DielsAlder Reactions 1 2 3 4 Tetraphenylnaphthalene Hexaphenylbenzene

Diels-Alder Reactions • • • 1, 2, 3, 4 -Tetraphenylnaphthalene Hexaphenylbenzene Dimethyl Tetraphenylphthalate Triptycene Experimental Write-up

1, 2, 3, 4 -Tetraphenylnaphthalene Large rxn tube & boiling chip 1. 500 mg tetraphenylcyclopentadienone (purple solid) 2. 3 m. L glyme (1, 2 -dimethoxyethane) 3. 0. 35 m. L isoamyl nitrite Caution: Remove any plastic coatings from reaction tubes. 1. Heat to a gentle reflux and then time for 2 -5 min. 2. In a separate small vial dissolve 250 mg anthranilic acid in 2 m. L glyme. 3. Add the acid/glyme mixture to the refluxing solution dropwise with a pipette. • Bubbling should occur. • Look for a color change from dark brown to yellow. • If color change does not occur add an additional 0. 35 m. L isoamyl nitrite. • Heat for an additional 2 min. 4. Cap and shake tube and let it stand at room temperature. (Move on to next reaction while compound precipitates out. ) 5. Add 10 m. L ethanol and 5 m. L saturated sodium bicarbonate, shake well and allow solid to form. 6. Vacuum filter: wash with cold water twice followed by cold ethanol. 7. Recrystallize with nitrobenzene/ethanol. 8. Weigh and record MP.
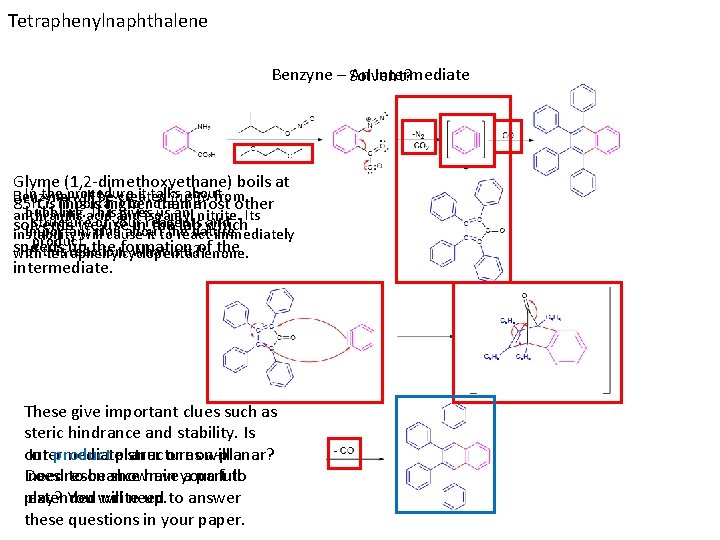
Tetraphenylnaphthalene Benzyne – Solvent? An Intermediate Glyme (1, 2 -dimethoxyethane) boils at In the procedure it talks about Benzyne willisbe created inthe situ from It is This important to note 85ᵒC. higher than most other bubbling. acid Thisand givesisoamyl us an nitrite. Its anthranilic structure ofuse your and solvents weclue inreagents thethe lab which important about nature instability will cause it to react immediately product. speeds up the formation of of this reaction. What is it? the with tetraphenylcyclopentadienone. intermediate. These give important clues such as steric hindrance and stability. Is our product planar or non-planar? Intermediate structures will Does have a partfull to needresonance to be shown in your play? You will need to answer extended write-up. these questions in your paper.

