DIAGNOSIS AND MANAGEMENT OF GUILLAIN BARRE SYNDROME AN

DIAGNOSIS AND MANAGEMENT OF GUILLAIN –BARRE SYNDROME – AN UPDATE 1 Prof. Sumana Kanjilal Department of Pediatrics IPGMER & SSKM Hospital

Guillain-Barré syndrome (GBS) is an autoimmune disorder that is thought to be a post infectious polyneuropathy, involving mainly motor but also sensory and sometimes autonomic nerves. Incidence : 0. 6 -4 per 100000 population. Affects people of all ages – mean age 40 years. In paediatric population 0. 34 -1. 34 per 100000. Slightly male preponderance. 2

TYPES OF PRESENTATION Acute onset CIDP 3

SUBTYPES Classic variety: - Acute inflammatory demyelinating polyradiculoneuropathy (AIDP) - Acute motor axonal neuropathy (AMAN) - Acute motor sensory axonal neuropathy (AMSAN) Variants of GBS: 4
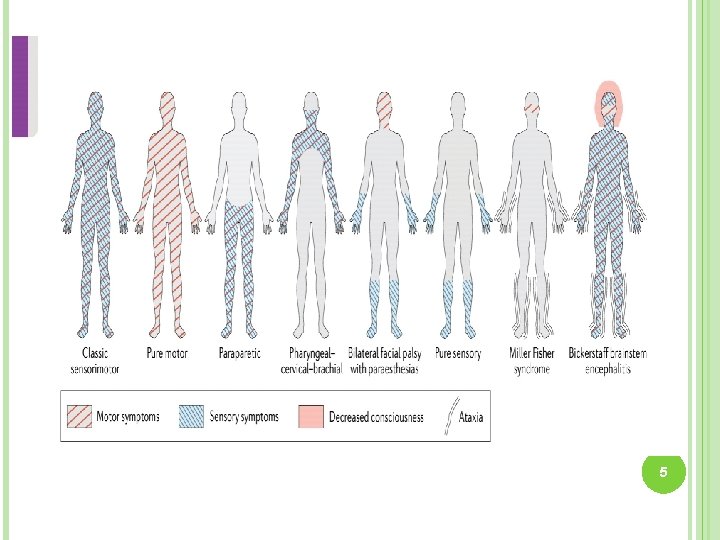
5
6

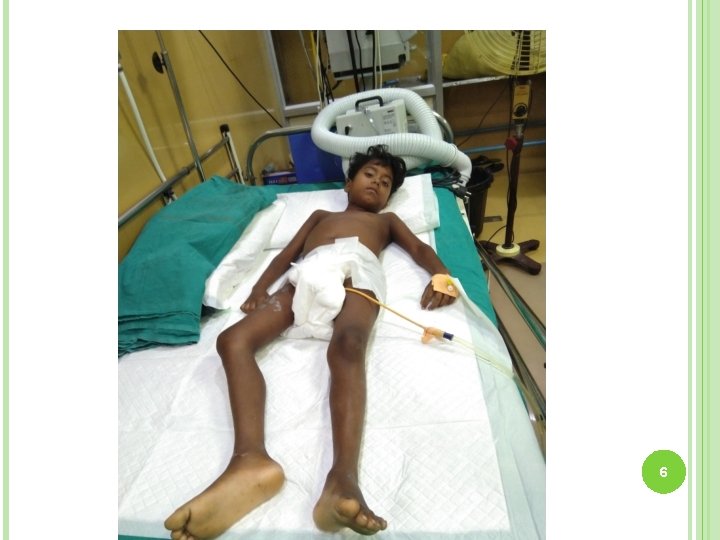
PRESENTING COMPLAINTS A nine-year old female child presented with- Progressive weakness of 1. Bilateral lower limbs followed by upper limbs 2. Sudden onset after waking up in the morning 3. Gradually progressed upwards reaches maximum within 24 hrs. Retention of urine which developed after 3 days of admission

It was not associated with: Fever, headache, vomiting, photophobia Loss of consciousness Any tingling, numbness Difficulty in swallowing or difficulty in speech Any neck pain or stiffness or visual problems No complaint of deviation of mouth to any side

Past history No significant past medical or surgical history was present. No recent past history of upper respiratory infection or loose motion. Family history Birth history Developmental history No delay Immunization history Up to date. No history of recent immunization.

ON EXAMINATION • Patient was conscious, alert, cooperative • Mild pallor • Vitals: PR- 68/min BP- 100/64 (between 50 P and 90 P) RR- 26/min Temperature- not raised Sp. O 2 – 98% at room air

CNS EXAMINATION Mental status- Alert & co-operative. Thought process coherent. Oriented to person, place and time. Cranial nerves- Intact. No neck stiffness Motor system Upper limbs Lower limbs Tone Power Deep Reflexes Plantar Sensation Decreased Grade 2 Absent Intact Decreased Grade 1 Absent Flexor Intact
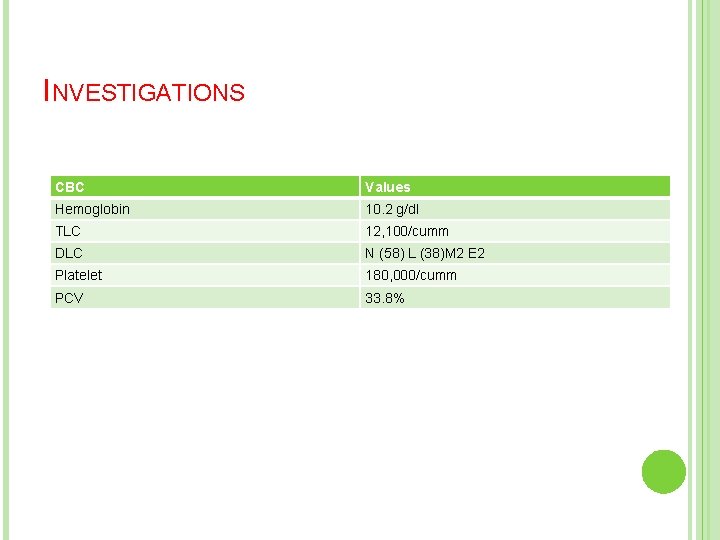
INVESTIGATIONS CBC Values Hemoglobin 10. 2 g/dl TLC 12, 100/cumm DLC N (58) L (38)M 2 E 2 Platelet 180, 000/cumm PCV 33. 8%

