Diamondlike carbon DLC films Amorphous Carbon Film Mixture


Diamond-like carbon (DLC) films • Amorphous Carbon Film • Mixture of sp 1, sp 2 and sp 3 Hybridized Bonds • High Content of Hydrogen (20 -60%) • Synonyms – Diamond-like Carbon – (Hydrogenated) amorphous carbon (a-C: H) – i-Carbon – Tetrahedral Amorphous Carbon (ta-C) sp 3 ta-C: H ta-C DAC sp 2 GA C PAC No film H

Diamond-like carbon (DLC) films • High hardness • High wear resistance • Low friction coefficient • Optical transparency • Chemical inertness • Smooth surface • Bio-compatibility Hard disk Video Head Drum • Protective coating • Bio materials Coronary Artery Stent Hip Joint
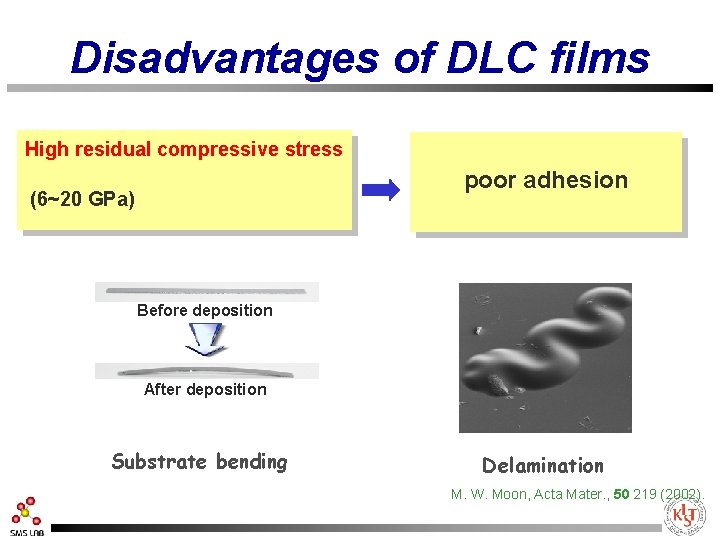
Disadvantages of DLC films High residual compressive stress poor adhesion (6~20 GPa) Hard disk Before deposition After deposition Substrate bending Delamination M. W. Moon, Acta Mater. , 50 219 (2002).
Structure and property relationship H a rd n e s s Substrate biasing Post-annealing Metal incorporation ; Ti, W, Mo, Cr, Al….
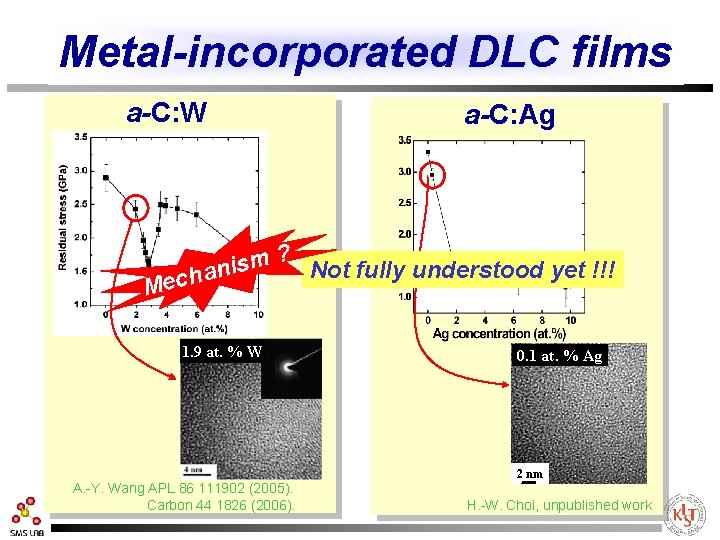
Metal-incorporated DLC films a-C: W a-C: Ag ? m s i Not fully understood yet !!! chan Me 1. 9 at. % W A. -Y. Wang APL 86 111902 (2005). Carbon 44 1826 (2006). 0. 1 at. % Ag 2 nm H. -W. Choi, unpublished work
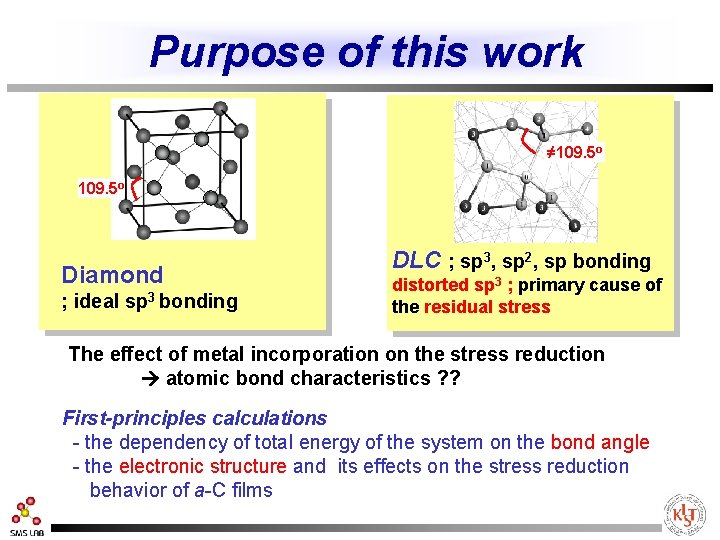
