Dielectric Materials Prerequisite Capacitive behavior Polarization Dielectric Loss

Dielectric Materials • Pre-requisite • Capacitive behavior • Polarization • Dielectric Loss • Insulating behavior • Dielectric Breakdown • Refractive Index • Piezoelectricity & Ferroelectricity EE 3406/NUS 79

Pre-requisite Gauss’s Law: Where: the integral refers to that over the whole of the surface enclosing the charge Qtotal and En is the electric field normal to a small area d. A on the closed surface. D is the displacement (surface charge density). EE 3406/NUS 80

Polarization • Microscopic View • Macroscopic View • Clausius-Mossotti Equation: The link between Microscopic and Macroscopic Views • Types of Polarizations EE 3406/NUS 81

Polarization vs Charge Flow • Dielectric materials typically have an energy gap of greater than 2. 5 e. V. • Insulating at room temperature • No need to be concerned with the transport of charge carriers. • Need to consider how the bound charges are polarized in the presence of an applied electric field EE 3406/NUS 82

Polarization: Microscopic View “polarization” process • Consider a negative and a positive charge separated by a distance a, as shown below. • We define the electric dipole moment, p, of a pair of equal charges as the product of the charge and the separation. Direction of vector p points from the negative to the positive charge. p=Qa • The separation of negative and positive charges resulting in an induced dipole is termed polarization. EE 3406/NUS 83
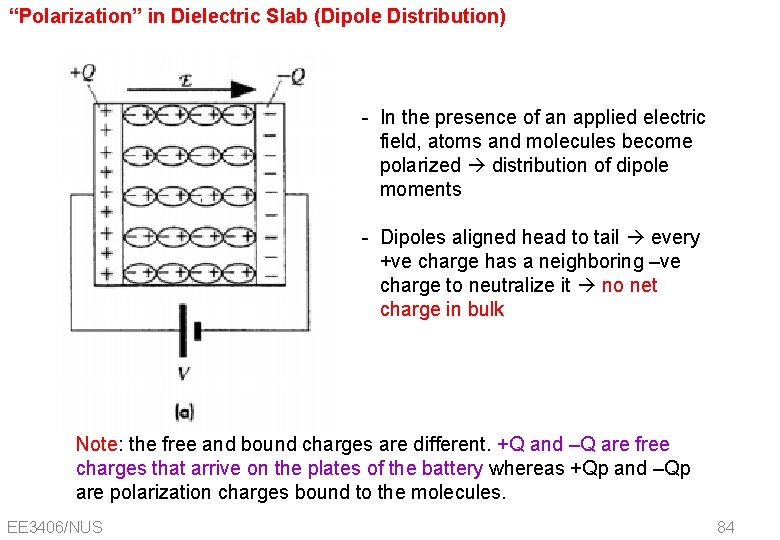
“Polarization” in Dielectric Slab (Dipole Distribution) - In the presence of an applied electric field, atoms and molecules become polarized distribution of dipole moments - Dipoles aligned head to tail every +ve charge has a neighboring –ve charge to neutralize it no net charge in bulk Note: the free and bound charges are different. +Q and –Q are free charges that arrive on the plates of the battery whereas +Qp and –Qp are polarization charges bound to the molecules. EE 3406/NUS 84

“Polarization” in Dielectric Slab (polarization “P”) - However, there is a net +Qp on RHS and –Qp on LHS face. These bound charges are a result of polarization and termed as surface polarization charges. - The polarization P is equal to the dipole moment per unit volume, given by (b) (c) where p 1, p 2, …, p. N are the average dipole moments induced at N molecules in the volume. - If pav is the average dipole moment per Notes: molecules, then an equivalent definition - Polarization “P” is a macroscopic parameter. of P is - dipole moments pi (i=1, 2…, N), pav are P=Npav microscopic parameters. - The equations link “macro-” and “micro-” where N is number of molecules per parameters. unit volume. EE 3406/NUS 85