Renal function tests: Urea - 27 mg/ dl Creatinine - 0. 5 mg/ dl Serum sodium - 135 mmol/ L Serum potassium - 3. 4 mmol/ L CRP - <0. 314 mg /d. L LFT – WNL

CSF Study Physical appearance- colorless, No coagulum Microscopy – Cell count- 8 cells/ml, Cell type- mononuclear Biochemistry – Protein – 230 mg/dl, Sugar- 76 mg/dl Nerve conduction study NCS revealed electrophysiological features consistent with demyelination distal motor neuropathy The associated clinical presentation intermingled with this finding might suggest AIDP

OTHER INVESTIGATIONS MRI Brain & Whole Spine- NMO with MOG antibody profile. Anti- Aquaporin 4 Ig. G MOG antibodies

HOW TO DIAGNOSE GBS Preceding events: Infections- Six pathogens are associated with GBS: Campylobactor jejuni, Mycoplasma pneumoniae, CMV, Zika EB virus & Hepatitis –A Vaccine- Influenza vaccine, Conjugated meningococcal vaccine Drugs. Immunobiologicals e. g, TNF-antagonist, Type I interferon etc. Clinical history and examination 16

INVESTIGATIONS Lab tests: - CBC, CBG, S, electrolytes, CRP kidney function tests, liver enzymes - Anti-ganglioside antibodies- diagnostic value is limited, Anti- GQ 1 b antibodies sre found in 90% of patients with Miller Fisher Syndrome 17

CSF EXAMINATION: -Classic finding is albumin-cytologic dissociation. - Protein levels are normal in 30 -50% of patients in the 1 st week and in 10 -30% of patients in the 2 nd week after disease onset. - Mild pleocytosis (10 -50 cell/μl)- consider infectious causes of polyradiculopathy. - Marked pleocytosis (>50 cells/μl)- suggests other pathologies e. g, leptomeningeal malignancy or infectious or inflammatory diseases of spinal cord or nerve root. 18

OTHER TESTS Electrophysiological studies-Helpful in patients with atypical presentation -To differentiate between 3 subtypes of classical GBS i. e, AIDP, AMAN and AMSAN - May be normal; a. When performed within 1 week of symptom onset b. In patients with initial proximal weakness c. Mild disease d. Slow progression Or e. Clinical variants 19
MRI – BRAIN & SPINAL CORD Not a routine test Presence of nerve root enhancement on gladolinium enhanced MRI is non-specific but sensitive feature of GBS in young children. Helpful in differential diagnosis of acute flaccid myelitis and others like -Brainstem infections or inflammation - Stroke - Nerve root compression - Leptomeningeal malignancy - Spinal cord or anterior horn cell inflammation 20

DIAGNOSTIC CRITERIA FOR GB SYNDROME Features required for diagnosis- Progressive bilateral weakness of arms & legs Absent or ↓ tendon reflexes in affected limbs Features that strongly support diagnosis- Progressive phase lasts from days to 4 weeks (usually < 2 wks) Relative symmetry of symptoms & signs Relatively mild sensory symptoms & signs (absent in pure motor variant) 21

Cranial nerve involvement, Specially bilateral facial palsy Autonomic dysfunction Muscular or radicular back or limb pain Increased protein level in CSF; normal protein level do not rule out the diagnosis Electrodiagnostic features of motor or sensorymotor neuropathy Ref: National Institute of Neurological Disorders & Stroke (NINDS) criteria 22

FEATURES THAT CAST DOUBT ON DIAGNOSIS Increased number of mononuclear or polynuclear cells in CSF (>50/μl) Marked, persistent asymmetry of weakness Bladder or bowel dysfunction at onset or persistent during disease course Severe respiratory dysfunction with limited limb weakness at onset Sensory signs with limited weakness at onset Fever at onset Nadir <24 hrs Sharp sensory level indicating spinal cord injury 23

