Diabetic Retinopathy Clinical Research Network Factors Associated with

Diabetic Retinopathy Clinical Research Network Factors Associated with Visual Acuity and Central Subfield Thickness Changes when Treating DME with Anti. VEGF: An Exploratory Analysis of the Protocol T Randomized Clinical Trial 1

Objectives Ø Identify baseline factors that are associated with more successful 2 -year outcomes, regardless of specific anti. VEGF agent used • Secondary objective: identify interactions – factors that have differing effects on the outcomes based on the anti-VEGF assignment Ø Outcomes: 1. Change in visual acuity (VA) 2. Change in VA Area under the Curve (AUC) 3. Change in Central Subfield Thickness (CST) 2

Why do we Explore a Predictive Factor Analysis Ø To refine clinician/patient expectations of therapy outcomes Ø To look for any situations in which therapy has an adverse outcome Ø To determine if there may be a situation in which a particular drug is less effective 3

Background • Baseline Factors Previously identified to Predict more Successful Outcomes in Protocol T • New analysis aims to identify influential factors beyond what we know already • Baseline Factors Previously identified to be associated with more Successful Outcomes in Protocol I • VA: younger age, absence of ERM, less severe DR level 4
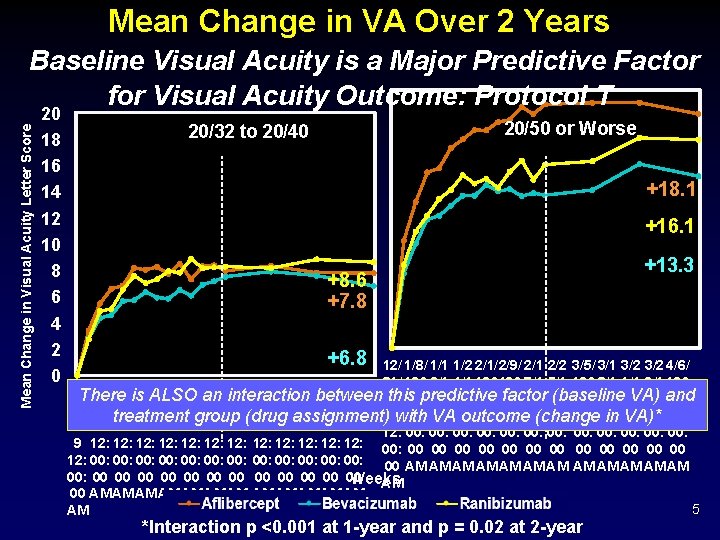
Mean Change in VA Over 2 Years Mean Change in Visual Acuity Letter Score Baseline Visual Acuity is a Major Predictive Factor for Visual Acuity Outcome: Protocol T 20 18 16 14 12 10 8 6 4 2 0 20/50 or Worse 20/32 to 20/40 +18. 1 +16. 1 +8. 6 +7. 8 +6. 8 +13. 3 12/ 1/8/ 1/1 1/2 2/1/2/9/ 2/1 2/2 3/5/ 3/1 3/2 4/6/ 31/ 190 6/1 4/1 190190 7/1 5/1 190 3/1 1/1 9/1 190 12/ 1/8/ 1/1 1/2 2/1/ 2/9/ 2/1 2/2 3/5/ 3/1 3/2 4/6/ There is ALSO an interaction between 189 this 0 predictive (baseline VA) 900 0 factor 0 900 900 900 and 0 31/ 190 6/1 4/1 190190 7/1 5/1 190 3/1 1/1 9/1 190 12: 12: 12: treatment group (drug assignment)9 with VA 12: outcome (change in 12: VA)* 189 0 900 900 0 12: 00: 00: 00: 9 12: 12: 12: 00: 00 00 00 12: 00: 00: 00: 00 AM AM AM 00: 00 00 00 Weeks AM 00 AM AM AM 5 AM *Interaction p <0. 001 at 1 -year and p = 0. 02 at 2 -year
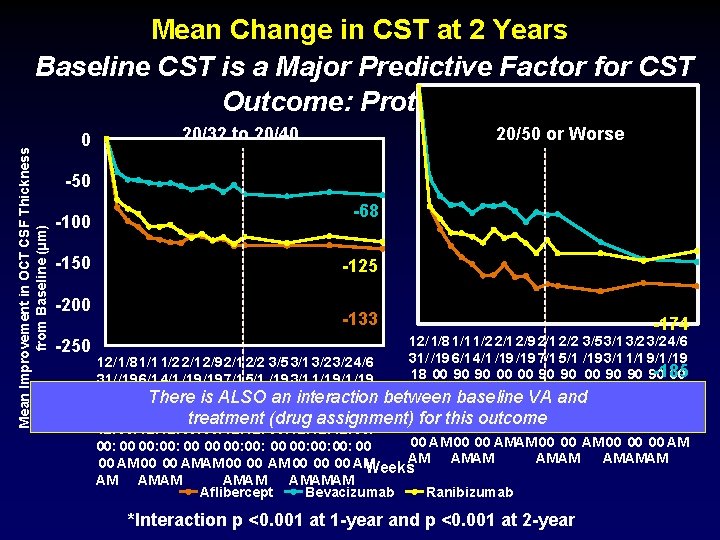
Mean Improvement in OCT CSF Thickness from Baseline (µm) Mean Change in CST at 2 Years Baseline CST is a Major Predictive Factor for CST Outcome: Protocol T 20/32 to 20/40 0 20/50 or Worse -50 -68 -100 -150 -125 -200 -250 -133 -174 12/ 1/8 1/1 1/2 2/1 2/9 2/1 2/2 3/5 3/1 3/2 4/6 31/ /19 6/1 4/1 /19 7/1 5/1 /19 3/1 1/1 9/1 /19 12/ 1/8 1/1 1/2 2/1 2/9 2/1 2/2 3/5 3/1 3/2 4/6 -185 18 00 90 90 90 00 31/ /19 6/1 4/1 /19 7/1 5/1 /19 3/1 1/1 9/1 /19 99 12: baseline 0 0 12: 0 and 0 12: 0 0 0 12: 18 00 90 90 00 is 00 ALSO 90 90 an 00 90 90 90 00 between There interaction VA -211 12: 00: 12: 12: 00: 99 12: 0 0 0 12: treatment (drug assignment)00: for 00 this outcome 00: 00: 00 12: 00: 12: 12: 00 AM 00 00 00 AM 00: 00: 00 00 00: 00: 00: 00 AM AMAMAM 00 00 AMAM 00 00 00 AM AM AMAM Aflibercept Weeks AMAMAM Bevacizumab Ranibizumab *Interaction p <0. 001 at 1 -year and p <0. 001 at 2 -year