Purpose of this work ≠ 109. 5 o Diamond ; ideal sp 3 bonding DLC ; sp 3, sp 2, sp bonding distorted sp 3 ; primary cause of the residual stress The effect of metal incorporation on the stress reduction atomic bond characteristics ? ? First-principles calculations - the dependency of total energy of the system on the bond angle - the electronic structure and its effects on the stress reduction behavior of a-C films
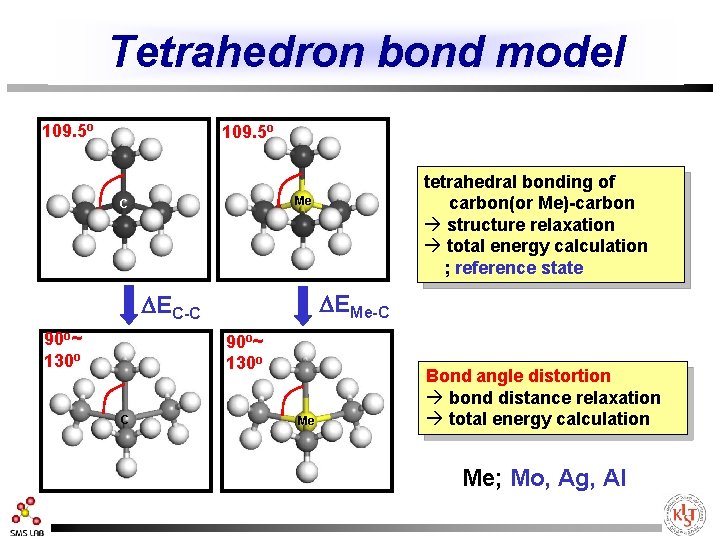
Tetrahedron bond model 109. 5 o Me C DEMe-C DEC-C 90 o~ 130 o C tetrahedral bonding of carbon(or Me)-carbon à structure relaxation à total energy calculation ; reference state Me Bond angle distortion à bond distance relaxation à total energy calculation Me; Mo, Ag, Al

Calculation condition by VASP § DFT scheme § Ecut = 550 e. V § Exchange-correlation potential; GGA (PBE) § Projector Augmented-Wave (PAW) potential § Gaussian smearing factor = 0. 05 e. V § Spin-unrestricted calculations § Convergence = 10 -5 e. V § Ionic relaxation; CG method (force < 0. 01 e. V/Å) § Gamma point calculation (15 x 15 Å3)
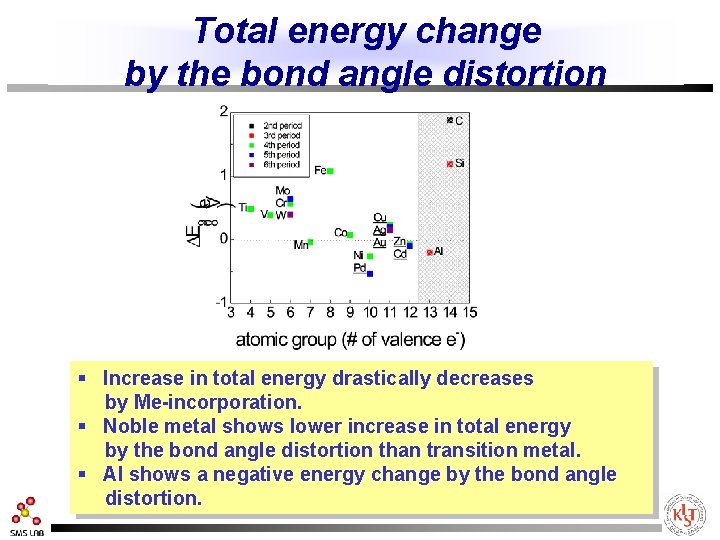
Total energy change by the bond angle distortion § Increase in total energy drastically decreases by Me-incorporation. § Noble metal shows lower increase in total energy by the bond angle distortion than transition metal. § Al shows a negative energy change by the bond angle distortion.