Hexaphenylbenzene A neat reaction (without solvent) Small DRY rxn tube 100 mg tetraphenylcyclopentadienone 500 mg diphenylacetylene Caution: Remove any plastic coatings from reaction tubes. 1. Turn heat up all the way on sand bath. Reaction requires a temperature of ~300ᵒC. • Use a spatula to build up sand around the base of the tube to increase heat. • Lightly place a cap on reaction tube in case it bumps. 2. Heat mixture until mixture changes color from purple to brown. 3. Pull out of sand lightly shake to observe if any white solid forms at the bottom while mixture is cooling. • Reflux longer if no solid forms upon cooling. 4. Cool and then add 2 m. L diphenyl ether and heat in sand bath until solid is dissolved. 5. Cool tube to RT before placing 2 m. L of toluene into the product. • Let tube cool or ice bath will make tube shatter if it’s hot. 6. Vacuum filter and wash white solid with toluene.
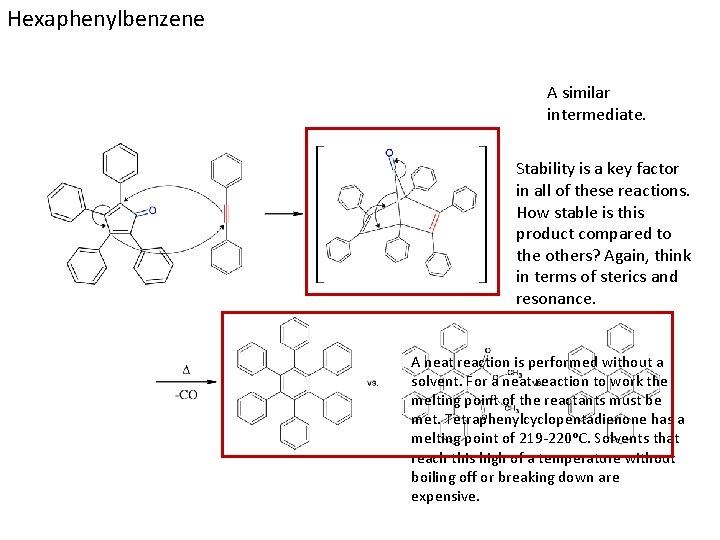
Hexaphenylbenzene A similar intermediate. Stability is a key factor in all of these reactions. How stable is this product compared to the others? Again, think in terms of sterics and resonance. A neat reaction is performed without a solvent. For a neat reaction to work the melting point of the reactants must be met. Tetraphenylcyclopentadienone has a melting point of 219 -220ᵒC. Solvents that reach this high of a temperature without boiling off or breaking down are expensive.
Dimethyl Tetraphenylphthalate Small rxn tube & boiling stick 100 mg tetraphenylcyclopentadienone 0. 1 m. L dimethyl acetylenedicarboxylate 1 m. L nitrobenzene Caution: Remove any plastic coatings from reaction tubes. 1. Heat rxn tube & contents until reflux. Color will change from purple to a tan. 2. Cool to room temp FIRST then add 3 m. L ethanol. 3. Cool in an ice bath. 4. Vacuum filter w/ cold ethanol, dry, and weigh.
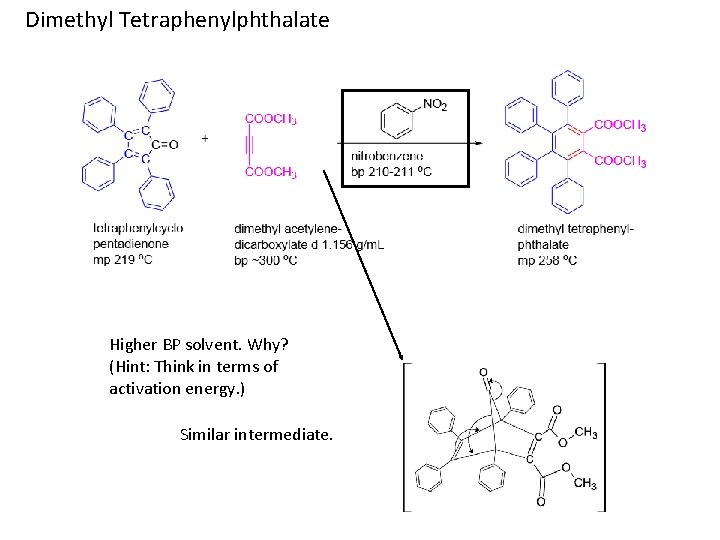
Dimethyl Tetraphenylphthalate Higher BP solvent. Why? (Hint: Think in terms of activation energy. ) Similar intermediate.