29 Baseline Factors Evaluated: Protocol T Demographics (12) Gender Race/Ethnicity** Diabetes Type** DM Duration** Age Insulin Used** Mean Arterial BP Hb. A 1 c Current or former smoker** Hypertension** Prior MI, TIA, Stroke, or CAD** Urine Albumin Creatinine Ratio Ocular History or Exam (7) OCT (6) (Reading Center) Prior Treatment for DME (any)** Central Subfield Thickness Prior anti-VEGF for DME** Macular Volume Prior Laser for DME** DR Severity (judged by investigator) Visual Acuity Lens Status Type of DME (judged by investigator) Subretinal Fluid in Central 1 mm Intraretinal fluid. Cystoid Edema in Central 1 mm Epiretinal Membrane, Center 1 mm Deformed VMT-Vitreous Attached, Center Fundus Photo (4) (Reading Center) PRP + DR Severity Hard Exudates within 6 mm zone Surface Wrinkling Retinopathy Hemorrhages/ Microaneurysm within 6 mm zone 1 mm deformed* **Patient reported 7

29 Baseline Factors Evaluated: Protocol T Demographics (12) Gender Race/Ethnicity** Diabetes Type*, ** DM Duration** Age Insulin Used** Mean Arterial BP Hb. A 1 c Current or former smoker** Hypertension** Prior MI, TIA, Stroke, or CAD** Urine Albumin Creatinine Ratio Ocular History or Exam (7) OCT (6) (Reading Center) Prior Treatment for DME (any)** Central Subfield Thickness Prior anti-VEGF for DME** Macular Volume Prior Laser for DME** DR Severity (judged by investigator) Visual Acuity Lens Status Type of DME (judged by investigator) Fundus Photo (4) (Reading Center) PRP + DR Severity Subretinal Fluid in Central 1 mm* Hard Exudates within 6 mm Intraretinal fluid. Cystoid Edema in Surface Wrinkling Central 1 mm* Retinopathy* Epiretinal Membrane, Center 1 mm Deformed Hemorrhages/ Microaneurysm within 6 mm VMT-Vitreous Attached, Center 1 mm deformed* *Subgroup N <20; the factor will not be analyzed **Patient reported 8

