Diagnosing Diabetes In Adults Type 1 LADA or

Diagnosing Diabetes In Adults– Type 1, LADA, or Type 2? Part 2 2 Stanley Schwartz MD, FACE, FACP Affiliate Main Line Health Emeritus, Clinical Assoc. Prof. of Medicine Perlman School of Medicine, University of Pennsylvania Struan F. A. Grant, Ph. D Children’s Hospital of Philadelphia Associate Professor, University of Pennsylvania Vanessa Guy Children’s Hospital of Philadelphia Senior Clinical Research Coordinator Co-Investigators NIH RO-1, Genes in LADA

PROPOSAL: ‘β-Cell Centric’ Classification of Diabetes Ø Intuitively obvious approach Ø Didn’t we know that all along! It can help define diagnosis and therapy better, especially as our knowledge-base increases
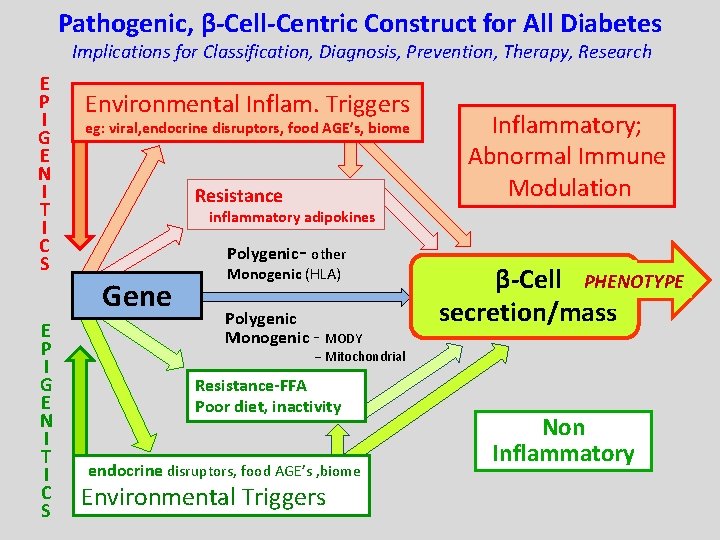
Pathogenic, β-Cell-Centric Construct for All Diabetes Implications for Classification, Diagnosis, Prevention, Therapy, Research E P I G E N I T I C S Environmental Inflam. Triggers eg: viral, endocrine disruptors, food AGE’s, biome Resistance inflammatory adipokines Polygenic- other Gene E P I G E N I T I C S Inflammatory; Abnormal Immune Modulation Monogenic (HLA) Polygenic Monogenic - MODY β-Cell PHENOTYPE secretion/mass − Mitochondrial Resistance-FFA Poor diet, inactivity endocrine disruptors, food AGE’s , biome Environmental Triggers Non Inflammatory
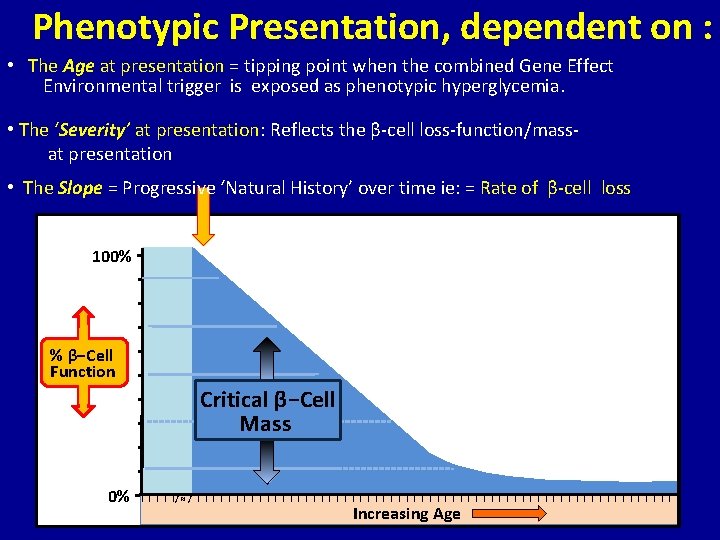