CONTD…. Hyperreflexia or clonus Extensor plantar responses Abdominal pain Slow progression with limited weakness without respiratory involvement Continued progression for >4 weeks after start of symptoms Alterations of consciousness (except in Bickerstaff brainstem encephalitis) 24
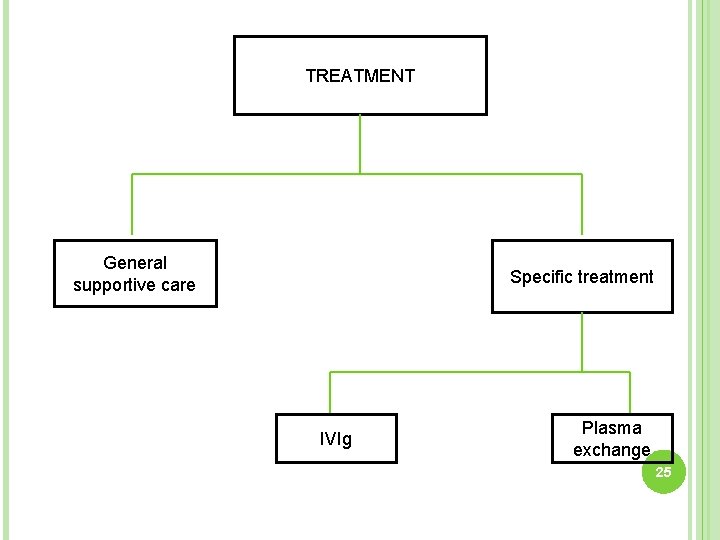
TREATMENT General supportive care Specific treatment IVIg Plasma exchange 25
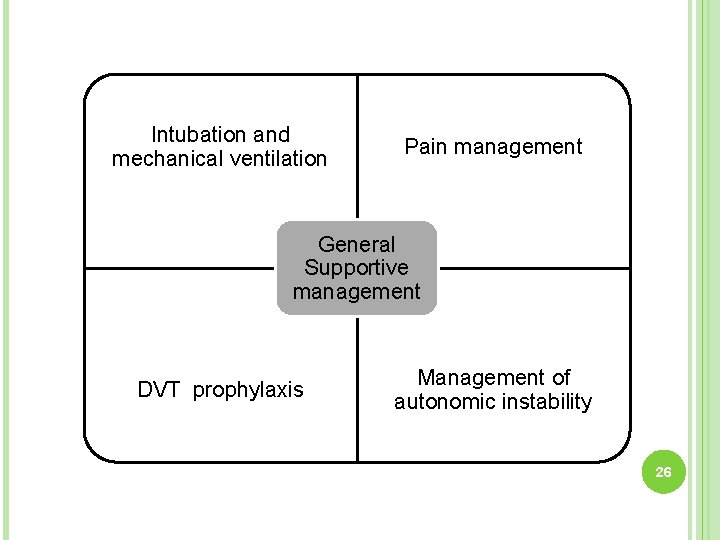
Intubation and mechanical ventilation Pain management General Supportive management DVT prophylaxis Management of autonomic instability 26
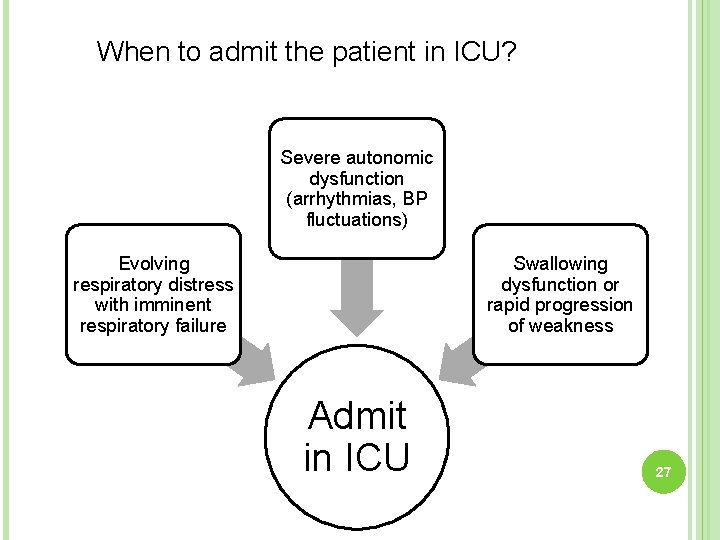
When to admit the patient in ICU? Severe autonomic dysfunction (arrhythmias, BP fluctuations) Evolving respiratory distress with imminent respiratory failure Swallowing dysfunction or rapid progression of weakness Admit in ICU 27
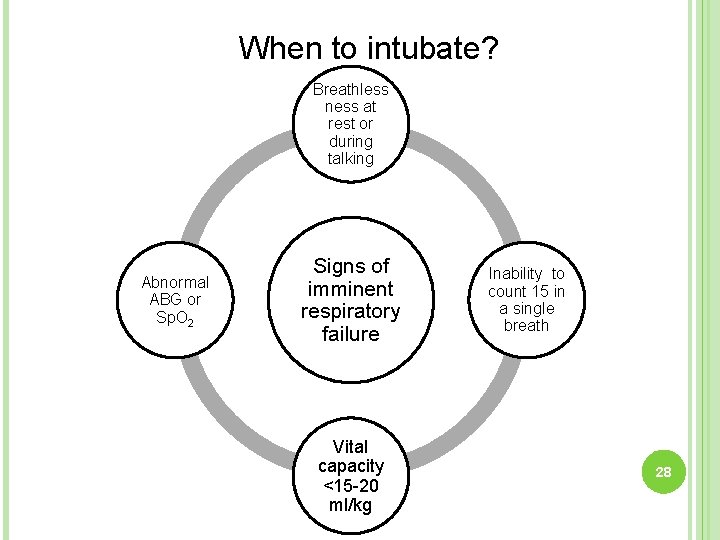
When to intubate? Breathless ness at rest or during talking Abnormal ABG or Sp. O 2 Signs of imminent respiratory failure Vital capacity <15 -20 ml/kg Inability to count 15 in a single breath 28

Can we predict the probability of requirement of ventilation? ERASMUS GBS RESPIRATORY INSUFFICIENCY SCORE (EGRIS)- It calculates the probability that a patient with GBS will require mechanical ventilation within 1 wk of assessment. Although this model is based on Dutch population of patients with GBS of > 6 yrs of age and has not been validated internationally. 29
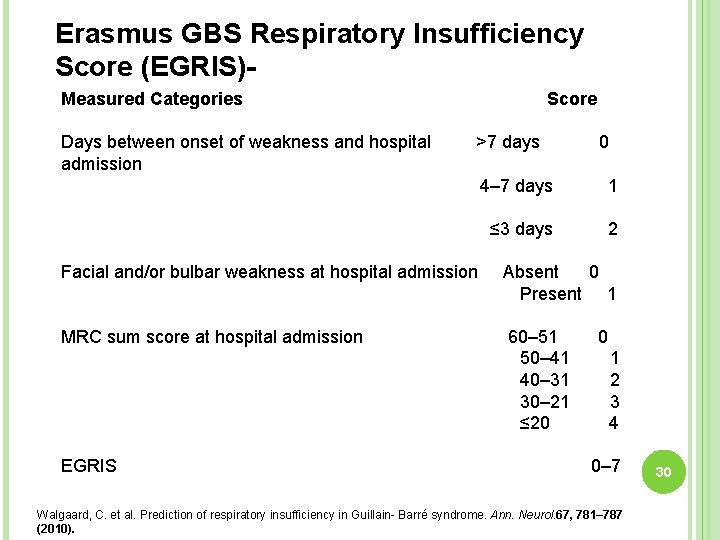
Erasmus GBS Respiratory Insufficiency Score (EGRIS)Measured Categories Days between onset of weakness and hospital admission Score >7 days 0 4– 7 days 1 ≤ 3 days 2 Facial and/or bulbar weakness at hospital admission Absent 0 Present 1 MRC sum score at hospital admission 60– 51 50– 41 40– 31 30– 21 ≤ 20 EGRIS 0 1 2 3 4 0– 7 Walgaard, C. et al. Prediction of respiratory insufficiency in Guillain- Barré syndrome. Ann. Neurol. 67, 781– 787 (2010). 30
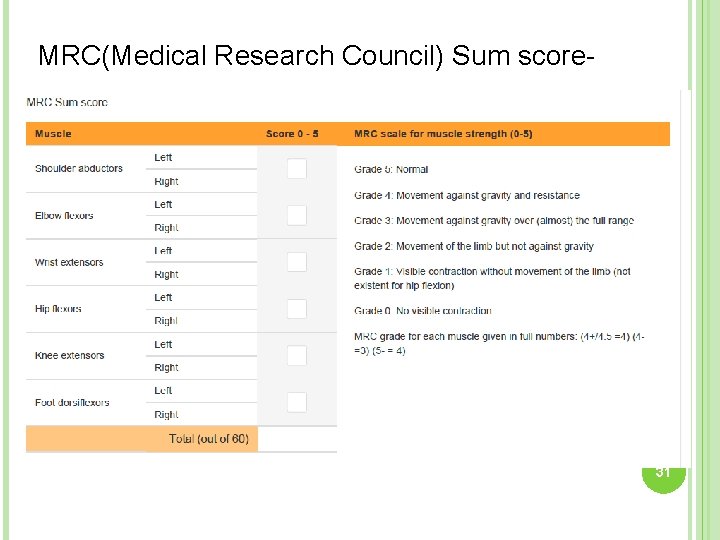
MRC(Medical Research Council) Sum score- 31
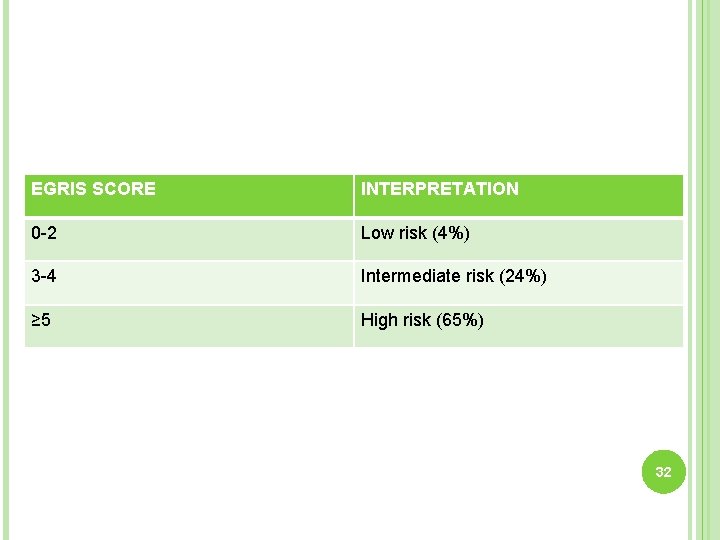
EGRIS SCORE INTERPRETATION 0 -2 Low risk (4%) 3 -4 Intermediate risk (24%) ≥ 5 High risk (65%) 32