Results 9

Change in VA at 2 Year Visit 10

Final Predictive Factors for Change in VA from Baseline to 2 Years Adjusted for baseline VA, treatment group, and their interaction Predictor Age (each 10 yr increment) R-square = 0. 28 N Model Estimate ETDRS letters (95% CI) P value 564 -2. 1 (-3. 0, -1. 2) <0. 001 11
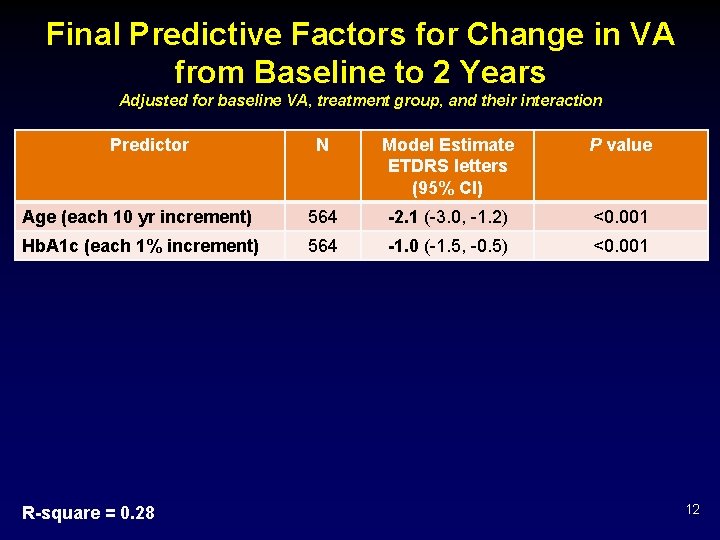
Final Predictive Factors for Change in VA from Baseline to 2 Years Adjusted for baseline VA, treatment group, and their interaction Predictor N Model Estimate ETDRS letters (95% CI) P value Age (each 10 yr increment) 564 -2. 1 (-3. 0, -1. 2) <0. 001 Hb. A 1 c (each 1% increment) 564 -1. 0 (-1. 5, -0. 5) <0. 001 R-square = 0. 28 12
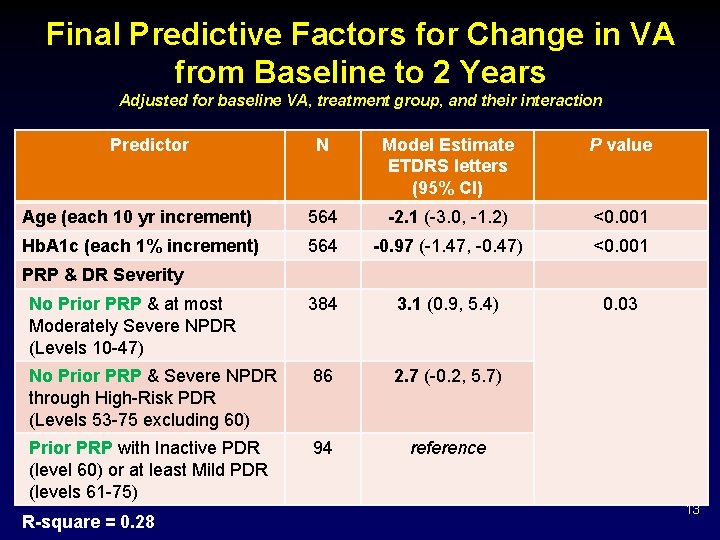
Final Predictive Factors for Change in VA from Baseline to 2 Years Adjusted for baseline VA, treatment group, and their interaction Predictor N Model Estimate ETDRS letters (95% CI) P value Age (each 10 yr increment) 564 -2. 1 (-3. 0, -1. 2) <0. 001 Hb. A 1 c (each 1% increment) 564 -0. 97 (-1. 47, -0. 47) <0. 001 No Prior PRP & at most Moderately Severe NPDR (Levels 10 -47) 384 3. 1 (0. 9, 5. 4) 0. 03 No Prior PRP & Severe NPDR through High-Risk PDR (Levels 53 -75 excluding 60) 86 2. 7 (-0. 2, 5. 7) Prior PRP with Inactive PDR (level 60) or at least Mild PDR (levels 61 -75) 94 reference PRP & DR Severity R-square = 0. 28 13
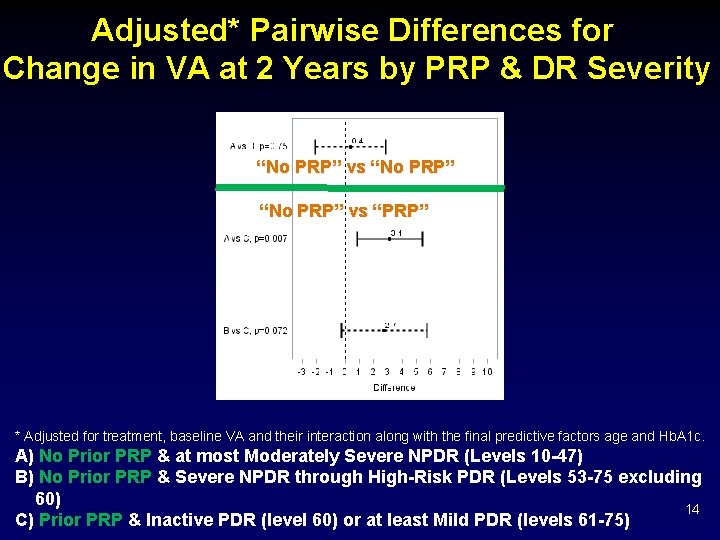
Adjusted* Pairwise Differences for Change in VA at 2 Years by PRP & DR Severity “No PRP” vs “PRP” * Adjusted for treatment, baseline VA and their interaction along with the final predictive factors age and Hb. A 1 c. A) No Prior PRP & at most Moderately Severe NPDR (Levels 10 -47) B) No Prior PRP & Severe NPDR through High-Risk PDR (Levels 53 -75 excluding 60) 14 C) Prior PRP & Inactive PDR (level 60) or at least Mild PDR (levels 61 -75)

