DIBORANE Methods Of Preparetion It is prepared by
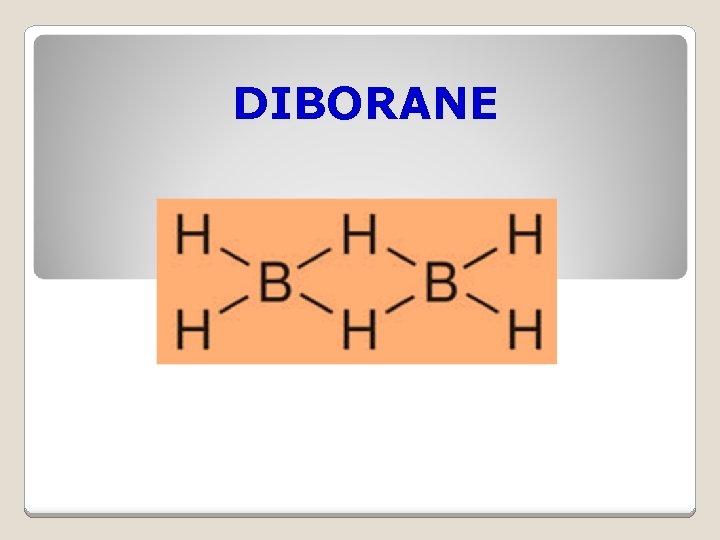
DIBORANE

Methods Of Preparetion It is prepared by treating boron Trifluoride with Li. Al. H 4 in diethyl ether 4 BF 3 + 3 Li. Al. H 4 2 B 2 H 6 +3 Li. F + 3 ALF 3 In a laboratory the diborane is prepared by the oxidation of sodium borobhydride with iodine 2 Na. BH 4 + I 2 B 2 H 6 +2 Na. I +H 2 In industry it is prepared by the reaction of BF 3 with sodium hydride 450 K 2 BF 3 + 6 Na. H B 2 H 6 + 6 Na. F

Properties 1) Diborane is a colorless, highly toxic gas. It has boiling point ok 180 K 2) Diborane catches fire spontaneously when it is exposed to atmospheric air it burns with oxygen. The reaction is exothermic releasing a lager amount of energy B 2 H 6 + 3 O 2 B 2 O 3 + 3 H 2 O ΔH= - 1976 K J mol-1 3) it is readily hydrolyzed by water to form boric acid B 2 H 6 + 6 H 2 O 2 B(OH)3 + 6 H 2
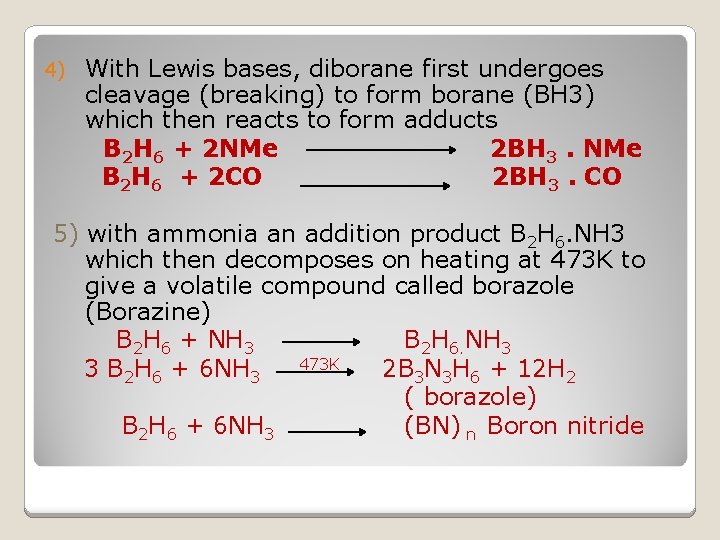
4) With Lewis bases, diborane first undergoes cleavage (breaking) to form borane (BH 3) which then reacts to form adducts B 2 H 6 + 2 NMe 2 BH 3. NMe B 2 H 6 + 2 CO 2 BH 3. CO 5) with ammonia an addition product B 2 H 6. NH 3 which then decomposes on heating at 473 K to give a volatile compound called borazole (Borazine) B 2 H 6 + NH 3 B 2 H 6. NH 3 473 K 3 B 2 H 6 + 6 NH 3 2 B 3 N 3 H 6 + 12 H 2 ( borazole) B 2 H 6 + 6 NH 3 (BN) n Boron nitride
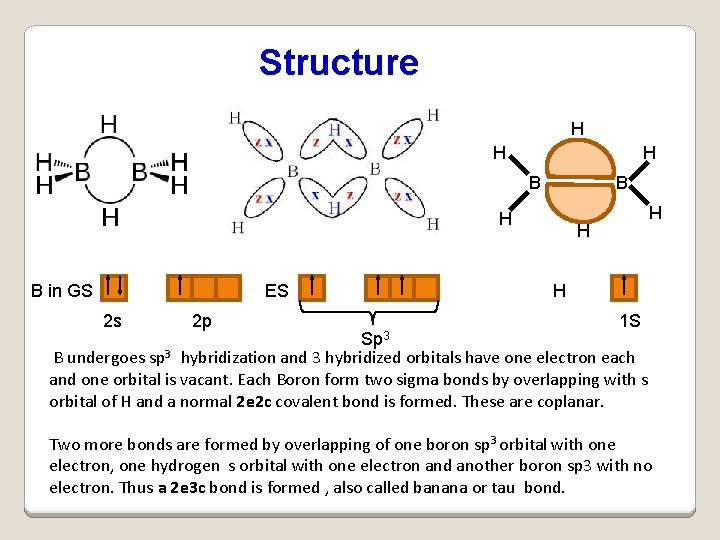
Structure H H H B in GS ES 2 s 2 p H B H H Sp 3 1 S B undergoes sp 3 hybridization and 3 hybridized orbitals have one electron each and one orbital is vacant. Each Boron form two sigma bonds by overlapping with s orbital of H and a normal 2 e 2 c covalent bond is formed. These are coplanar. Two more bonds are formed by overlapping of one boron sp 3 orbital with one electron, one hydrogen s orbital with one electron and another boron sp 3 with no electron. Thus a 2 e 3 c bond is formed , also called banana or tau bond.