RISK FACTORS FOR PROLONGED MECHANICAL VENTILATION: Inability to lift the arms from the bed at one week after intubation and An axonal subtype OR Unexcitable nerves in electrophysiological studies Early tracheostomy should be considered in patients who have this risk factors 33
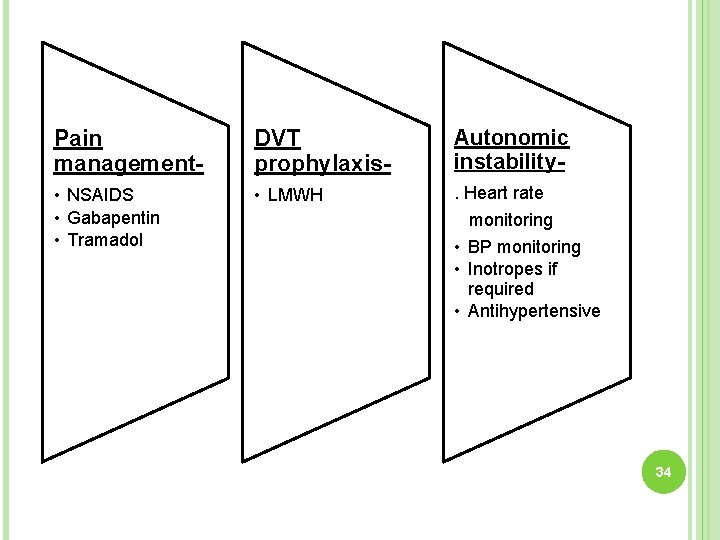
Pain management- DVT prophylaxis- Autonomic instability- • NSAIDS • Gabapentin • Tramadol • LMWH . Heart rate monitoring • BP monitoring • Inotropes if required • Antihypertensive 34
When to start specific treatment? Patient is unable to walk independently for 10 meters. Rapidly progressive weakness Signs of bulbar failure or respiratory insufficiency 35
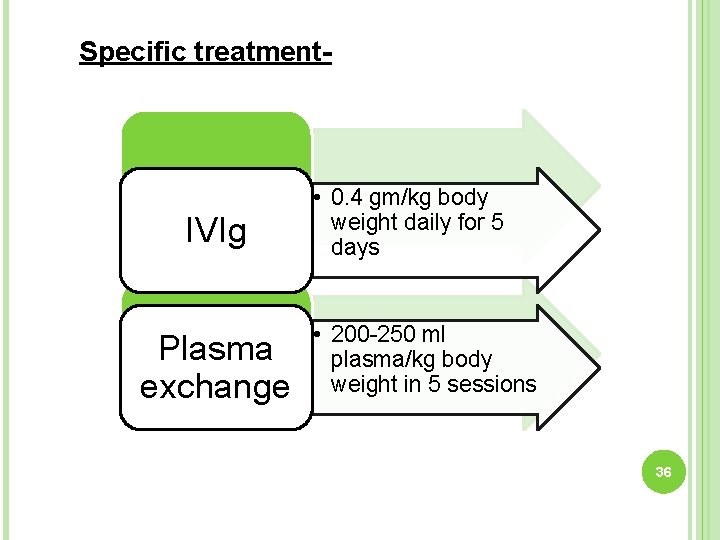
Specific treatment- IVIg Plasma exchange • 0. 4 gm/kg body weight daily for 5 days • 200 -250 ml plasma/kg body weight in 5 sessions 36

IVIG THERAPY IN GBS- Contraindications. Selective Ig. A deficiency Anaphylaxis following previous intravenous immunoglobulin infusion Relative contraindications. Severe congestive cardiac failure Renal insufficiency 37

MECHANISM OF ACTION OF IVIG IN GBSIVIG interferes with co-stimulatory molecules involved in antigen presentation and modulation of auto-antibodies, cyotokines and adhesion molecules production. Disrupts complement activation and membrane attack complex formation. Upregulates surface expression of the inhibitory Fc receptor, Fc gamma receptor IIb, on inflammatory cells, thereby attenuating autoantibody-initiated inflammation. 38