Change in VA Through 2 Years Area Under the Curve 15

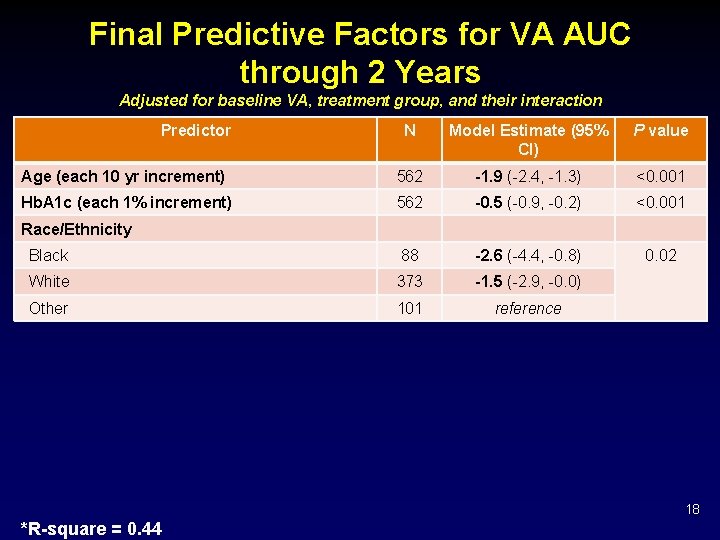
Final Predictive Factors for VA AUC through 2 Years Adjusted for baseline VA, treatment group, and their interaction Predictor Age (each 10 yr increment) N Model Estimate (95% CI) P value 562 -1. 9 (-2. 4, -1. 3) <0. 001 16 *R-square = 0. 44