“Polarization” in Dielectric Slab (Surface polarization charge density) - A large dipole between –Qp and +Qp separated with d, its total dipole moment ptotal is ptotal=Qpd - From the definition of the polarization “P”, we have P=(Qpd)/(Ad)=Qp/A (b) (c) Notes: - Polarization “P” (magnitude) equals “surface charge density” (charge per unit area) appearing on the surface of a dielectric slab EE 3406/NUS - Introducing σp as surface (polarization) charge density, we have P=σp - Direction of “P” is normal to the surface 86
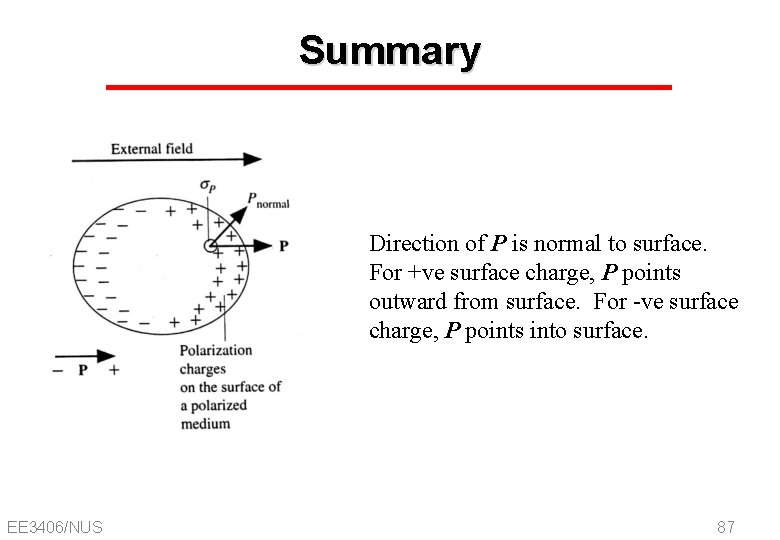
Summary Direction of P is normal to surface. For +ve surface charge, P points outward from surface. For -ve surface charge, P points into surface. EE 3406/NUS 87
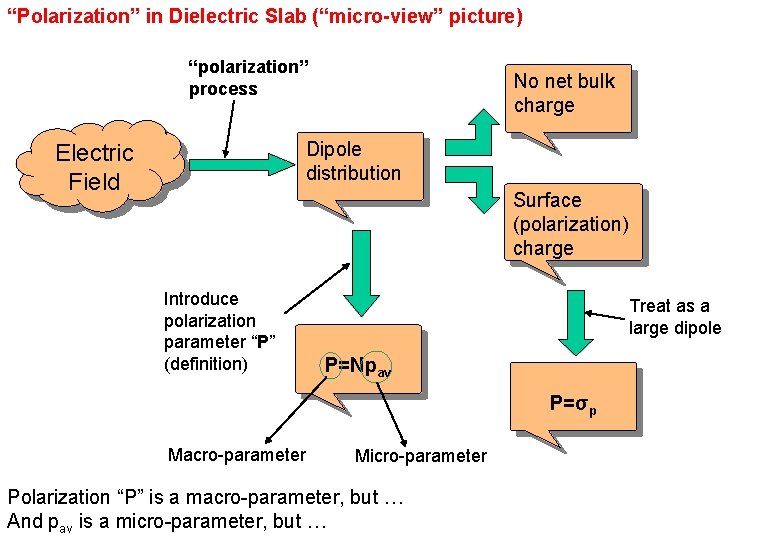
“Polarization” in Dielectric Slab (“micro-view” picture) “polarization” process No net bulk charge Dipole distribution Electric Field Surface (polarization) charge Introduce polarization parameter “P” (definition) Treat as a large dipole P=Npav P=σp Macro-parameter Micro-parameter Polarization “P” is a macro-parameter, but … And pav is a micro-parameter, but … EE 3406/NUS 88

Polarization: Macroscopic View Dielectric displacement (or surface charge density) - When vacuum is present between two parallel pates, the surface charge density (or dielectric displacement) D is given by D= 0 E - Hence at the macroscopic level, a dielectric is characterized by its permittivity which relates the surface charge density (or dielectric displacement D’) to the electric field via D’= E= 0 r. E is the product of the permittivity of free space 0 and the relative dielectric constant r. EE 3406/NUS 89