HOW TO GIVE IVIG? The total dose of IVIg is 2 gm/kg administered over two to five days. There is no advantage to giving it in less than five days for this disorder. While the side effects are usually mild, the infusions are generally better tolerated if given over five days. The first infusion to be started at a very slow rate of 25 to 50 ml/hr for 30 minutes and then increasing it progressively by 50 ml/hr every 15 to 20 minutes up to 150 to 200 ml/per hr. One study indicated that TRFs were more in 2 -days regimen than 5 -days regimen. 39
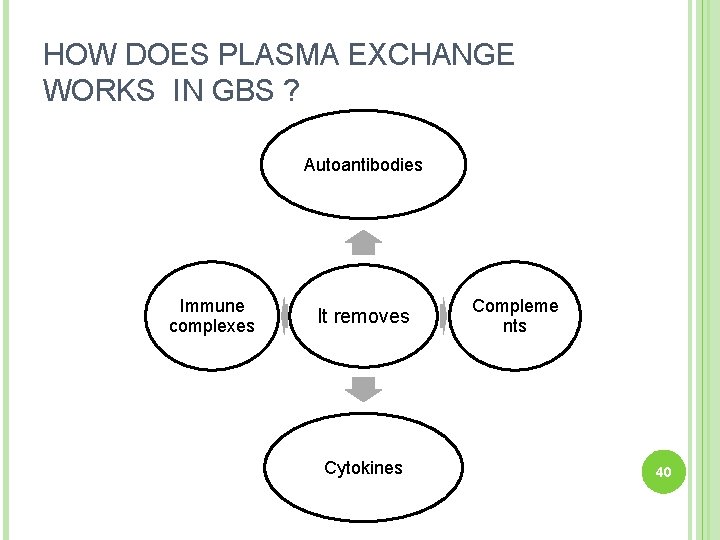
HOW DOES PLASMA EXCHANGE WORKS IN GBS ? Autoantibodies Immune complexes It removes Cytokines Compleme nts 40
HOW TO DO PLASMA EXCHANGE IN GBS? Start with 50 ml/kg For 5 times- daily/alternate day Over 5 -10 days, total 250 ml/kg. 41
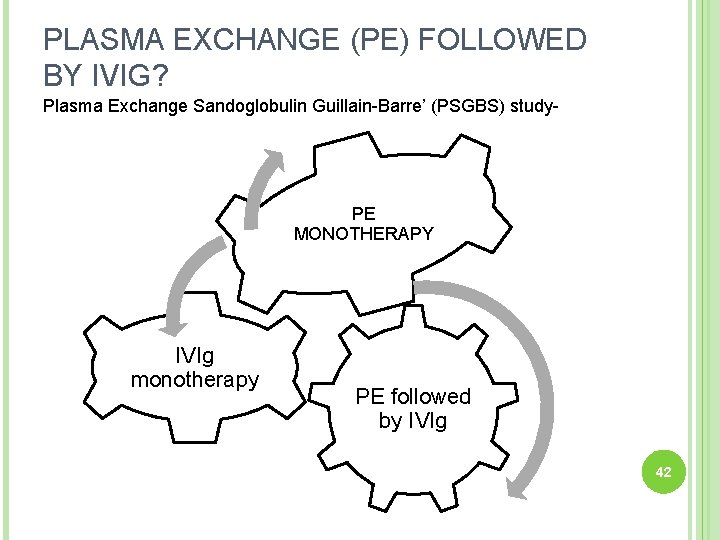
PLASMA EXCHANGE (PE) FOLLOWED BY IVIG? Plasma Exchange Sandoglobulin Guillain-Barre’ (PSGBS) study- PE MONOTHERAPY IVIg monotherapy PE followed by IVIg 42