Final Predictive Factors for VA AUC through 2 Years Adjusted for baseline VA, treatment group, and their interaction Predictor N Model Estimate (95% CI) P value Age (each 10 yr increment) 562 -1. 9 (-2. 4, -1. 3) <0. 001 Hb. A 1 c (each 1% increment) 562 -0. 5 (-0. 9, -0. 2) <0. 001 17 *R-square = 0. 44
Final Predictive Factors for VA AUC through 2 Years Adjusted for baseline VA, treatment group, and their interaction Predictor N Model Estimate (95% CI) P value Age (each 10 yr increment) 562 -1. 9 (-2. 4, -1. 3) <0. 001 Hb. A 1 c (each 1% increment) 562 -0. 5 (-0. 9, -0. 2) <0. 001 Black 88 -2. 6 (-4. 4, -0. 8) 0. 02 White 373 -1. 5 (-2. 9, -0. 0) Other 101 reference Race/Ethnicity 18 *R-square = 0. 44
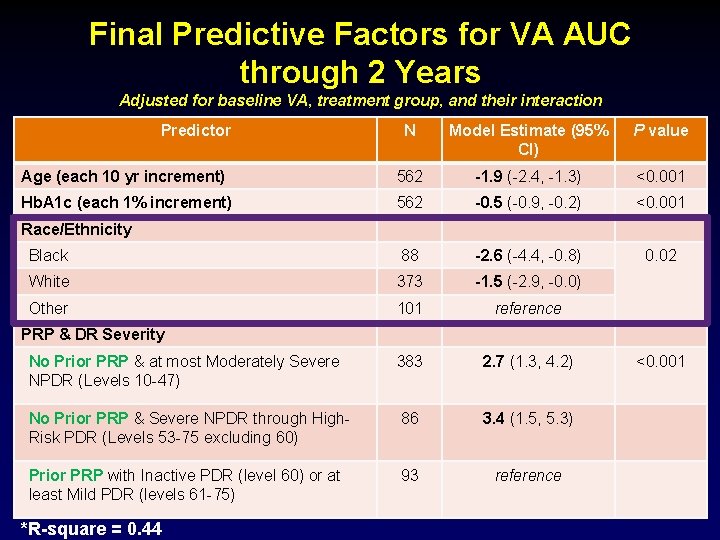
Final Predictive Factors for VA AUC through 2 Years Adjusted for baseline VA, treatment group, and their interaction Predictor N Model Estimate (95% CI) P value Age (each 10 yr increment) 562 -1. 9 (-2. 4, -1. 3) <0. 001 Hb. A 1 c (each 1% increment) 562 -0. 5 (-0. 9, -0. 2) <0. 001 Black 88 -2. 6 (-4. 4, -0. 8) 0. 02 White 373 -1. 5 (-2. 9, -0. 0) Other 101 reference No Prior PRP & at most Moderately Severe NPDR (Levels 10 -47) 383 2. 7 (1. 3, 4. 2) No Prior PRP & Severe NPDR through High. Risk PDR (Levels 53 -75 excluding 60) 86 3. 4 (1. 5, 5. 3) Prior PRP with Inactive PDR (level 60) or at least Mild PDR (levels 61 -75) 93 reference Race/Ethnicity PRP & DR Severity *R-square = 0. 44 <0. 001 19
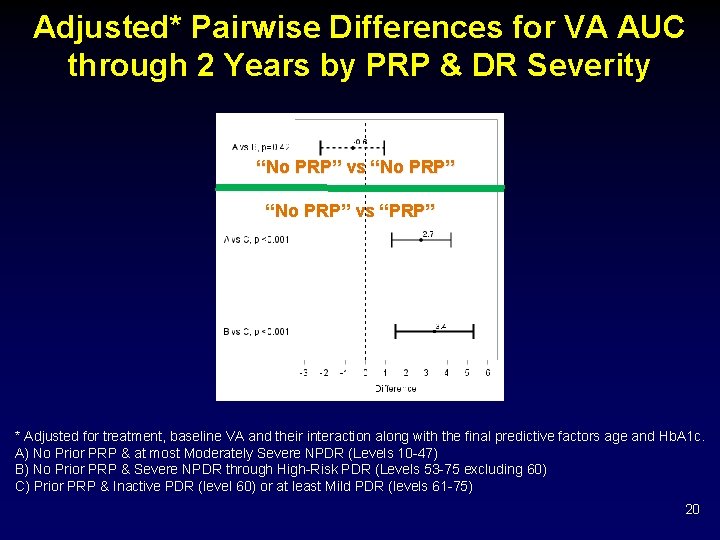
Adjusted* Pairwise Differences for VA AUC through 2 Years by PRP & DR Severity “No PRP” vs “PRP” * Adjusted for treatment, baseline VA and their interaction along with the final predictive factors age and Hb. A 1 c. A) No Prior PRP & at most Moderately Severe NPDR (Levels 10 -47) B) No Prior PRP & Severe NPDR through High-Risk PDR (Levels 53 -75 excluding 60) C) Prior PRP & Inactive PDR (level 60) or at least Mild PDR (levels 61 -75) 20
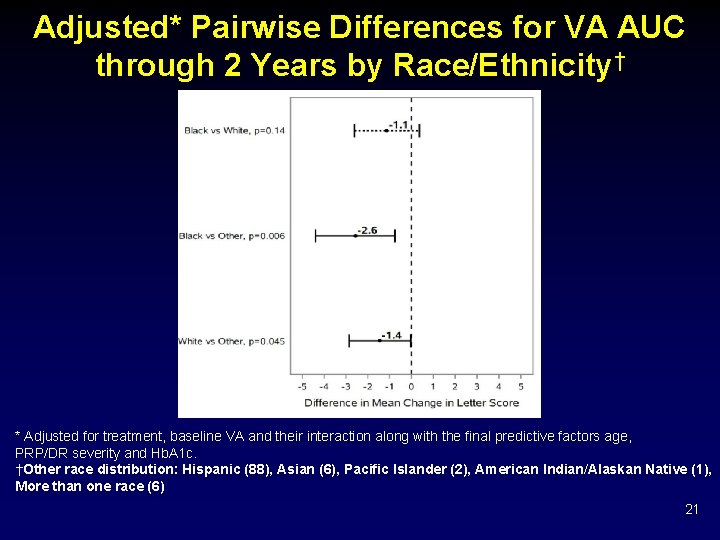
Adjusted* Pairwise Differences for VA AUC through 2 Years by Race/Ethnicity† * Adjusted for treatment, baseline VA and their interaction along with the final predictive factors age, PRP/DR severity and Hb. A 1 c. †Other race distribution: Hispanic (88), Asian (6), Pacific Islander (2), American Indian/Alaskan Native (1), More than one race (6) 21