Dielectric constant r (macro parameter) - D’ can also be expressed as D’ = 0 r. E = 0 E+P = 0 E+ 0( r-1)E here, P is the increase in charge density above that for a vacuum, and its value equals to 0( r-1)E - The meaning of “P” here is “additional” charge density in dielectric compared to a vacuum case, thus has the same meaning as the polarization “P” (surface charge density). - To express the dependence of P on E, we define the dielectric susceptibility χ, by P= 0χE - Thus, we have χ= r-1 EE 3406/NUS or r=χ+1 90
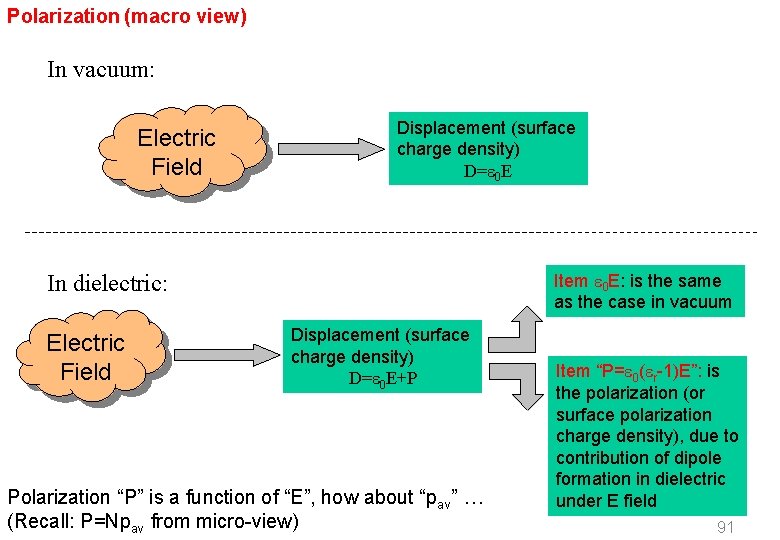
Polarization (macro view) In vacuum: Electric Field Displacement (surface charge density) D= 0 E Item 0 E: is the same as the case in vacuum In dielectric: Electric Field Displacement (surface charge density) D= 0 E+P Polarization “P” is a function of “E”, how about “pav” … (Recall: P=Npav from micro-view) EE 3406/NUS Item “P= 0( r-1)E”: is the polarization (or surface polarization charge density), due to contribution of dipole formation in dielectric under E field 91

Clausius-Mossotti Equation • Local (or Internal) Field: A re-visit of micro-view • Link between “macro-” and “micro-” • Clausius-Mossotti Equation EE 3406/NUS 92
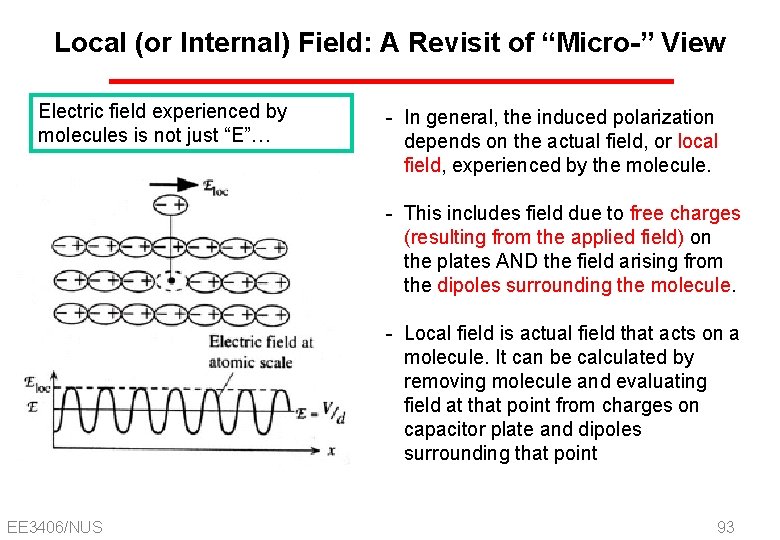
Local (or Internal) Field: A Revisit of “Micro-” View Electric field experienced by molecules is not just “E”… - In general, the induced polarization depends on the actual field, or local field, experienced by the molecule. - This includes field due to free charges (resulting from the applied field) on the plates AND the field arising from the dipoles surrounding the molecule. - Local field is actual field that acts on a molecule. It can be calculated by removing molecule and evaluating field at that point from charges on capacitor plate and dipoles surrounding that point EE 3406/NUS 93

