Differential scanning calorimetry study of maize starch gelatinization














- Slides: 24

Differential scanning calorimetry study of maize starch gelatinization in presence of glucose 18 th European Young Cereal Scientists and Technologists Workshop Brecht Verbauwhede, Lomme Deleu, Jan Delcour, Bart Goderis Laboratory of Food Chemistry and Biochemistry 15/04/2019

Many thanks to… Dr. Lomme Deleu Prof. Dr. Bart Goderis Prof. Dr. Jan Delcour Laboratory of Food Chemistry and Biochemistry 2 Polymer chemistry and materials Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Overview • Introduction • Research question • Results and discussion • Conclusion 3 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Introduction 4

Introduction Starch sources and composition • Sources • Cereals • Roots and tubers • Legumes • Composition Amylose (AM) 18 -30% • • Amylopectin (AP) 82 -70% Depends on botanical origin Waxy starches: almost no AM present High Amylose starches: > 30% Minor constituents: lipids (only in cereal starches), protein, water (Buléon et al. , 1998) 5 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

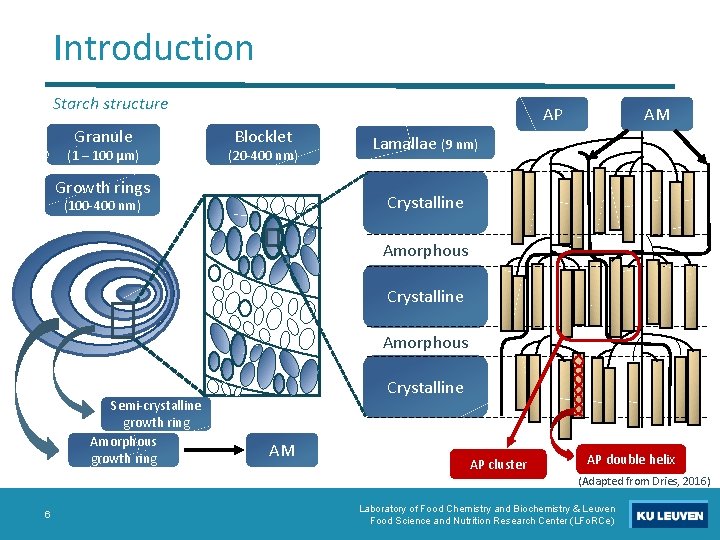
Introduction Starch structure Granule (1 – 100 µm) AP Blocklet (20 -400 nm) Growth rings AM Lamallae (9 nm) Crystalline (100 -400 nm) Amorphous Crystalline Amorphous Semi-crystalline growth ring Amorphous growth ring Crystalline AM AP cluster AP double helix (Adapted from Dries, 2016) 6 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

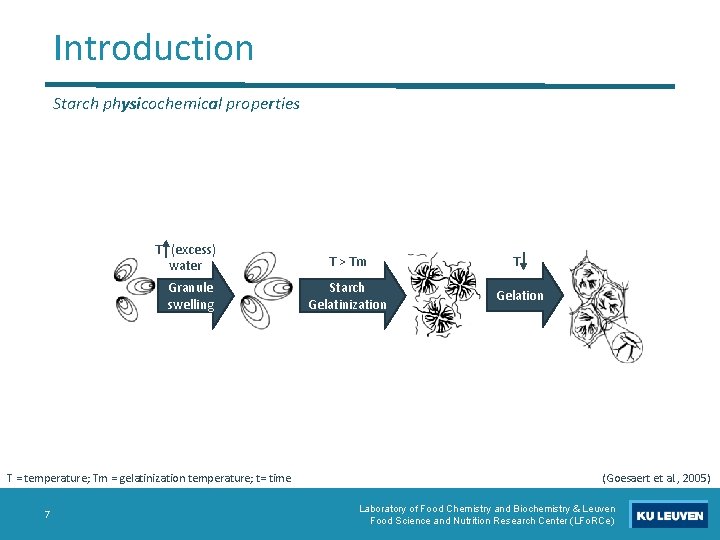
Introduction Starch physicochemical properties T (excess) water Granule swelling T = temperature; Tm = gelatinization temperature; t= time 7 T > Tm T Starch Gelatinization Gelation (Goesaert et al. , 2005) Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