Phenotypic Presentation, dependent on : • The Age at presentation = tipping point when the combined Gene Effect Environmental trigger is exposed as phenotypic hyperglycemia. • The ‘Severity’ at presentation: Reflects the β-cell loss-function/massat presentation • The Slope = Progressive ‘Natural History’ over time ie: = Rate of β-cell loss 100% − − % β−Cell − Function − − Critical β−Cell − Mass − − 0% −I I I/ ≈ / I I I I I I I I I I I I I I I Increasing Age
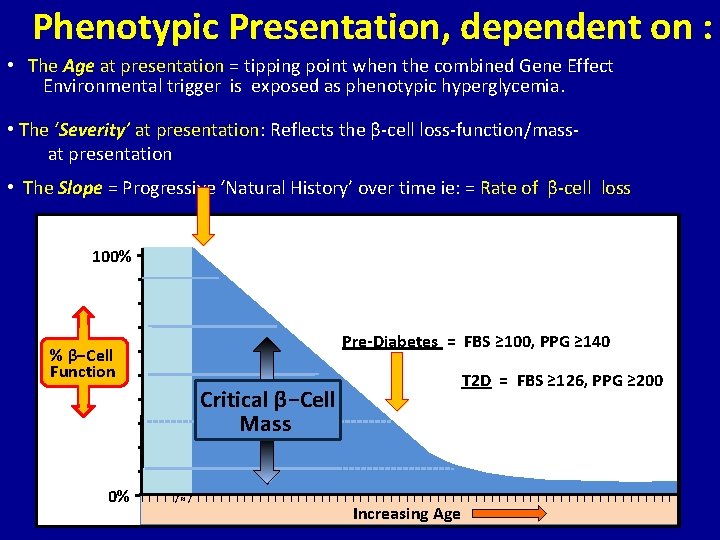
Phenotypic Presentation, dependent on : • The Age at presentation = tipping point when the combined Gene Effect Environmental trigger is exposed as phenotypic hyperglycemia. • The ‘Severity’ at presentation: Reflects the β-cell loss-function/massat presentation • The Slope = Progressive ‘Natural History’ over time ie: = Rate of β-cell loss 100% − − Pre-Diabetes = FBS ≥ 100, PPG ≥ 140 − % β−Cell Function − T 2 D = FBS ≥ 126, PPG ≥ 200 − Critical β−Cell − Mass − − 0% −I I I/ ≈ / I I I I I I I I I I I I I I I Increasing Age

New β-Cell Centric Construct: Implications Genetics 101 for Diabetes Phenotype: is DEPENDENT ON Genotype: 1. Number of Genes, which genes-their nature, even Epigenetics recent article on epigenetics in T 1 DM-DCCT/EDIC ie: Genes influencing: insulin secretory dynamics, insulin resistance, sites of susceptibility of β-Cell to destruction by Endogenous/Exogenous Triggers eg: inflammation, etc. i. e. Genetics is: Which genes, how many different ones, the ‘severity/intensity’ of expression!

