Dielectrics II ClausiusMossotti relation The LangevinDebye Formula Dr

Dielectrics II Clausius-Mossotti relation The Langevin-Debye Formula Dr. Ashok Kumar Dimri M S College Saharanpur U. P.
Clausius-Mossotti relation Non polar molecules No permanent dipole moment Material as a whole is neutral Polarization occurs in presence of applied field Ethane , A non polar molecule Paraffin paper, Mica, Pure Water , Bakelite, Fused quartz Pyrex glass, Strontium titanate and Teflon
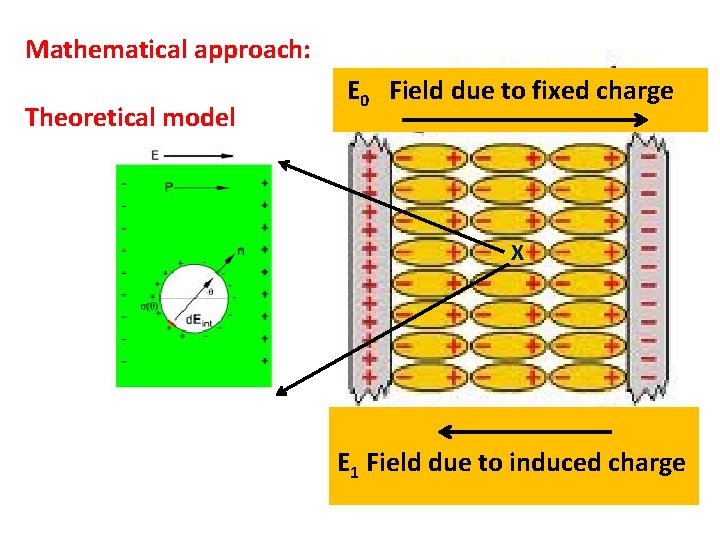
Mathematical approach: Theoretical model E 0 Field due to fixed charge X E 1 Field due to induced charge

Local electric field at an atom at X Elocal=E 0+E 1+E 2+E 3 The field E 1 is called the depolarization field, for within the body it tends to oppose the applied field E 0 E 2 Lorentz cavity field: field from polarization charges on inside of a spherical cavity cut (as a mathematical fiction) out of the specimen with the reference atom as center. E 3 field of atoms inside cavity
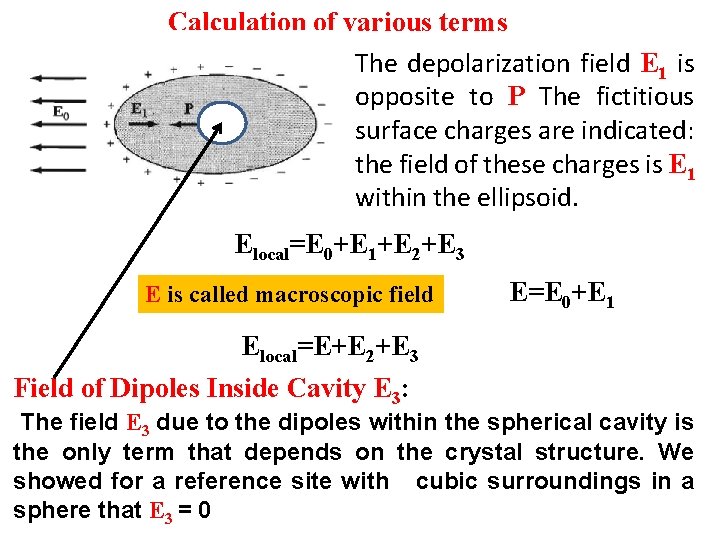
Calculation of various terms The depolarization field E 1 is opposite to P The fictitious surface charges are indicated: the field of these charges is E 1 within the ellipsoid. Elocal=E 0+E 1+E 2+E 3 E is called macroscopic field E=E 0+E 1 Elocal=E+E 2+E 3 Field of Dipoles Inside Cavity E 3: The field E 3 due to the dipoles within the spherical cavity is the only term that depends on the crystal structure. We showed for a reference site with cubic surroundings in a sphere that E 3 = 0
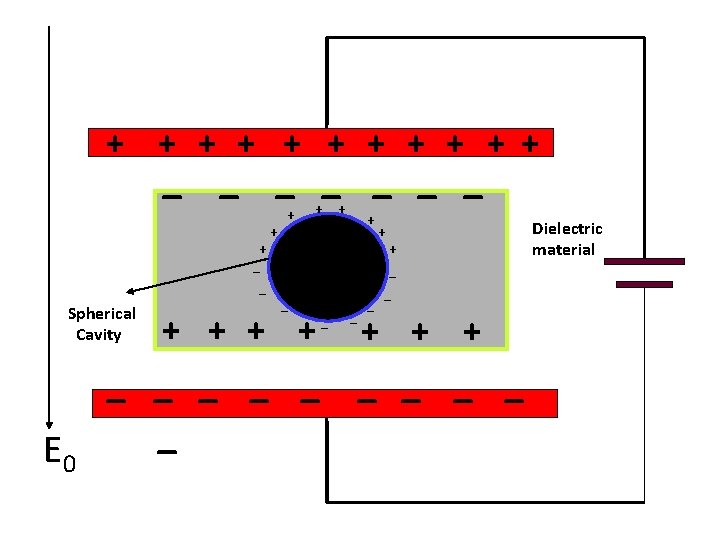
+ + + _ _ _ _ + _ _ Spherical Cavity E 0 + + + A _ + + _ _ + + + _ _ _ _ _ Dielectric material