Change in Central Subfield Thickness at the 2 Year Visit 22
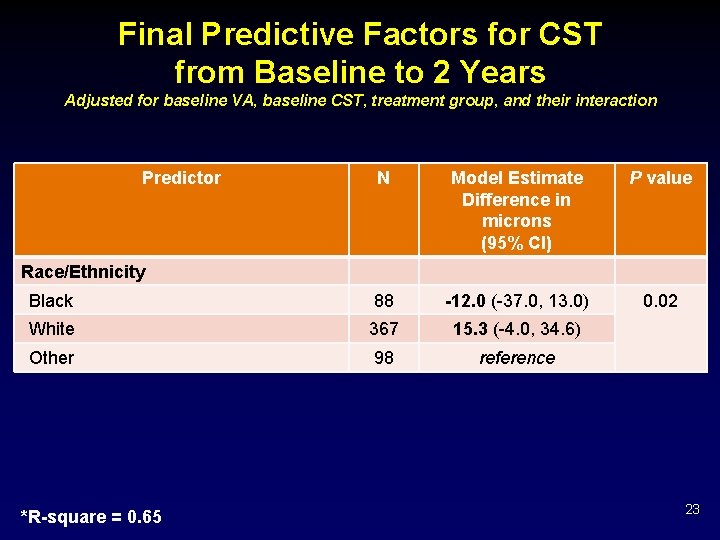
Final Predictive Factors for CST from Baseline to 2 Years Adjusted for baseline VA, baseline CST, treatment group, and their interaction Predictor N Model Estimate Difference in microns (95% CI) P value Black 88 -12. 0 (-37. 0, 13. 0) 0. 02 White 367 15. 3 (-4. 0, 34. 6) Other 98 reference Race/Ethnicity *R-square = 0. 65 23
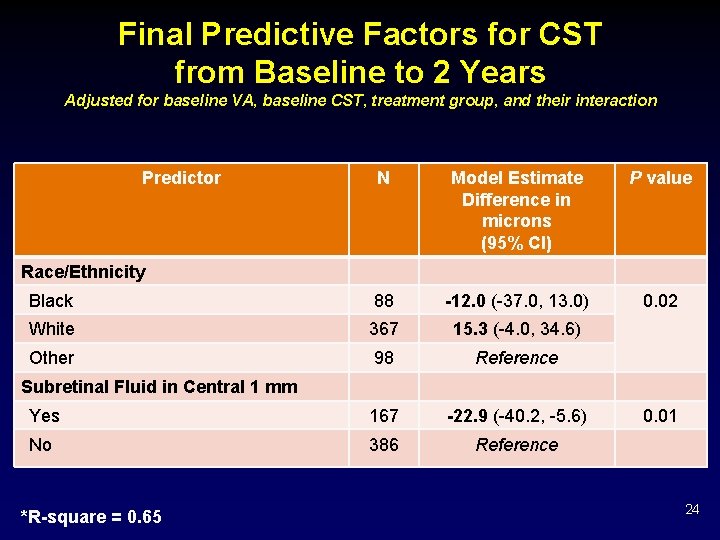
Final Predictive Factors for CST from Baseline to 2 Years Adjusted for baseline VA, baseline CST, treatment group, and their interaction Predictor N Model Estimate Difference in microns (95% CI) P value Black 88 -12. 0 (-37. 0, 13. 0) 0. 02 White 367 15. 3 (-4. 0, 34. 6) Other 98 Reference Yes 167 -22. 9 (-40. 2, -5. 6) No 386 Reference Race/Ethnicity Subretinal Fluid in Central 1 mm *R-square = 0. 65 0. 01 24
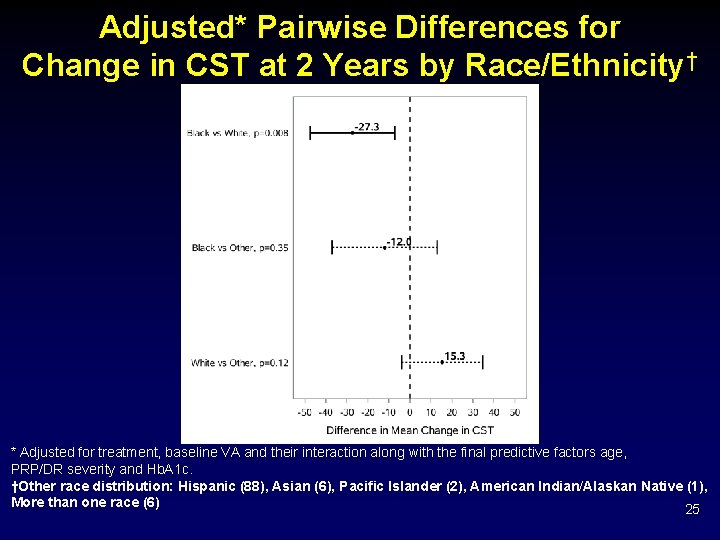
Adjusted* Pairwise Differences for Change in CST at 2 Years by Race/Ethnicity† * Adjusted for treatment, baseline VA and their interaction along with the final predictive factors age, PRP/DR severity and Hb. A 1 c. †Other race distribution: Hispanic (88), Asian (6), Pacific Islander (2), American Indian/Alaskan Native (1), More than one race (6) 25