Local (or Internal) Field: Some comments - For solids and many liquids where the number of atoms or molecules per unit volume is large, the local field is greater than the applied field. - The greater the polarization, the greater the local field because there are bigger dipoles around the point of interest. Thus Elocal depends on the arrangement of polarized molecules around the point and hence the crystal structure. - On the other hand, with dilute gases where the concentration of molecules or atoms is very low (surrounding dipoles are very sparse and far away), then the influence of other dipoles on the local electric field is negligible, and the local field is effectively the same as the applied field. How does the “local” field relate to applied field “E”? EE 3406/NUS 94

Relation of “Elocal” to “E” - The relation between the local field and applied field: “P” including contribution of “dipoles” in dielectric to local field where E is the applied field, P is the polarization induced by the applied field. (Note: the equation is derived by considering the simplest cubic crystal structure or a liquid). - Polarizability At low electric fields, we assume that the dipole moment p is proportional to the local electric field Elocal p=a. Elocal where a is a constant called polarizability which depends on the polarization mechanism of the material concerned. EE 3406/NUS 95
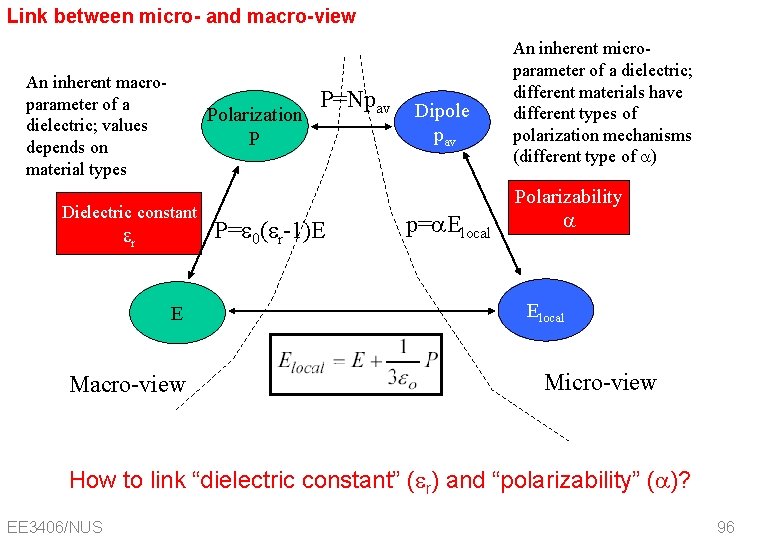
Link between micro- and macro-view An inherent macroparameter of a dielectric; values depends on material types Polarization P Dielectric constant r E Macro-view P=Npav P= 0( r-1)E Dipole pav p=a. Elocal An inherent microparameter of a dielectric; different materials have different types of polarization mechanisms (different type of a) Polarizability a Elocal Micro-view How to link “dielectric constant” ( r) and “polarizability” (a)? EE 3406/NUS 96

Clausius-Mossotti Equation - CM equation relates the dielectric constant ( r) and polarizability (a) directly - A higher a will cause a higher dielectric constant. DIY + P=Npav=Na. Elocal + P= 0( r-1)E EE 3406/NUS - Four different types of polarization mechanisms exist in dielectric Results in different dielectric constant Results in different capacitive behavior in response to frequency and temperature 97