This study showed- Efficacy Side effects Outcome No difference 43 Hughes RA. Plasma exchange versus intravenous immunoglobulin for Guillain-Barre syndrome. Ther Apher. 1997; 1: 129– 130. [Pub. Med: 10225757]
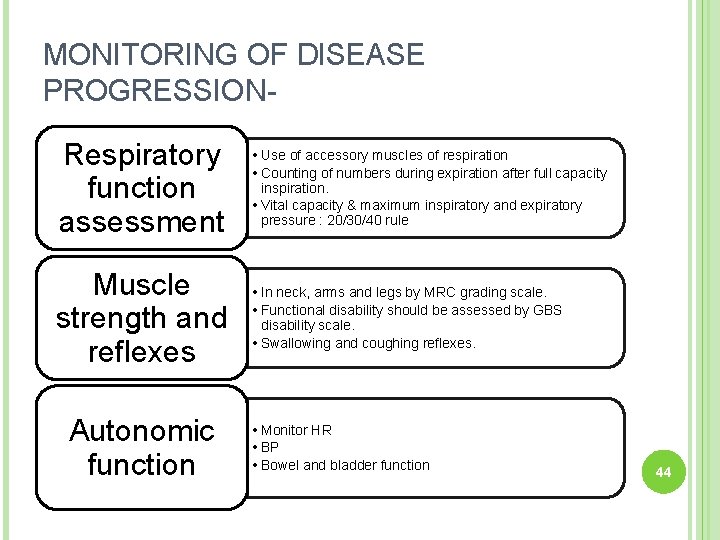
MONITORING OF DISEASE PROGRESSION- Respiratory function assessment • Use of accessory muscles of respiration • Counting of numbers during expiration after full capacity inspiration. • Vital capacity & maximum inspiratory and expiratory pressure : 20/30/40 rule Muscle strength and reflexes • In neck, arms and legs by MRC grading scale. • Functional disability should be assessed by GBS disability scale. • Swallowing and coughing reflexes. Autonomic function • Monitor HR • BP • Bowel and bladder function 44
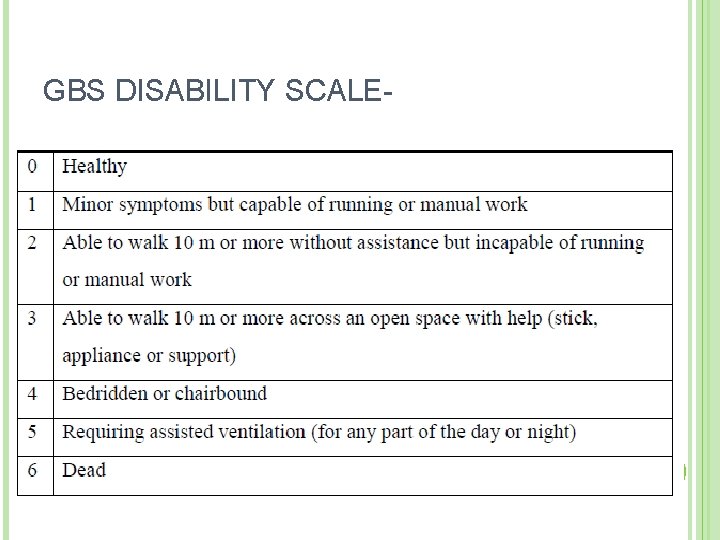
GBS DISABILITY SCALE- 45
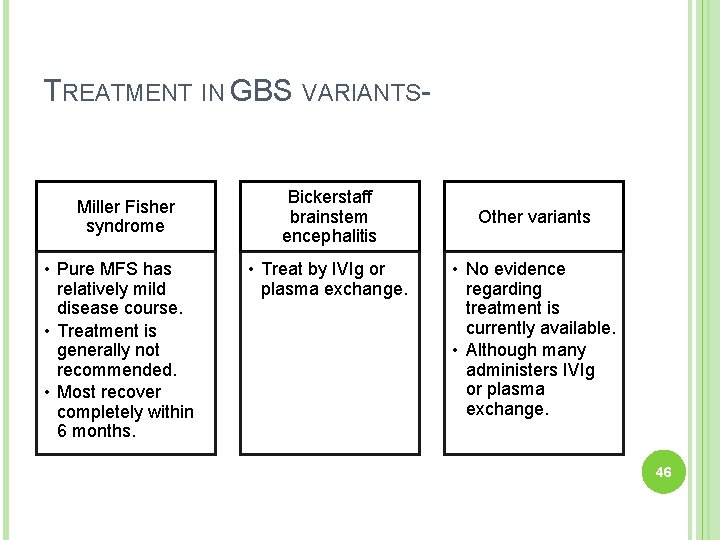
TREATMENT IN GBS VARIANTS- Miller Fisher syndrome • Pure MFS has relatively mild disease course. • Treatment is generally not recommended. • Most recover completely within 6 months. Bickerstaff brainstem encephalitis • Treat by IVIg or plasma exchange. Other variants • No evidence regarding treatment is currently available. • Although many administers IVIg or plasma exchange. 46
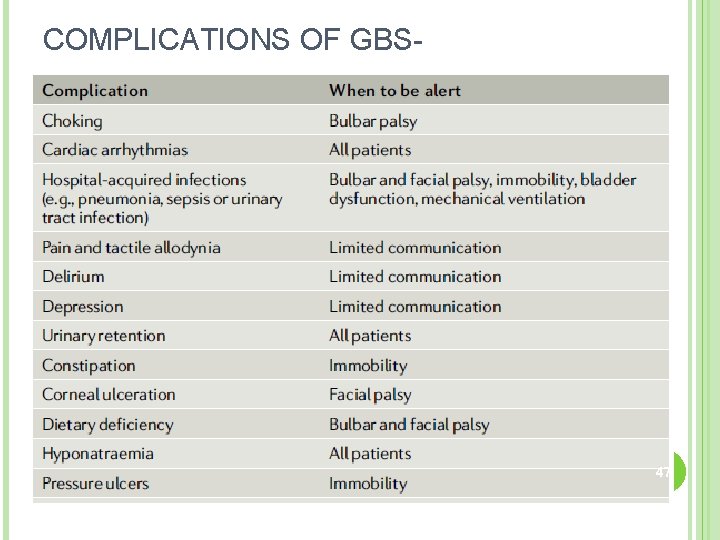
COMPLICATIONS OF GBS- 47

INSUFFICIENT RESPONSE TO TREATMENT About 40%* of patients treated with standard doses of plasma exchange or IVIg do not improve in the first 4 weeks following treatment. It does not imply that the treatment is ineffective, as progression might have been worse without therapy. One may consider repeating the treatment or changing to an alternative treatment, but no evidence exists that this approach will improve the outcome. 48 *Verboon, C. , van Doorn, P. A. & Jacobs, B. C. Treatment dilemmas in Guillain- Barré syndrome. J. Neurol. Neurosurg. Psychiatry 88, 346– 352 (2017).

TREATMENT RELATED FLUCTUATIONS TRFs are observed in 6– 10%* of patients with GBS. It is defined as disease progression occurring within 2 months following an initial treatment- induced clinical improvement or stabilization. The general view is that a TRF indicates that the treatment effect has worn off while the inflammatory phase of the disease is still ongoing. So, patients with GBS who display TRFs might benefit from further treatment, and repeating the full course of IVIg or plasma exchange in these patients is a common practice. 49 *Kleyweg, R. P. & van der Meche, F. G. Treatment related fluctuations in Guillain- Barré syndrome after high- dose immunoglobulins or plasma- exchange. J. Neurol. Neurosurg. Psychiatry 54, 957– 960(1991).
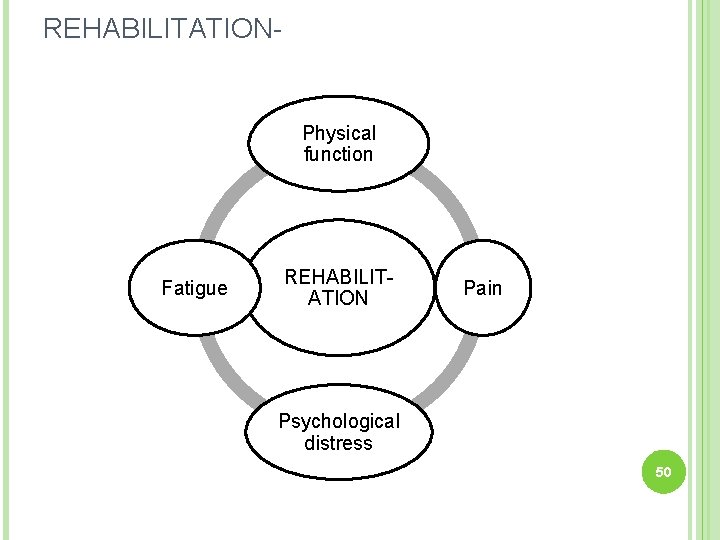
REHABILITATION- Physical function Fatigue REHABILITATION Pain Psychological distress 50
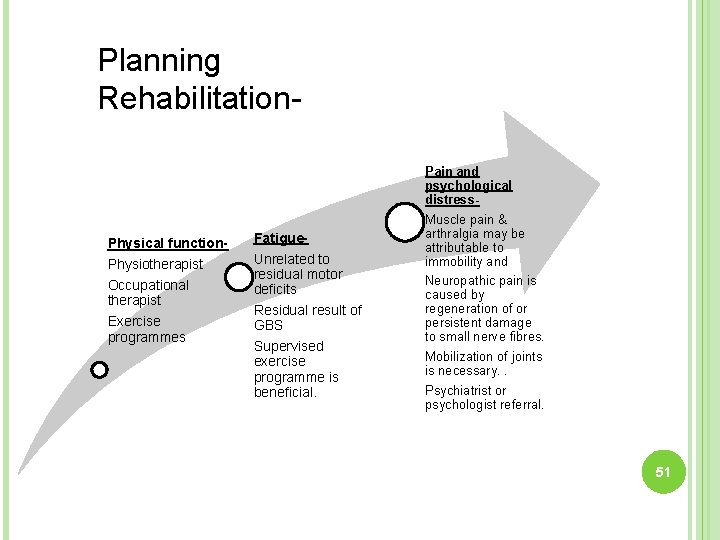
Planning Rehabilitation- Physical function- Fatigue- Physiotherapist Unrelated to residual motor deficits Residual result of GBS Supervised exercise programme is beneficial. Occupational therapist Exercise programmes Pain and psychological distress. Muscle pain & arthralgia may be attributable to immobility and Neuropathic pain is caused by regeneration of or persistent damage to small nerve fibres. Mobilization of joints is necessary. . Psychiatrist or psychologist referral. 51
52