Structure of Higher Boranes Types of Bonds in Boranes 1. Normal covalent bonds -2 c-2 e- B-H 2. Normal covalent bonds - 2 c-2 e- B-B 3. Bridge bonds -3 c-2 e- B-H-B 4. Bridge bonds - 3 c-2 e- B-B-B 5. Closed bridge bonds - 3 c-2 e. B B B
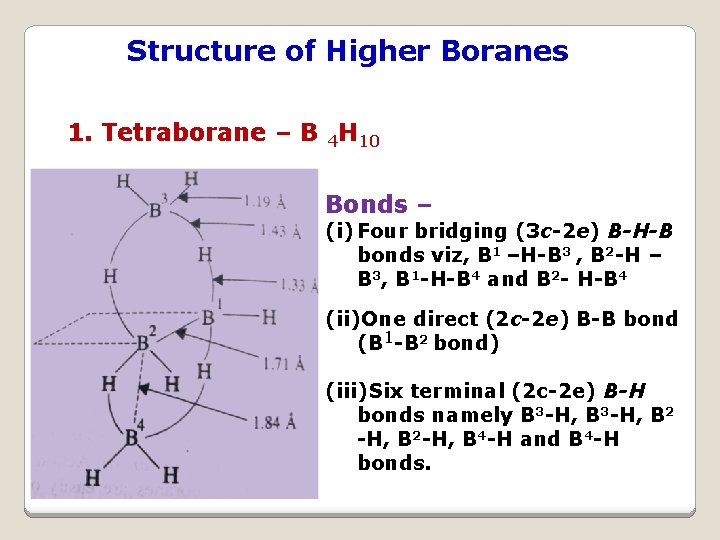
Structure of Higher Boranes 1. Tetraborane – B 4 H 10 Bonds – (i) Four bridging (3 c-2 e) B-H-B bonds viz, B 1 –H-B 3 , B 2 -H – B 3, B 1 -H-B 4 and B 2 - H-B 4 (ii)One direct (2 c-2 e) B-B bond (B 1 -B 2 bond) (iii)Six terminal (2 c-2 e) B-H bonds namely B 3 -H, B 2 -H, B 4 -H and B 4 -H bonds.
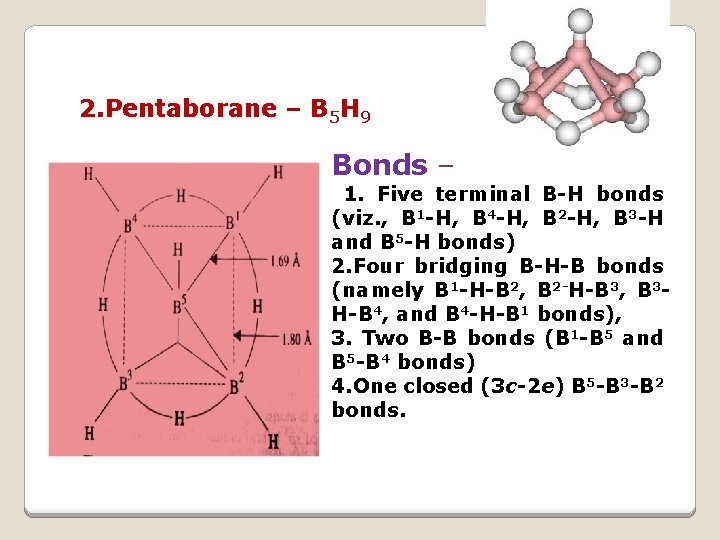
2. Pentaborane – B 5 H 9 Bonds – 1. Five terminal B-H bonds (viz. , B 1 -H, B 4 -H, B 2 -H, B 3 -H and B 5 -H bonds) 2. Four bridging B-H-B bonds (namely B 1 -H-B 2, B 2 -H-B 3, B 3 H-B 4, and B 4 -H-B 1 bonds), 3. Two B-B bonds (B 1 -B 5 and B 5 -B 4 bonds) 4. One closed (3 c-2 e) B 5 -B 3 -B 2 bonds.
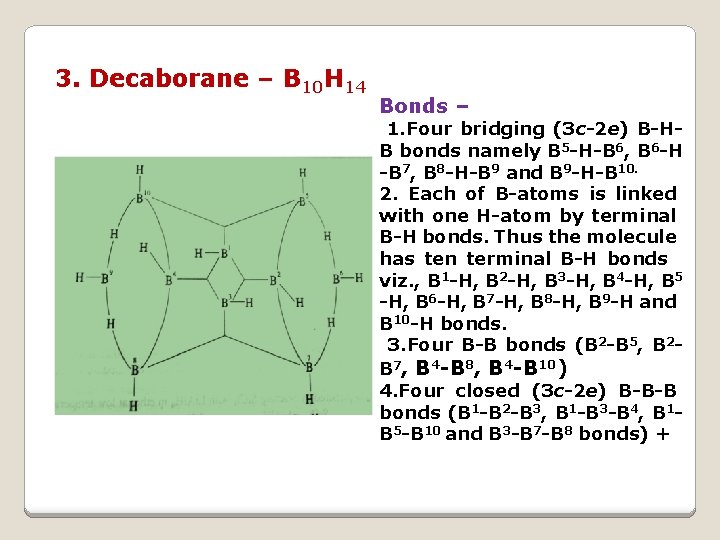
3. Decaborane – B 10 H 14 Bonds – 1. Four bridging (3 c-2 e) B-HB bonds namely B 5 -H-B 6, B 6 -H -B 7, B 8 -H-B 9 and B 9 -H-B 10. 2. Each of B-atoms is linked with one H-atom by terminal B-H bonds. Thus the molecule has ten terminal B-H bonds viz. , B 1 -H, B 2 -H, B 3 -H, B 4 -H, B 5 -H, B 6 -H, B 7 -H, B 8 -H, B 9 -H and B 10 -H bonds. 3. Four B-B bonds (B 2 -B 5, B 2 B 7, B 4 -B 8, B 4 -B 10) 4. Four closed (3 c-2 e) B-B-B bonds (B 1 -B 2 -B 3, B 1 -B 3 -B 4, B 1 B 5 -B 10 and B 3 -B 7 -B 8 bonds) +
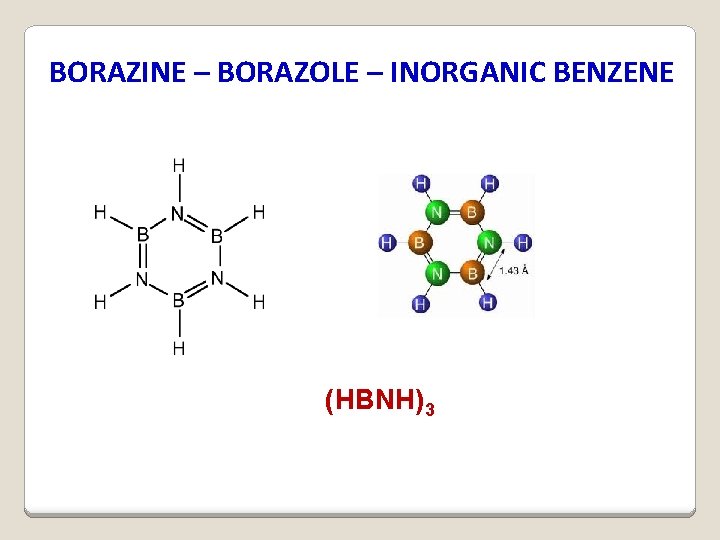
BORAZINE – BORAZOLE – INORGANIC BENZENE (HBNH)3