Types of Polarization - Four main mechanisms for polarization (i) Electronic polarization (ii) Ionic polarization (iii) Orientational/dipolarization (iv) Interfacial polarization - Need to understand: (i) origin of polarization (ii) magnitude of polarization in various materials (iii) speed with which dipoles can align EE 3406/NUS 98
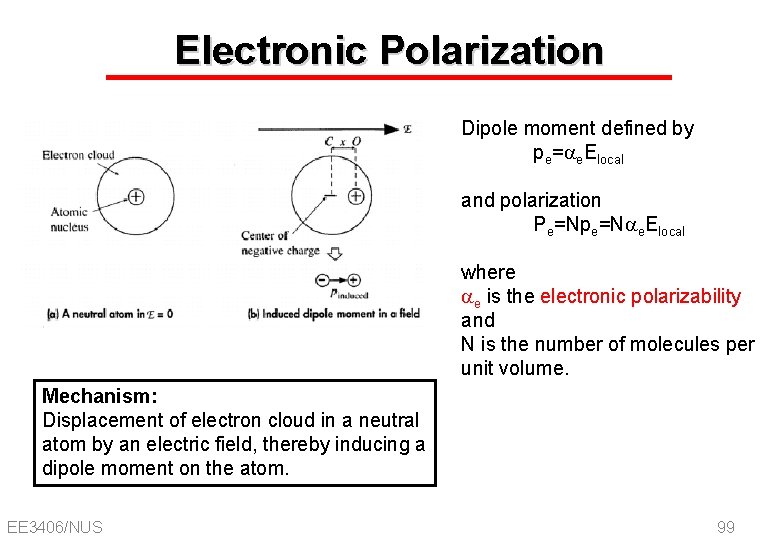
Electronic Polarization Dipole moment defined by pe=ae. Elocal and polarization Pe=Npe=Nae. Elocal where ae is the electronic polarizability and N is the number of molecules per unit volume. Mechanism: Displacement of electron cloud in a neutral atom by an electric field, thereby inducing a dipole moment on the atom. EE 3406/NUS 99

Properties of Electronic Polarization - Clausius Mossotti (CM) Equation of Electronic polarization - Type of Materials Since this form of polarization is atomic in nature, it is present in all materials regardless of type of bonding. When field is removed, polarization vanishes. - Temperature dependence of ae Electronic structure of an atom is independent of the temperature ae has no dependence on temperature. - Operational frequency Light electron cloud the response speed to the electric field is very fast. Typically up to ~1015 -1016 Hz. (optical frequency) EE 3406/NUS 100
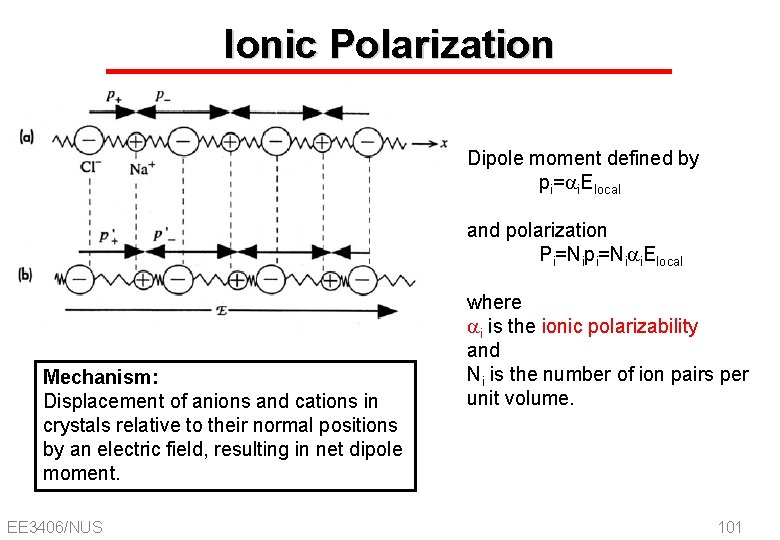
Ionic Polarization Dipole moment defined by pi=ai. Elocal and polarization Pi=Nipi=Niai. Elocal Mechanism: Displacement of anions and cations in crystals relative to their normal positions by an electric field, resulting in net dipole moment. EE 3406/NUS where ai is the ionic polarizability and Ni is the number of ion pairs per unit volume. 101