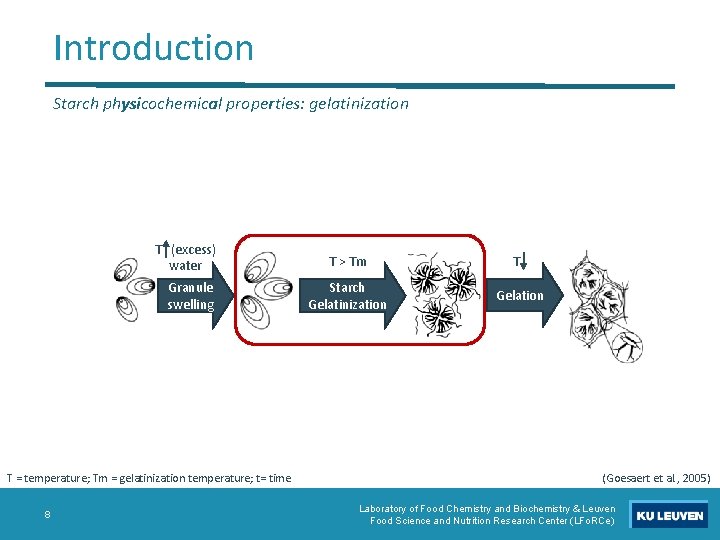
Introduction Starch physicochemical properties: gelatinization T (excess) water Granule swelling T = temperature; Tm = gelatinization temperature; t= time 8 T > Tm T Starch Gelatinization Gelation (Goesaert et al. , 2005) Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Introduction Starch physicochemical properties: gelatinization T > Tm Starch Gelatinization • Loss of birefringence • Loss of molecular order • AP crystal melting • Breakage of intramolecular hydrogen bonds • Amylose leaching T = temperature; Tm = gelatinization temperature 9 (Jenskins and Donald, 1998); (Goesaert et al. , 2005); (Dries, 2016) Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Introduction Starch physicochemical properties: gelatinization T > Tm Starch Gelatinization • Studied by differential scanning calorimetry (DSC) • Detection of phase transitions through monitoring energy consumption and release • Reference and sample cell heated at same rate • During phase transitions in sample difference in applied heat T = temperature; Tm = gelatinization temperature 10 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

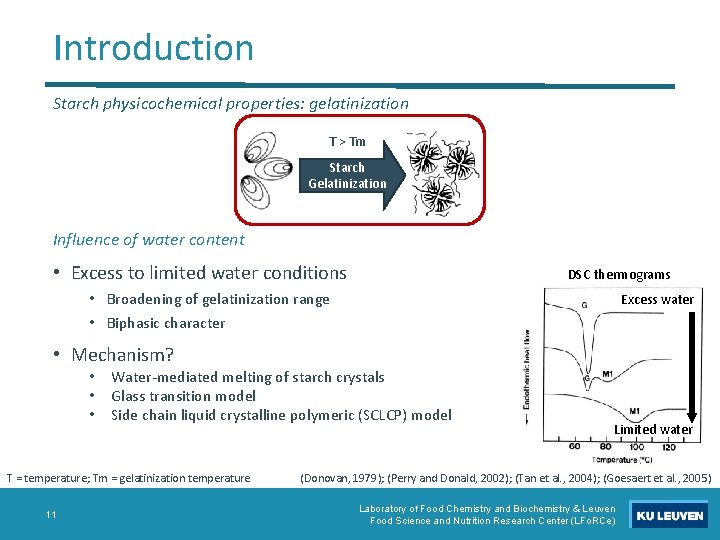
Introduction Starch physicochemical properties: gelatinization T > Tm Starch Gelatinization Influence of water content • Excess to limited water conditions DSC thermograms • Broadening of gelatinization range • Biphasic character Excess water • Mechanism? • • • Water-mediated melting of starch crystals Glass transition model Side chain liquid crystalline polymeric (SCLCP) model T = temperature; Tm = gelatinization temperature 11 Limited water (Donovan, 1979); (Perry and Donald, 2002); (Tan et al. , 2004); (Goesaert et al. , 2005) Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Introduction Starch physicochemical properties: gelatinization T > Tm Starch Gelatinization DSC thermograms Glucose Influence of sugars 0% 5% 10% 20% 40% • Dependent on LMWCH and concentration • Decrease, increase or no influence on gelatinization enthalpy Heat Flow (W/g) • Delay gelatinization (higher Tm) Sucrose • No consensus on mechanism Temperature (°C) DSC = differential scanning calorimetry 12 0% 5% 10% 20% 40% (Baek et al. , 2004); (Gunaratne et al. , 2007); (Perry and Donald, 2002); (Goesaert et al. , 2005) Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Research question 13