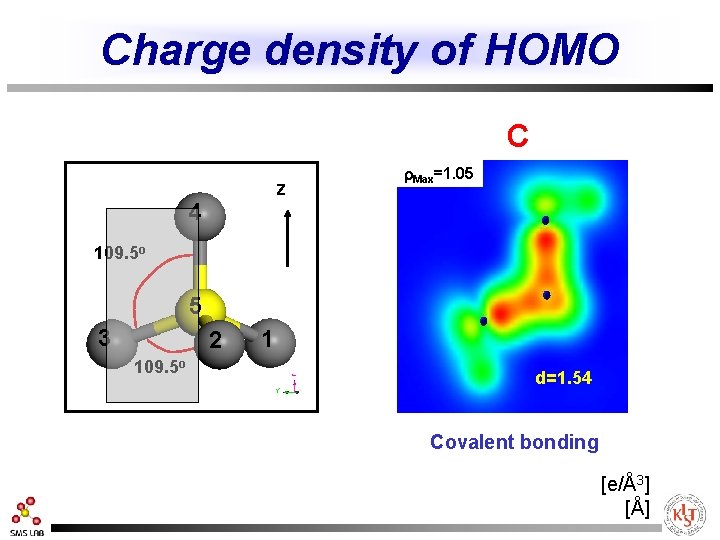
Charge density of HOMO C z 4 r. Max=1. 05 109. 5 o 5 3 2 109. 5 o 1 d=1. 54 Covalent bonding [e/Å3] [Å]
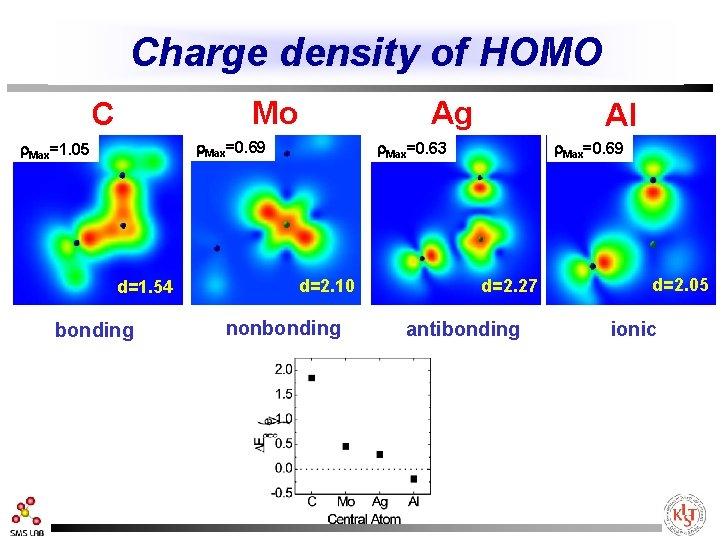
Charge density of HOMO Mo C Ag r. Max=0. 69 r. Max=1. 05 d=1. 54 bonding Al r. Max=0. 63 d=2. 10 nonbonding r. Max=0. 69 d=2. 27 antibonding d=2. 05 ionic
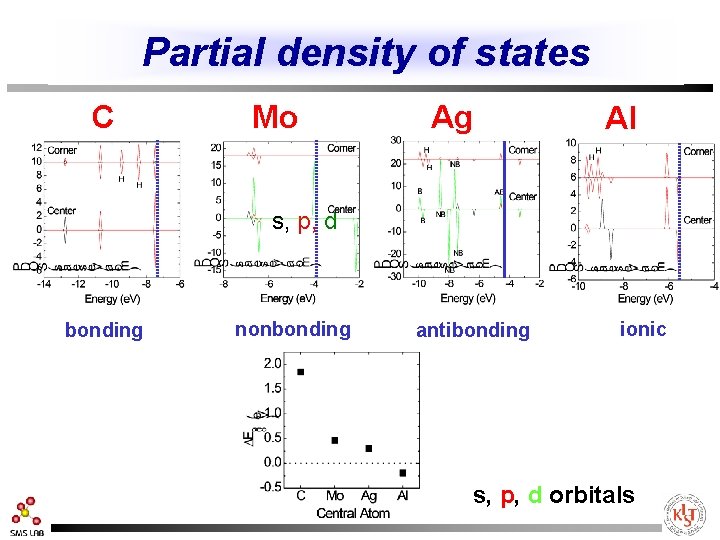
Partial density of states C Mo Ag Al s, p, d bonding nonbonding antibonding ionic s, p, d orbitals
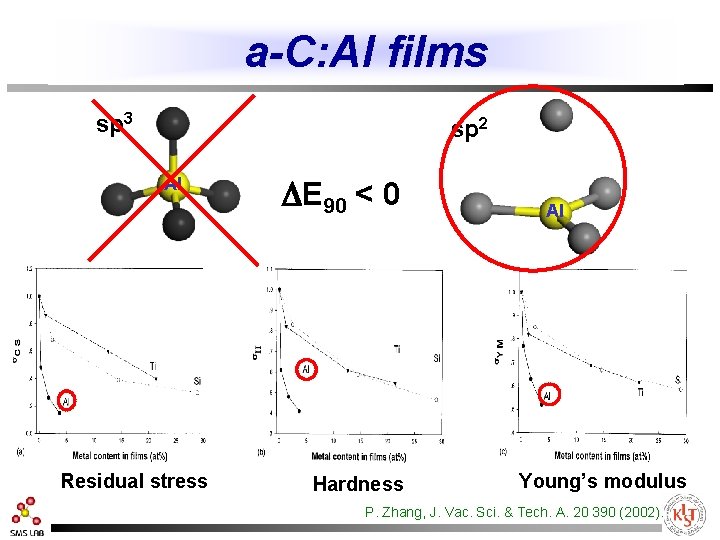