Genotyping Should Be a Standard Diagnostic Marker to be obtained For DX of DM: (Cost now $100) eg: Pharmacogenetics Pick right drug for right patient • Find Gene action/ Function- Leads to understanding mechanisms Ø eg: TCF 7 L 2 -Potential Therapy ØPARP-1 Inhibitor? ? , incretin Ø Or Gene/Mechanism/ Therapy Ø low BMR- results in morbid obesity Ø Asian/ Eastern Europeans- store more Visceral Fat at Lower BMI
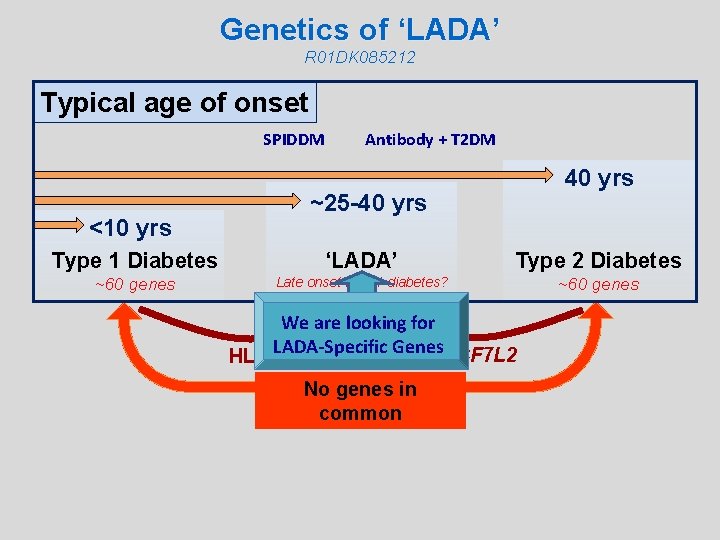
Genetics of ‘LADA’ R 01 DK 085212 Typical age of onset SPIDDM <10 yrs Antibody + T 2 DM 40 yrs ~25 -40 yrs Type 1 Diabetes ‘LADA’ Type 2 Diabetes ~60 genes Late onset type 1 diabetes? ~60 genes We are looking for HLA LADA-Specific Genes. TCF 7 L 2 No genes in common

New β-Cell Centric Construct: Implications β-cell Issues Ø Usual use of Glycemic Criteria Ø Usual/Occasional Use of C-Peptide Ø Try to Determine Mono-Genetic Causes Ø Development of therapies aimed at • Improving β-cell function: • Reduce Glucotoxicity • Reduce Lipotoxicity • Reduce IR (treat inflammation, gut biome change)
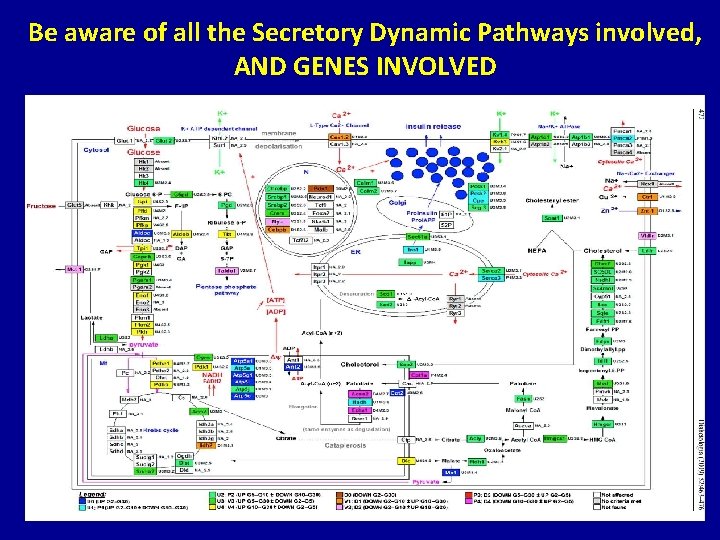
Be aware of all the Secretory Dynamic Pathways involved, AND GENES INVOLVED
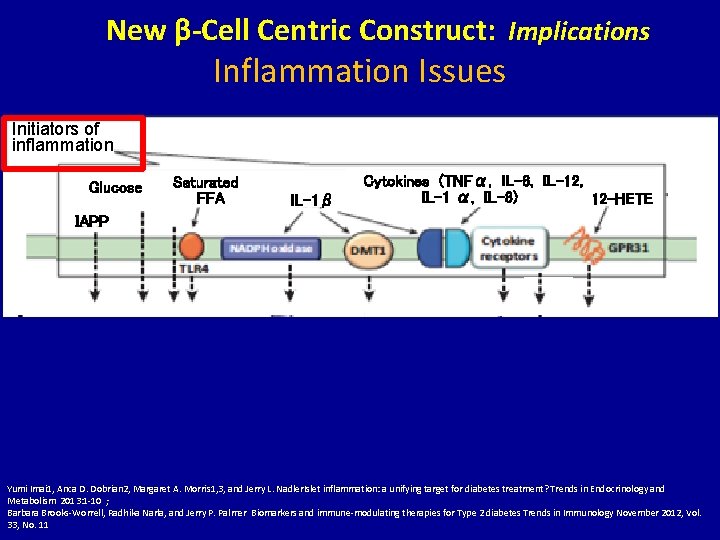
New β-Cell Centric Construct: Implications Inflammation Issues Initiators of inflammation Glucose Saturated FFA IL-1β Cytokines (TNFα, IL-6, IL-12, IL-1 α, IL-8) 12 -HETE IAPP Yumi Imai 1, Anca D. Dobrian 2, Margaret A. Morris 1, 3, and Jerry L. Nadler. Islet inflammation: a unifying target for diabetes treatment? Trends in Endocrinology and Metabolism 2013: 1 -10 ; Barbara Brooks-Worrell, Radhika Narla, and Jerry P. Palmer Biomarkers and immune-modulating therapies for Type 2 diabetes Trends in Immunology November 2012, Vol. 33, No. 11