PREPARATION
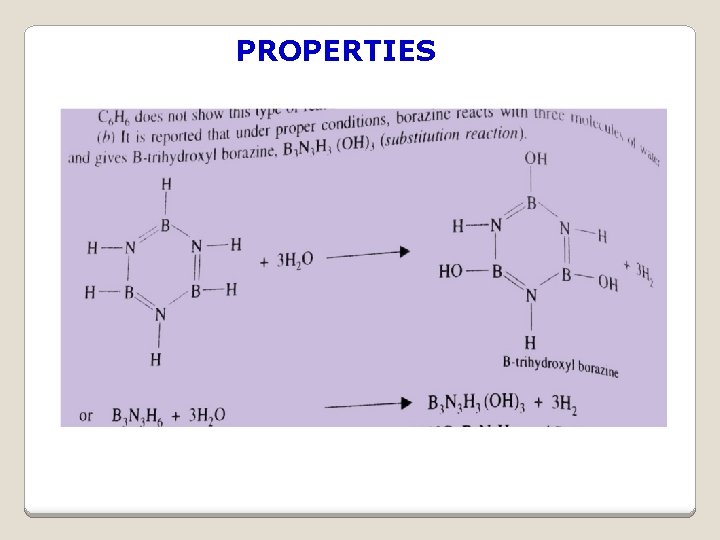
PROPERTIES





- Slides: 17