Properties of Ionic Polarization - Type of Materials Occurs predominantly in ionic materials. Like Na. Cl, Cs. Cl, … - Temperature dependence of ai None. - Operational frequency Since entire ions (much heavier than electrons) are being displaced, the mechanism only operates up to ~1012 – 1013 Hz (infra-red frequencies). - Clausius Mossotti equation also valids for (ai+ae) and r. EE 3406/NUS 102
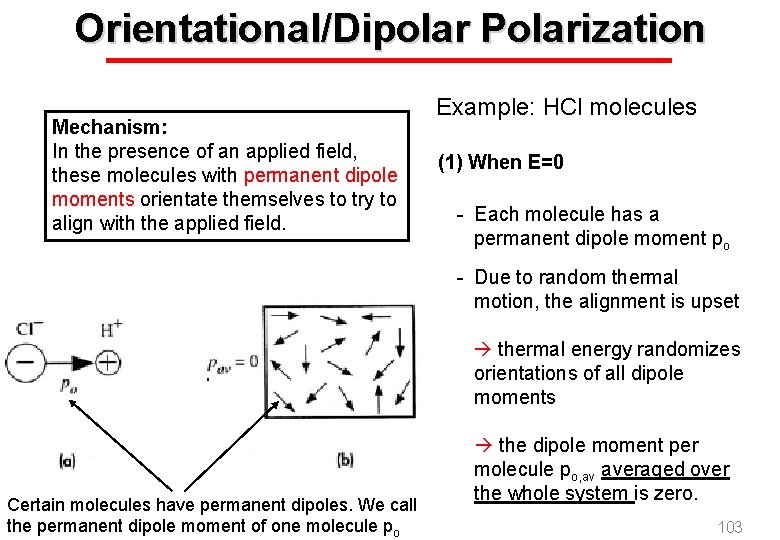
Orientational/Dipolar Polarization Mechanism: In the presence of an applied field, these molecules with permanent dipole moments orientate themselves to try to align with the applied field. Example: HCl molecules (1) When E=0 - Each molecule has a permanent dipole moment po - Due to random thermal motion, the alignment is upset thermal energy randomizes orientations of all dipole moments Certain molecules have permanent dipoles. We call the permanent dipole moment of one molecule po EE 3406/NUS the dipole moment per molecule po, av averaged over the whole system is zero. 103
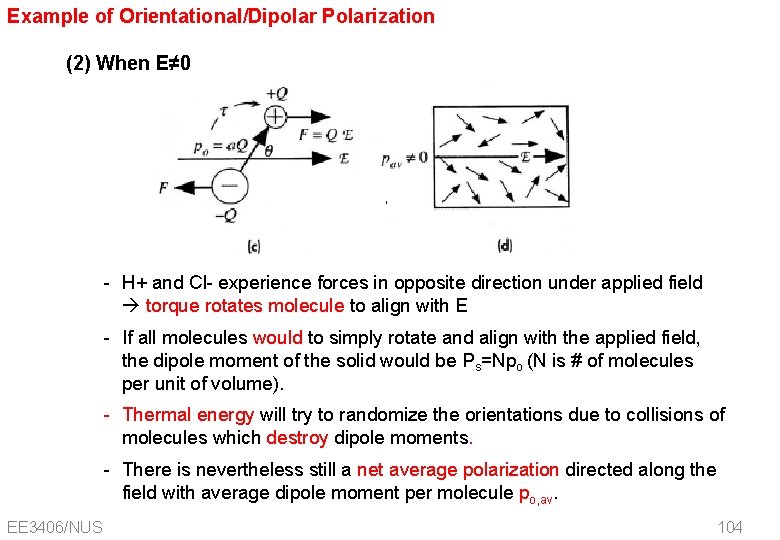
Example of Orientational/Dipolar Polarization (2) When E≠ 0 - H+ and Cl- experience forces in opposite direction under applied field torque rotates molecule to align with E - If all molecules would to simply rotate and align with the applied field, the dipole moment of the solid would be Ps=Npo (N is # of molecules per unit of volume). - Thermal energy will try to randomize the orientations due to collisions of molecules which destroy dipole moments. - There is nevertheless still a net average polarization directed along the field with average dipole moment per molecule po, av. EE 3406/NUS 104
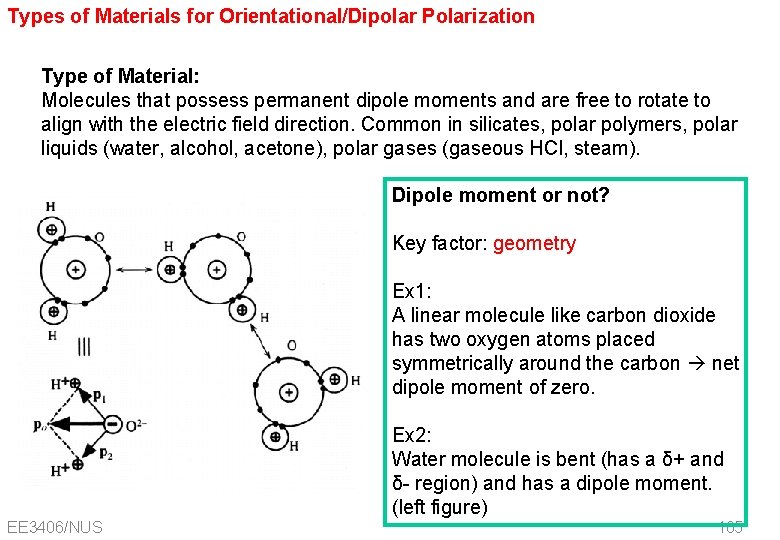
Types of Materials for Orientational/Dipolar Polarization Type of Material: Molecules that possess permanent dipole moments and are free to rotate to align with the electric field direction. Common in silicates, polar polymers, polar liquids (water, alcohol, acetone), polar gases (gaseous HCl, steam). Dipole moment or not? Key factor: geometry Ex 1: A linear molecule like carbon dioxide has two oxygen atoms placed symmetrically around the carbon net dipole moment of zero. Ex 2: Water molecule is bent (has a δ+ and δ- region) and has a dipole moment. (left figure) EE 3406/NUS 105