Triptycene Rxn Tube 1 – w/ boiling chip Rxn Tube 2 400 mg anthracene 0. 4 m. L isoamyl nitrite 4 m. L glyme (1, 2 -dimethoxyethane) 520 mg anthranilic acid 2 m. L glyme (1, 2 -dimethoxyethane) 1. Add contents of tube 2 into tube 1 dropwise over 20 -minute period through a septum. 2. After 20 min addition, add 0. 4 m. L isoamyl nitrite. 3. Reflux for 10 min and then cool to RT. 4. Add 5 m. L Et. OH followed by 10 m. L sodium hydroxide soln (3 M). 5. Vacuum filter and wash with cold ethanol then cold water (brown color should fade). Weigh and record this crude product. 6. Place your crude product in 25 m. L round bottom flask and add 200 mg maleic anhydride & 4 m. L triglyme (NOT glyme). Fit with a reflux condenser and reflux on sand bath for 5 min. Cool to RT. 7. Add 2 m. L Et. OH and 6 m. L sodium hydroxide soln (3 M). Filter and rinse with cold Et. OH followed by cold water. 8. Recrystallize with methanol and filter, dry and weigh.
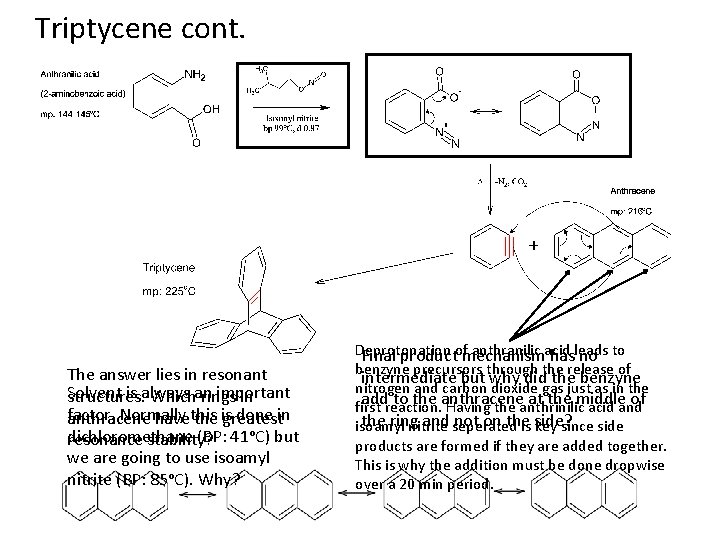
Triptycene cont. The answer lies in resonant Solvent is always important structures. Whichan rings in factor. Normally done in anthracene havethis the is greatest dichloromethane (BP: 41ᵒC) but resonance stability? we are going to use isoamyl nitrite (BP: 85ᵒC). Why? Deprotonation anthranilic acid Final productofmechanism hasleads no to benzyne precursors through the release of intermediate but why did the benzyne nitrogen and carbon dioxide gas just as in the addreaction. to the anthracene at the middle of first Having the anthrinilic acid and the ring and seperated not on theis side? isoamyl nitrite key since side products are formed if they are added together. This is why the addition must be done dropwise over a 20 min period.

Experimental Write-Up Significant figures must be a part Reaction title is boldsince and there is of your experimental Compound names are not they acapitalized period every title. be Title is could playafter aunless limiting role. Solvent volumes atshould the Decimals always have zeros in part of the placed after solvent name, beginning ofparagraph. a sentence. Italicized front them. notof before. words will never be capitalized. Example: Notice that all reactants have the same reactants amount ONLY of sig figs while require solventsmmol only amounts, NOT solvents. Use Common abbreviations The only exception is %have the require. Always one. Your product will include spaces correct for m. L limiting (not are acceptable use. recovery. same number ofnotation sig figstoas your between amounts and units. or ML). your % yeild). reagent ml (including 2, 5 -Di-tert-butylacenaphthene. A soln of acenaphthene (15. 1 g, 97. 7 mmol) and aluminum chloride (2. 65 g, 19. 9 mmol) in carbon disulfide (100 m. L) were added to tert-butyl-chloride (20. m. L, 180 mmol) dropwise via syringe over 25 min. The soln was stirred for an additional 3 h, then refluxed for 1 h. Upon cooling, water (100 m. L) was cautiously added, followed by concentrated HCl (5 m. L) and stirring was continued until all of the dark solid had dissolved. The mixture was extracted with carbon disulfide, dried over Na 2 SO 4, and filtered. The solvent was removed under vacuum and the resulting yellowish wax was recrystallized once in glacial acetic acid and then once in toluene/ethanol to yield a white precipitate (7. 42 g, 27. 8 mmol, 30. 3%): mp 161 -162 ºC. Any color changes and descriptions must be given. Always include a description of your product. No space on % recovery Melting point must be given as a range ONLY if you recorded one.

A Write-Up Checklist What you do NOT need to do: • To explain processes such as recrystallization, how to set up an apparatus, or include what you think happened. Check these off after you have completed your write-up. o o o o Correct tense – Are there any “I”, “you”, “me”, “they”, “he”, “she” or present tense words (-ing, -es words), etc… in the experimental? Remove them and restructure your sentence(s) to reflect a past tense, 3 rd person point of view. All reactants are given mmol amounts while solvents were not. Liquid reactants should have mass included (e. g. “isoamyl nitrite (0. 35 m. L, 0. 30 g, 2. 6 mmol)”). Provide information about the product if it was requested in the procedure (mass, MP as a range, BP, % yield). A description of the product should always be included (color, texture, phase, smell, etc…). Crude weights are included in the same experimental paragraph. Shorthand may be used but always define it in its first mention (e. g. “hydrochloric acid (HCl)”). Include conditions such as time under reflux, ice bath, temperatures, crystallization solvents, filtration method, etc… Mass, mmol, and % yield of product reported at end along with MP range if recorded.
- Slides: 11