Lorentz Field, E 2 The field E 2 due to the polarization charges on the surface of the fictitious cavity was calculated by Lorentz. If is the polar angle (referred to the polarization direction, the surface charge density on the surface of the cavity is -P cos . The electric field at the center of the spherical cavity of radius a is Calculation of the field in a spherical cavity in a uniformly polarized medium.

Elocal= E + E 2 + E 3 This is the Lorentz relation The polarizability of an atom is defined in terms of the local electric field at the atom: p = E local where p is the dipole moment. P = n p = n E local Clausius-Mossotti relation This relates the dielectric constant to the electronic polarizability, but only for crystal structures for which the Lorentz local field obtains.
Langevin-Debye formula polar molecules permanent dipole moment Material as a whole is neutral Alignment of dipole occurs in presence of applied field
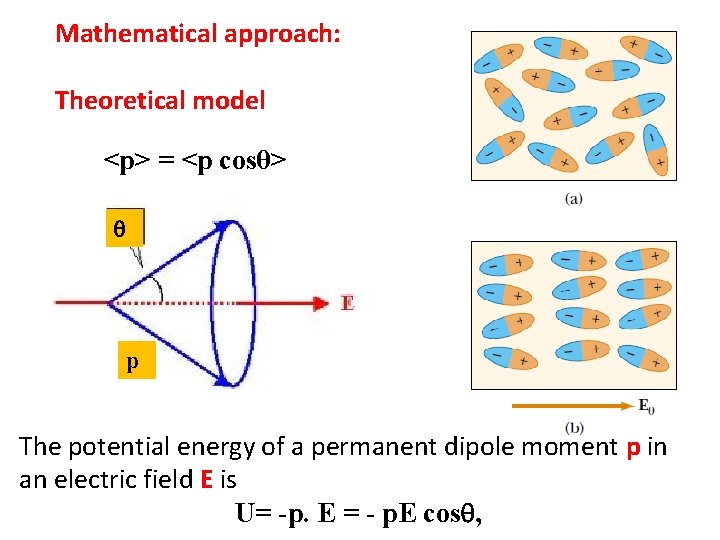
Mathematical approach: Theoretical model <p> = <p cosθ> p The potential energy of a permanent dipole moment p in an electric field E is U= -p. E = - p. E cos ,

The effective dipole moment of a molecular dipole is its' component along the field direction, i. e. , pcos. The distribution is Maxwell-Boltzmann’s distribution. Which gives the no. of dipoles per unit solid angle d = 2 sin d as exp (-U/k. BT) = exp (p. Ecos /k. BT) x = p. E /k. BT
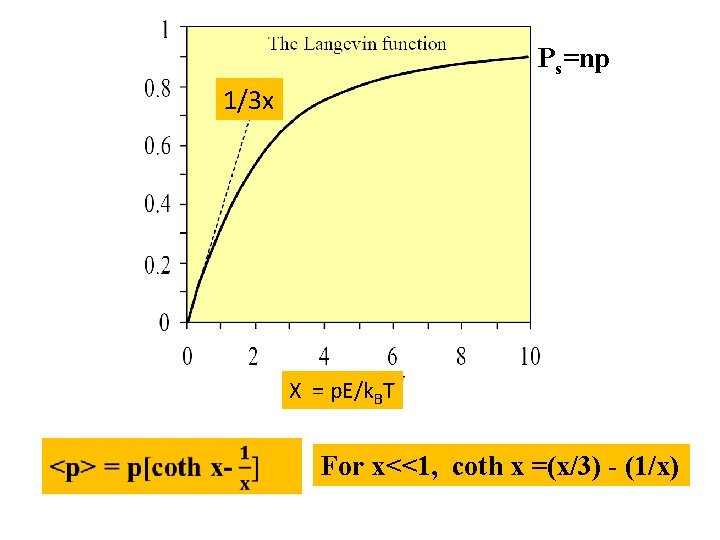
Ps=np 1/3 x X = p. E/k. BT For x<<1, coth x =(x/3) - (1/x)

<p> = p(x/3) = p 2 E/3 k. BT P/E= n=np 2 E/3 k. BT <p> =P/n= p 2 E/3 k. BT =p 2/3 k. BT This result has been derived by neglecting induced dipole moments, and represents. what we might call an orientational pblarizability. Induced dipoles effects, has been considered by Clausius-Mossotti relation say it is d, therefore total molecular polarizability is t = d + p 2/3 k. BT an expression which is known as the Langevin-Debye equation Thanks
- Slides: 13