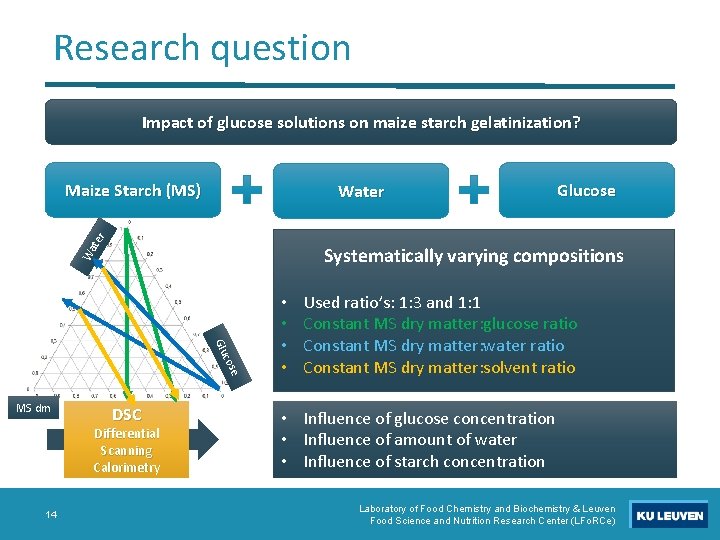
Research question Impact of glucose solutions on maize starch gelatinization? Maize Starch (MS) Wa ter Water Systematically varying compositions e cos Glu MS dm DSC Differential Scanning Calorimetry 14 Glucose • • Used ratio’s: 1: 3 and 1: 1 Constant MS dry matter: glucose ratio Constant MS dry matter: water ratio Constant MS dry matter: solvent ratio • Influence of glucose concentration • Influence of amount of water • Influence of starch concentration Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Method 15

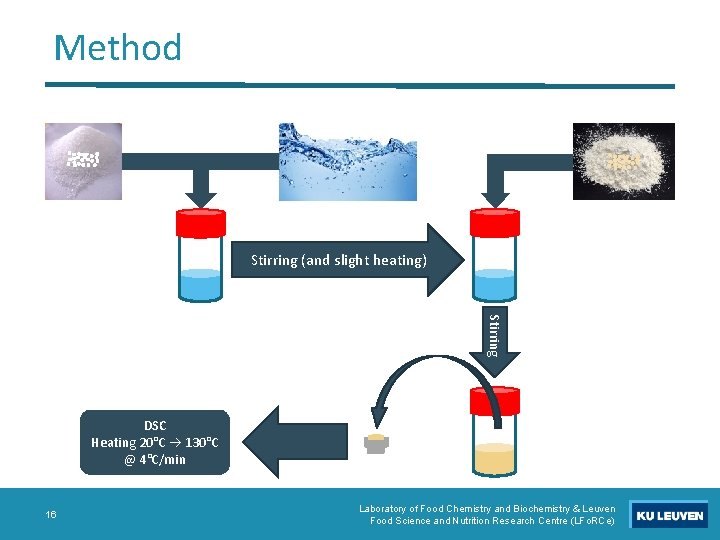
Method Stirring (and slight heating) Stirring DSC Heating 20°C 130°C @ 4°C/min 16 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Centre (LFo. RCe)

Results and discussion 17

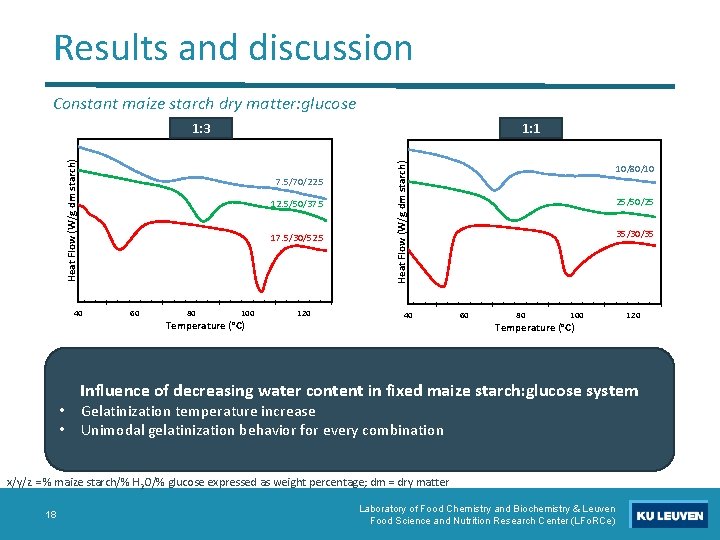
Results and discussion Constant maize starch dry matter: glucose 7. 5/70/22. 5 12. 5/50/37. 5 17. 5/30/52. 5 40 • • 60 80 100 Temperature (°C) 120 Heat Flow (W/g dm starch) 1: 1 Heat Flow (W/g dm starch) 1: 3 40 10/80/10 25/50/25 35/30/35 60 80 100 Temperature (°C) Influence of decreasing water content in fixed maize starch: glucose system Gelatinization temperature increase Unimodal gelatinization behavior for every combination x/y/z = % maize starch/% H 2 O/% glucose expressed as weight percentage; dm = dry matter 18 120 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