a-C: Al films sp 3 sp 2 Al Residual stress DE 90 < 0 Hardness Al Young’s modulus P. Zhang, J. Vac. Sci. & Tech. A. 20 390 (2002).
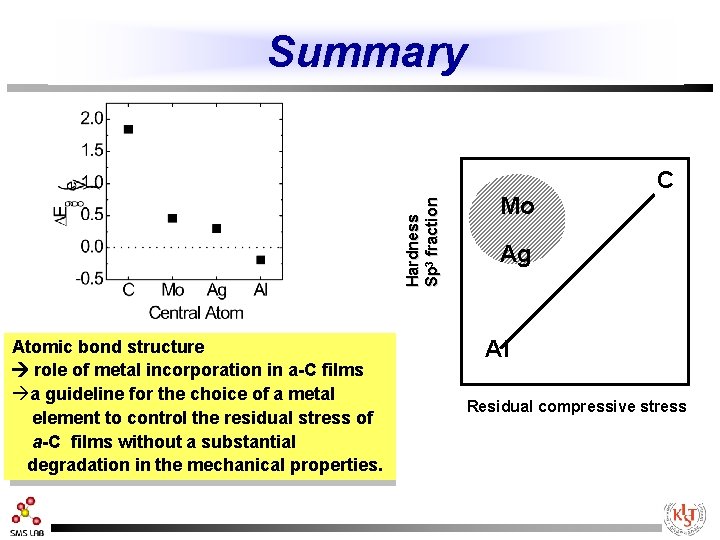
Summary C Mo r. Max=1. 05 r. Max=0. 69 Hard n ess S p 3 fr a c ti o n Mo Ag d=2. 10 d=1. 54 Al Ag Atomic bond structure role of metal incorporation in a-C films àa guideline for the choice of a metal element to control the residual stress of a-C films without a substantial degradation in the mechanical properties. C r. Max=0. 63 Al r. Max=0. 69 Residual compressive stress d=2. 27 d=2. 05
- Slides: 15