Diagnosing Acid Base Disorders without your PDA Daniel

Diagnosing Acid Base Disorders (without your PDA) Daniel Dilling, MD Adapted from: Morganroth, M. Journal of Critical Illness; 5(2): 138 -54.

When to be suspicious… and get an arterial blood gas • Most often indicated after an abnormal electrolyte panel – Low total CO 2 – High anion gap • Acute or chronic respiratory failure • Evaluate an ingestion • Always draw one at the same time as the electrolyte panel!!!

The elements of the ABG • • p. H p. CO 2 p. O 2 HCO 3 Base excess/deficit O 2 saturation Methemoglobin, Carboxyhemoglobin

The elements of the ABG • • p. H p. CO 2 p. O 2 HCO 3 Base excess/deficit O 2 saturation Methemoglobin, Carboxyhemoglobin

The elements of the ABG • • p. H p. CO 2 p. O 2 HCO 3 Base excess/deficit O 2 saturation Methemoglobin, Carboxyhemoglobin
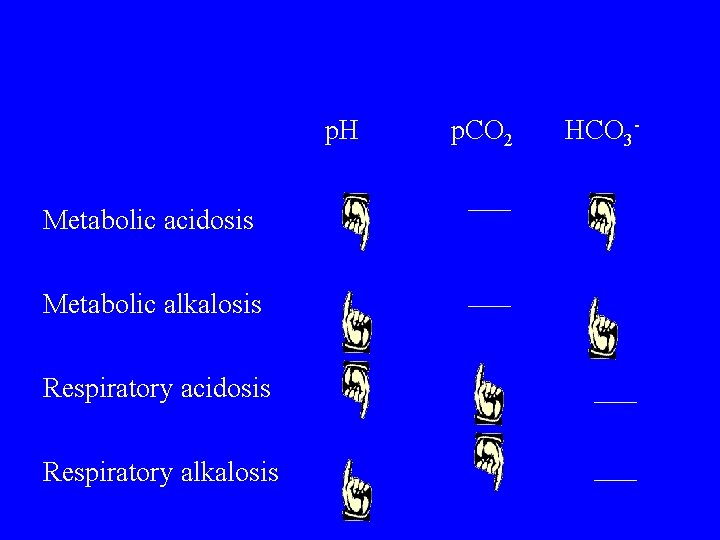
p. H Metabolic acidosis Metabolic alkalosis Respiratory acidosis Respiratory alkalosis p. CO 2 HCO 3 -

Metabolic Acidosis Anion gap M U D P I L E S Non-anion gap U S E D C A R S

Anion Gap Metabolic Acidosis M Methanol U Uremia Sulfuric, Phosphoric, Uric D DKA (AKA) Acetone, Acetoacetate, B-hydroxybuteric P Paraldehyde Organic acids I INH ? ? L Lactic (from what? ) Lactic E S Ethylene Glycol Salicylates Oxalic, Glycolic Formic

Anion Gap Metabolic Acidosis M Methanol U Uremia Sulfuric, Phosphoric, Uric D DKA (AKA) Acetone, Acetoacetate, B-hydroxybuteric P Paraldehyde Organic acids I INH ? ? L Lactic (from what? ) Lactic E S Ethylene Glycol Salicylates Oxalic, Glycolic Formic

Anion Gap Metabolic Acidosis M Methanol U Uremia Sulfuric, Phosphoric, Uric D DKA (AKA) Acetone, Acetoacetate, B-hydroxybuteric P Paraldehyde Organic acids I INH ? ? L Lactic (from what? ) Lactic E S Ethylene Glycol Salicylates Oxalic, Glycolic Salicylic, Lactic Formic

Non-Anion Gap Metabolic Acidosis U S Uretero-Sigmoid diversions (GI bicarbonate loss) E Ethanol or Enocrinopathies D Diarrhea C Carbonic Anhydrase Inhibitors A Hyper-Alimentation R S RTA Saline

Metabolic Alkalosis Vomiting or NG suction Fluid volume contraction (“contraction alkalosis”) Hypokalemia Cushing’s Disease, glucocorticoid, mineralocorticoid Post-chronic hypercapnia Bartter’s or Gitelman's Syndrome Massive blood transfusion (what preservative? ) Ingestion or administration of bicarbonate – “Milk-alkali Syndrome”

Respiratory Acidosis (Hypoventilation) • CNS – Sedatives (iatrogenic or street) – Obesity-hypoventilation • Pleural Disease – Effusion, PTX • Intrinsic Lung Disease – – COPD ARDS PE Pneumonia • Musculoskeletal or neurologic – Kyphoscoliosis – Guillain-Barre (flaccid paralysis) – Myesthenia Gravis – Botulism – Polymyositis – Multiple sclerosis – ALS • Other – OSA

Respiratory Alkalosis Catastrophic CNS event Anxiety or Pain Early asthma exacerbation Sepsis PE Cirrhosis Pulmonary Fibrosis Pregnancy Salicylates Progesterone

Six Step Approach to Acid-Base Analysis (fewer than AA!) 1. Is the patient acidemic or alkalemic? 2. Is the overriding disturbance respiratory or metabolic? 3. If respiratory… is it acute or chronic? 4. If metabolic… is there an high anion gap? 5. If metabolic… is the respiratory system compensating appropriately? 6. Is there a second metabolic disturbance present?

Six Step Approach 1. Is the patient acidemic or alkalemic? 2. 3. 4. 5. 6. Is the overriding disturbance respiratory or metabolic? If respiratory… is it acute or chronic? If metabolic… is there an high anion gap? If metabolic… is the respiratory system compensating appropriately? Is there a second metabolic disturbance present?