Properties of Orientational/Dipolar Polarization Temperature dependence of ao: - While dipole moments are trying to line up with the applied field, they are jostled by their thermal motion and not all of them succeed in lining up properly. - Assuming Boltzmann statistics, the average dipole moment per molecule is given (without proof) - Thus, the orientational polarizability given by - Hence the polarization due to orientational polarization EE 3406/NUS 106

Properties of Orientational/Dipolar Polarization - We see that polarization due to orientational polarization Po is inversely proportional to the absolute temperature. (1) Unlike electronic and ionic polarization, orientational polarization is strongly dependent on temperature (2) Orientational polarization can be retained after the field is removed. (3) ao decreases with increasing temperature r also decreases with increasing temperature. Operational frequency: Involves of rotation of whole molecules (or molecular groups) to align with the electric field, therefore only occurs at lower frequencies of ~1011 -1012 Hz (sub-infrared frequencies) EE 3406/NUS 107
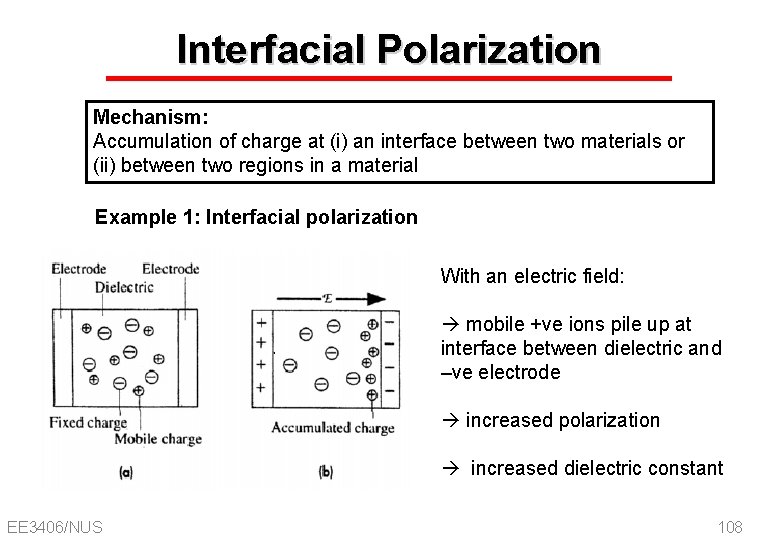
Interfacial Polarization Mechanism: Accumulation of charge at (i) an interface between two materials or (ii) between two regions in a material Example 1: Interfacial polarization With an electric field: mobile +ve ions pile up at interface between dielectric and –ve electrode increased polarization increased dielectric constant EE 3406/NUS 108
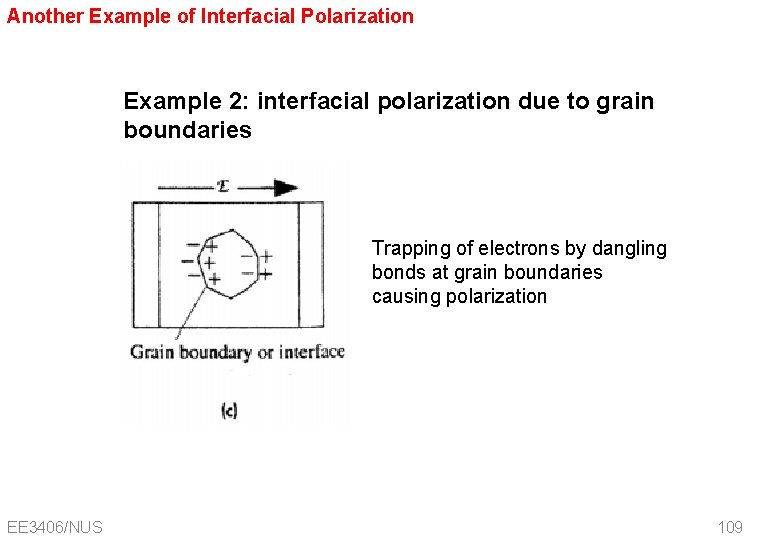
Another Example of Interfacial Polarization Example 2: interfacial polarization due to grain boundaries Trapping of electrons by dangling bonds at grain boundaries causing polarization EE 3406/NUS 109