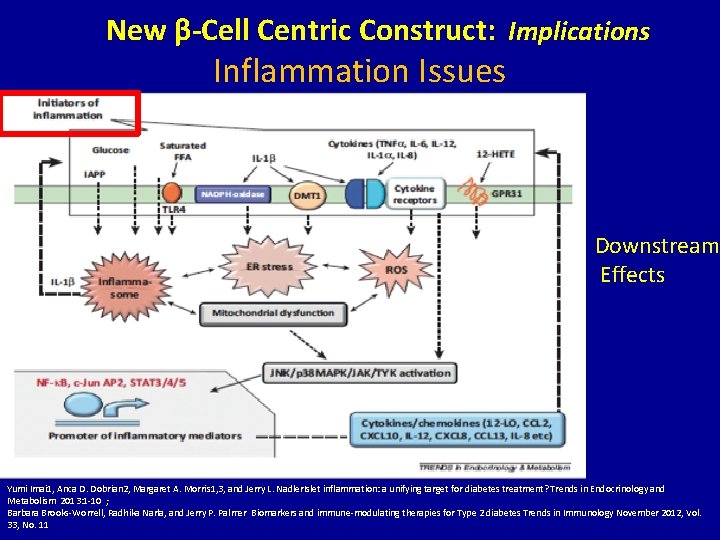
New β-Cell Centric Construct: Implications Inflammation Issues Downstream Effects Yumi Imai 1, Anca D. Dobrian 2, Margaret A. Morris 1, 3, and Jerry L. Nadler. Islet inflammation: a unifying target for diabetes treatment? Trends in Endocrinology and Metabolism 2013: 1 -10 ; Barbara Brooks-Worrell, Radhika Narla, and Jerry P. Palmer Biomarkers and immune-modulating therapies for Type 2 diabetes Trends in Immunology November 2012, Vol. 33, No. 11

Potential Immunomodulatory Therapy to Prevent / Treat / Reverse Diabetes- (and not just Type 1 DM) www. thelancet. com Published online July 26, 2013 http: //dx. doi. org/10. 1016/SO 140 -6736(13)60591 -7 A promising approach is the use of pharmacological agents, such as orally active chemical chaperones, which can stabilize protein conformation, improve ER folding capacity, and facilitate the trafficking of mutant proteins. 110– 113 Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy 2014: 7 25– 34 CLAUDIA CAVELTI-WEDER, Effects of Gevokizumab on Glycemia and Inflammatory Markers in Type 2 Diabetes Care 35: 1654– 1662, 2012 C. Levitan, , Proposal for generating new beta cells in a muted immune environment for type 1 diabetes [cyclosporin/PPI] Diabetes Metab Res Rev 2013; 29: 604
- Slides: 13