OUTCOME PREDICTIONCan be done by modified Erasmus GBS Outcome Score (m. EGOS)This prognostic model based on a Dutch cohort predicting the ability to walk independently at 6 months. A high score corresponds to a high risk of being unable to walk independently at 6 months. This has been validated in a Japanese cohort. Further validation is needed in other populations. Generally about 80%* patients with GBS regain the ability to walk independently at 6 months of disease onset. 53 *Doets, A. Y. et al. Regional variation of Guillain- Barré syndrome. Brain 141, 2866– 2877 (2018).
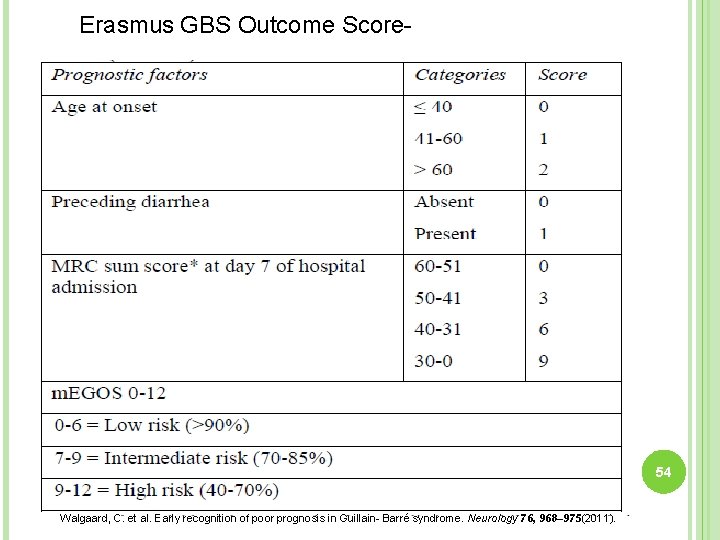
Erasmus GBS Outcome Score- 54 Walgaard, C. et al. Early recognition of poor prognosis in Guillain- Barré syndrome. Neurology 76, 968– 975(2011).

OUTCOME PREDICTION (CONTD. ) Death occurs in 3 -10% of cases (cardiovascular such as arrhythmia or fluctuations in BP or respiratory complications) either in acute or recovery phase. Recurrent episodes of GBS is rare affecting 2 -5% of cases. AMAN variety has more delayed recovery than AIDP. The alternative diagnosis of relapsing-remitting chronic inflammatory demyelinating polyneuropathy (CIDP) should be considered in relapsing cases. 55
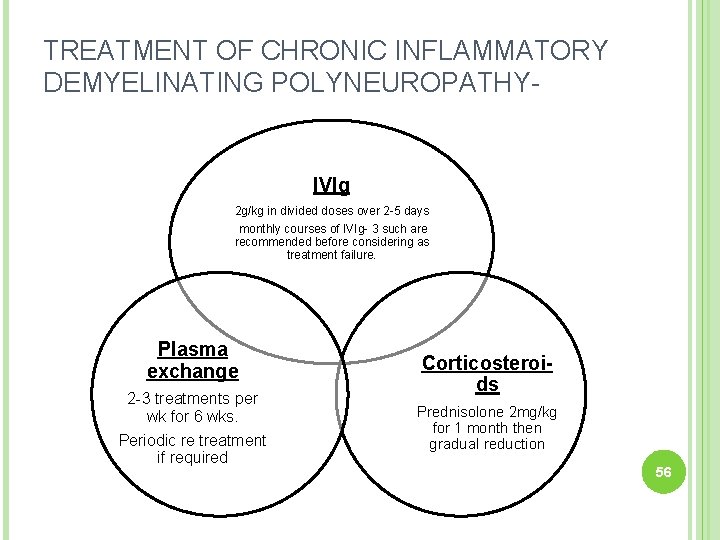
TREATMENT OF CHRONIC INFLAMMATORY DEMYELINATING POLYNEUROPATHY- IVIg 2 g/kg in divided doses over 2 -5 days monthly courses of IVIg- 3 such are recommended before considering as treatment failure. Plasma exchange 2 -3 treatments per wk for 6 wks. Periodic re treatment if required Corticosteroids Prednisolone 2 mg/kg for 1 month then gradual reduction 56