Six Step Approach 1. Is the patient acidemic or alkalemic? 2. Is the overriding disturbance respiratory or metabolic? 3. 4. 5. 6. If respiratory… is it acute or chronic? If metabolic… is there an high anion gap? If metabolic… is the respiratory system compensating appropriately? Is there a second metabolic disturbance present?

Six Step Approach 1. 2. Is the patient acidemic or alkalemic? Is the overriding disturbance respiratory or metabolic? 3. If respiratory… is it acute or chronic? Acute, p. H should change 0. 08 (p. CO 2 -40) / 10 Chronic, p. H should change 0. 03 (p. CO 2 -40) / 10 4. 5. 6. If metabolic… is there an high anion gap? If metabolic… is the respiratory system compensating appropriately? Is there a second metabolic disturbance present?

Acute, p. H should change 0. 08 (p. CO 2 -40) / 10 Chronic, p. H should change 0. 03 (p. CO 2 -40) / 10 So. . if the p. CO 2 is 80 the change is 80 – 40 = 40 40 / 10 = 4 the p. H should change by 0. 08 (4) = 0. 32 if acute the p. H should change by 0. 03 (4) = 0. 12 if chronic

Six Step Approach 1. 2. 3. Is the patient acidemic or alkalemic? Is the overriding disturbance respiratory or metabolic? If respiratory… is it acute or chronic? 4. If metabolic… is there an high anion gap? Anion gap = (Na+) – (Cl- + HCO 3 -) A normal anion gap is 5. 6. If metabolic… is the respiratory system compensating appropriately? Is there a second metabolic disturbance present?

Six Step Approach 1. 2. 3. Is the patient acidemic or alkalemic? Is the overriding disturbance respiratory or metabolic? If respiratory… is it acute or chronic? 4. If metabolic… is there an high anion gap? Anion gap = (Na+) – (Cl- + HCO 3 -) A normal anion gap is 12 5. 6. If metabolic… is the respiratory system compensating appropriately? Is there a second metabolic disturbance present?

Six Step Approach 1. 2. 3. 4. Is the patient acidemic or alkalemic? Is the overriding disturbance respiratory or metabolic? If respiratory… is it acute or chronic? If metabolic… is there an high anion gap? 5. If metabolic… is the respiratory system compensating appropriately? 1. 5 (HCO 3 -) +8 +/- 2 should equal p. CO 2 6. Is there a second metabolic disturbance present?

Six Step Approach 1. 2. 3. 4. Is the patient acidemic or alkalemic? Is the overriding disturbance respiratory or metabolic? If respiratory… is it acute or chronic? If metabolic… is there an high anion gap? 5. If metabolic… is the respiratory system compensating appropriately? 1. 5 (HCO 3 -) +8 +/- 2 should equal p. CO 2 If higher, then respiratory acidosis is also present If lower, then respiratory alkalosis is also present 6. Is there a second metabolic disturbance present?

Six Step Approach 1. 2. 3. 4. 5. Is the patient acidemic or alkalemic? Is the overriding disturbance respiratory or metabolic? If respiratory… is it acute or chronic? If metabolic… is there an high anion gap? If metabolic… is the respiratory system compensating appropriately? 6. Is there a second metabolic disturbance? • • “Delta anion gap” vs. “Delta bicarbonate” Measured HCO 3 - + (delta anion gap) should equal 24

Six Step Approach 1. 2. 3. 4. 5. Is the patient acidemic or alkalemic? Is the overriding disturbance respiratory or metabolic? If respiratory… is it acute or chronic? If metabolic… is there an high anion gap? If metabolic… is the respiratory system compensating appropriately? 6. Is there a second metabolic disturbance? • • “Delta anion gap” vs. “Delta bicarbonate” Measured HCO 3 - + (delta anion gap) should equal 24 If less than 24, then there is a non-gap acidosis present If more than 24, then there is a metabolic alkalosis present

Problem #1 “TRIPLE ACID-BASE DISORDER” 123 99 4. 0 5 1 o metabolic acidosis +AG 1 o respiratory alkalosis 1 o metabolic acidosis -AG ABG 7. 31/10/100/8 AG = 19 1. 5 (5) + 8 +/- 2 = 15. 5 +/- 2 The measured p. CO 2 is less than 13. 5… 5 + (19 -12) = 12 This is less than 24…

Problem #2 130 80 4. 0 10 1 o metabolic acidosis +AG 1 o metabolic alkalosis ABG 7. 20/25/100/8 AG = 40 1. 5 (10) + 8 +/- 2 = 23 +/- 2 The measured p. CO 2 is the same as the calculated 10 + (40 - 12) = 38 This is more than 24…

Problem #3 125 100 2. 5 8 1 o metabolic acidosis +AG 1 o respiratory acidosis 1 o metabolic acidosis -AG ABG 7. 07/28/100/10 AG = 17 1. 5 (8) + 8 +/- 2 = 20 +/- 2 The measured p. CO 2 is more than 22… 8 + (17 -12) = 13 This is less than 24…

Problem #4 135 93 4. 0 30 1 o respiratory acidosis (acute on chronic) ABG 7. 18/80/100/28 80 – 40 = 40 0. 03 (40/10)= 0. 12 0. 08 (40/10)= 0. 32 if chronic… if acute… 7. 40 – 7. 18 = 0. 22 is between 0. 12 and 0. 32…

Problem #5 “TRIPLE ACID-BASE DISORDER” 140 98 4. 0 17 1 o metabolic acidosis +AG 1 o respiratory acidosis 1 o metabolic alkalosis ABG 7. 14/45/100/18 AG = 25 1. 5 (17) + 8 +/- 2 = 25. 5 +/- 2 The measured p. CO 2 is more than 27. 5… 17 + (25 -12) = 30 This is more than 24…
- Slides: 31