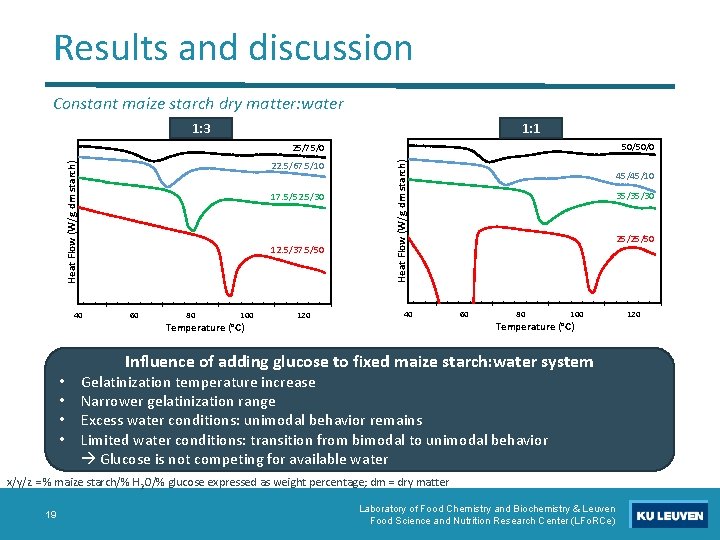
Results and discussion Constant maize starch dry matter: water 1: 3 1: 1 50/50/0 Heat Flow (W/g dm starch) 22. 5/67. 5/10 17. 5/52. 5/30 12. 5/37. 5/50 40 • • 60 80 100 Temperature (°C) 120 Heat Flow (W/g dm starch) 25/75/0 40 45/45/10 35/35/30 25/25/50 60 80 100 Temperature (°C) Influence of adding glucose to fixed maize starch: water system Gelatinization temperature increase Narrower gelatinization range Excess water conditions: unimodal behavior remains Limited water conditions: transition from bimodal to unimodal behavior Glucose is not competing for available water x/y/z = % maize starch/% H 2 O/% glucose expressed as weight percentage; dm = dry matter 19 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe) 120

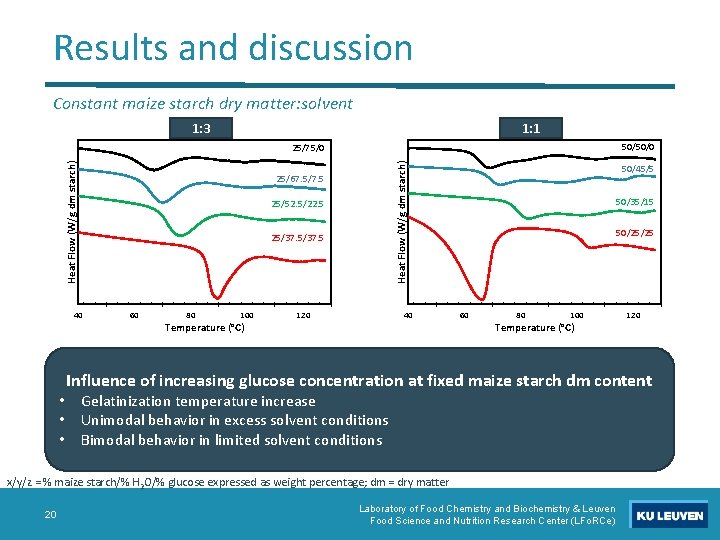
Results and discussion Constant maize starch dry matter: solvent 1: 3 1: 1 50/50/0 25/67. 5/7. 5 25/52. 5/22. 5 25/37. 5 40 • • • Heat Flow (W/g dm starch) 25/75/0 60 80 100 Temperature (°C) 120 40 50/45/5 50/35/15 50/25/25 60 80 100 Temperature (°C) Influence of increasing glucose concentration at fixed maize starch dm content Gelatinization temperature increase Unimodal behavior in excess solvent conditions Bimodal behavior in limited solvent conditions x/y/z = % maize starch/% H 2 O/% glucose expressed as weight percentage; dm = dry matter 20 120 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Conclusion 21

Conclusion • Increasing glucose concentration Increasing gelatinization temperature • Increasing starch concentration Transition from unimodal to bimodal gelatinization The glucose concentration in the solvent determines gelatinization temperature The amount of solvent determines gelatinization behavior 22 Laboratory of Food Chemistry and Biochemistry & Leuven Food Science and Nutrition Research Center (LFo. RCe)

Differential scanning calorimetry study of maize starch gelatinization in presence of glucose 18 th European Young Cereal Scientists and Technologists Workshop Brecht Verbauwhede, Lomme Deleu, Jan Delcour, Bart Goderis Laboratory of Food Chemistry and Biochemistry 15/04/2019