TREATMENT OF CIDP Mild cases- wait & observe. Monitor disease activity; clinical & NCS Moderate cases. One dose of IVIg -2 gm/kg over 2 -5 days. OR Oral prednisolone- 2 mg/kg for 4 wks, followed by tapering over 12 -24 wks. If worsening- consider other regime or plasma exchange. Severe cases- 3 consecutive course (4 -6 wks) of IVIg- 2 gm/kg, OR Oral prednisolone-2 mg/kg for 8 wks, followed by tapering over 52 -78 wks. If worsening- consider other regime or IVIg + 57 Prednisolone 0 r plasma exchange Ref: Oaklander AL et al. Treatment of chronic inflammatory demyelinating polyradiculo neuropathy : An overview of systematic reviews, Cochrane Systematic review. Jan, 2017

If the patient responds to corticosteroids the infusion intervals can be gradually increased or the dosage decreased. One-third of patients with CIDP fail to respond adequately. Patients who fail therapy with IVIg, PE, and glucocorticoids may benefit from treatment with azathioprine, methotrexate, cyclosporine, cyclophosphamide or Mycophenolate mofetil. CIDP associated with anti-CNTN 1(Contactin 1) and NF 155 (Neurofascin 155) antibodies is typically refractory to IVIg, but may respond to rituximab. 375 mg/m 2 once weekly 4 wks, followed by one dose per month for additional 2 doses. 58

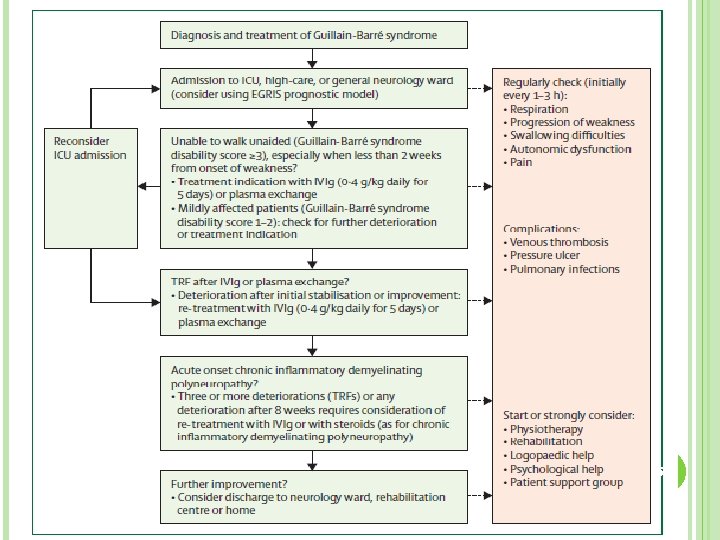
TEN STEPS IN DIAGNOSIS & MANAGEMENT OF GB SYNDROME 1. When to suspect GBS Rapidly progressive bilateral limb weakness and/or sensory deficits Hypo/areflexia Facial or bulbar palsy Ophthalmoplegia and ataxia 59

2. Check diagnostic criteria Exclude other causes Consider: - Routine laboratory tests - CSF examination - Electrophysiological studies 3. How to diagnose GBS- When to admit to ICU- One or more: - Rapid progression of weakness - Severe autonomic or swallowing dysfunction - Evolving respiratory distress - EGRIS > 4 60

4. When to start treatment- One or more: - Inability to walk >10 m independently - Rapid progression of weakness - Severe autonomic or swallowing dysfunction - Respiratory insufficiency 5. Treatment options. IVIg- 0. 4 mg/kg daily for 5 days Plasma exchange- (200 -250 ml/kg for 5 session) 61

6. Monitoring- Regularly assess : Muscle strength Respiratory function Swallowing function Autonomic function – - Blood pressure - Heart rate and rhythm - Bladder / bowel control 62
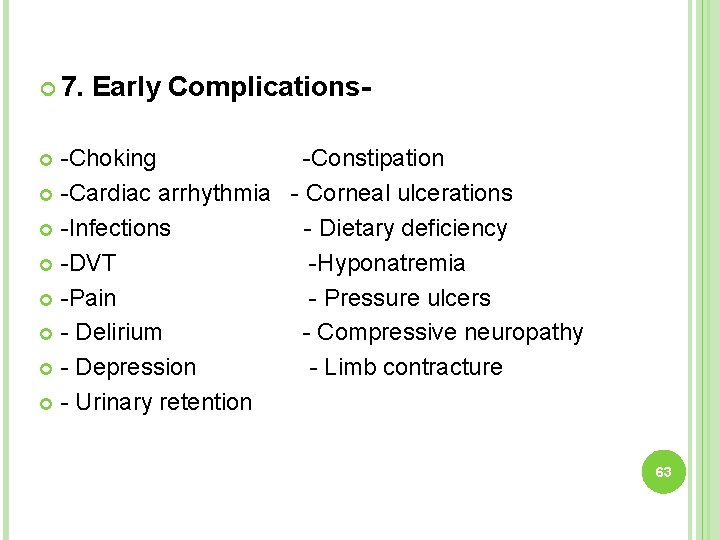
7. Early Complications- -Choking -Constipation -Cardiac arrhythmia - Corneal ulcerations -Infections - Dietary deficiency -DVT -Hyponatremia -Pain - Pressure ulcers - Delirium - Compressive neuropathy - Depression - Limb contracture - Urinary retention 63

8. Clinical progression- Treatment related fluctuations: - Repeat same treatment No initial response or incomplete recovery - No evidence for repeating treatment 9. Predicting outcome – Calculate m. EGOS on admission Recovery can continue > 3 years after onset Recurrence is rare (2 -5%) 64

10. Rehabilitation. Start rehabilitation programme early Manage long- term complaints: - Fatigue - Pain - Psychological stress Contact GBS patient organizations 65

SOME RARE PRESENTATIONS OF GBS 66

67

The variant most commonly reported to be associated with retained or brisk reflex is AMAN variety. 68
69

This patient was readmitted after 4 days of discharge with an acute onset flaccid quadruparesis, first involving lower limbs and evolving over the next 24 h to involve both upper limbs. In this patient serology for viral infections that predispose to GBS was negative. There was a clear temporal relationship between scrub typhus infection and the onset of flaccid weakness 70

71

This patient represented 3 days after discharge with worsening lower limb paraesthesia and unable to mobilize due to ascending lower limb weakness. He received intravenous immunoglobulin (IVIG) 0. 4 g/kg daily for 5 days and his symptoms stabilized. 72

THANK YOU 73
- Slides: 73