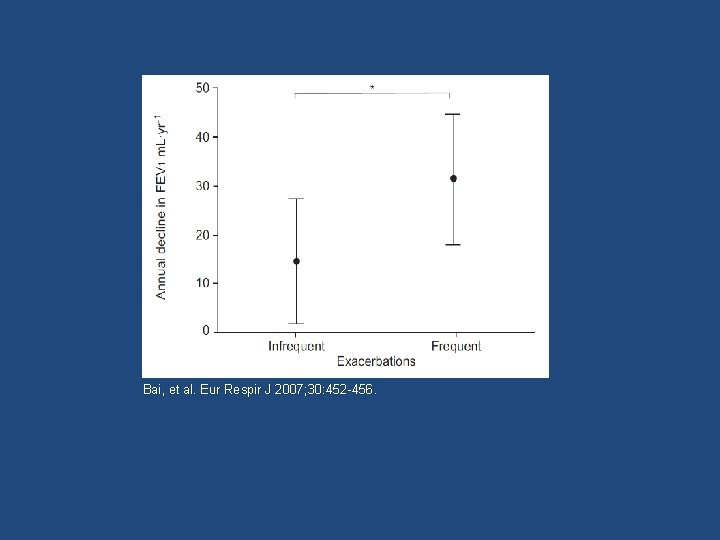
Bai, et al. Eur Respir J 2007; 30: 452 -456.

Why is the “smoking-related” overlap category important?
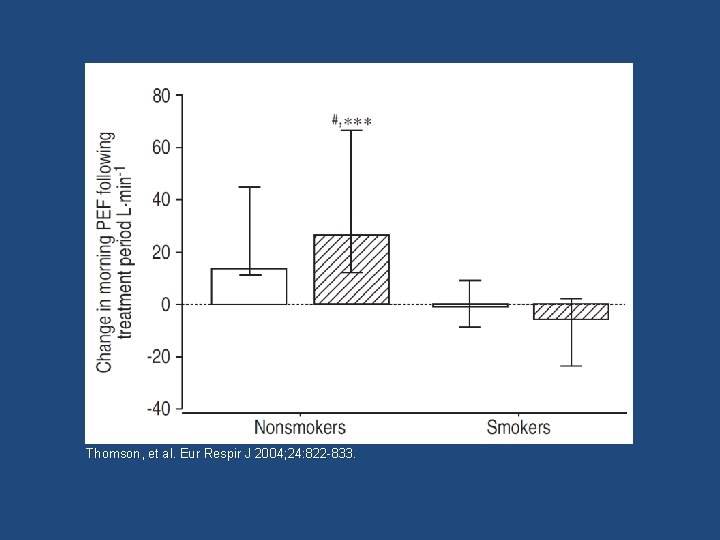
Thomson, et al. Eur Respir J 2004; 24: 822 -833.

Smoking-Related Overlap Category • Smokers in this overlap group tended to have more fixed obstruction – Less responsiveness to controller bronchodilator therapy as measured by a lack of change in morning peak expiratory flow after a treatment period when compared to non-smokers
- Slides: 56