Properties of Interfacial Polarization Type of Materials: Occurs predominantly at interfaces and in materials with defects, grain or phase boundaries, impurities. Operational frequency Occurs at even lower frequencies of ~10 -3 to 103 Hz. CM Equation is always not valid for interfacial polarization Interfacial polarization cannot be considered as a straightforward contribution because it occurs at interfaces (locally) and cannot be put into an average polarization per molecule in the bulk. EE 3406/NUS 110

Combined Polarization - The total polarization of a material is the sum of contributions from all components. - In general, the average polarization due to the electronic, ionic and orientational/dipolar components is - The dielectric constant under electronic, ionic and orientational polarization is given by the combined Clausius Mossotti equation. EE 3406/NUS 111

Some discussions on “Combined Polarization” - Not all atoms or molecules posses each of the above types of polarizability (i) All display electronic polarizability, since this aries from polarization of atoms. (ii) Many materials have ionic polarizations (exceptions are elements, eg. Ar, Ne, He, and molecules of the same atomic species, eg. H 2, O 2, Si). (iii) Those with permanent dipole moments have orientational/dipolarization. - Several factors determine the extent with which each dielectric is affected by each of the polarization mechanism: (1) Atomic scale structure (2) Type of bonding (3) Type of atoms (4) Frequency of applied electromagnetic field. EE 3406/NUS 112
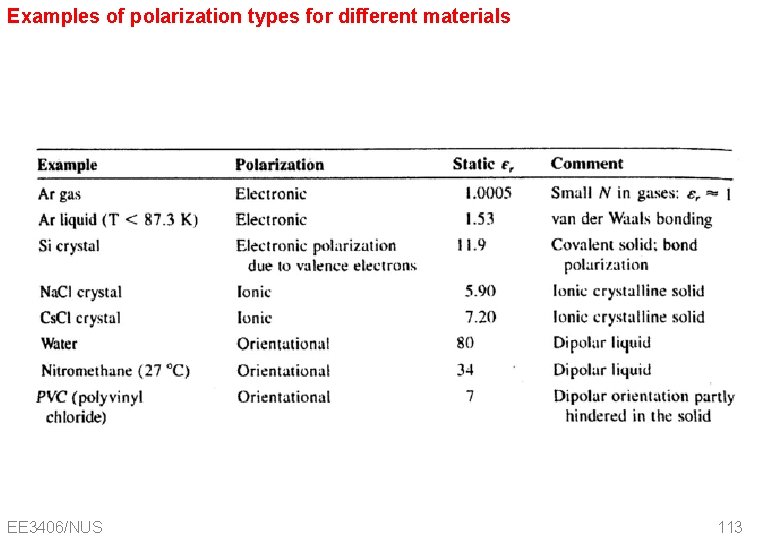
Examples of polarization types for different materials EE 3406/NUS 113
- Slides: 35