Summary 26

Final Predictive Factors for More Successful Outcomes Change in VA at 2 -Year 1 Younger age Lower Hb. A 1 C No Prior PRP/and any level of DR (excluding 60) 1 Adjusted for baseline VA, treatment, & their interaction 27

Final Predictive Factors for More Successful Outcomes Change in VA at 2 -Year 1 VA AUC through 2 -Yrs 2 Younger age Younger Age Lower Hb. A 1 C No Prior PRP/and any level of DR (excluding 60) Race: Other* 1 Adjusted for baseline VA, treatment, & 2 Adjusted for baseline VA, CST, treatment and their interaction, *88% Hispanic interaction of CST and treatment 28

Final Predictive Factors for More Successful Outcomes Change in VA at 2 -Year 1 VA AUC through 2 -Yrs 1 Younger age Change in CST at 2 -Yr 2 Younger Age Other*/Black Race/Ethnicity Lower Hb. A 1 C No Prior PRP/and any level of DR (excluding 60) Subretinal Fluid present Race: Other* 1 Adjusted for baseline VA, treatment, & 2 Adjusted for baseline VA, CST, treatment and their interaction, *88% Hispanic interaction of CST and treatment 29

Final Predictive Factors for More Successful Outcomes Change in VA at 2 -Year 1 VA AUC through 2 -Yrs 1 Younger age Change in CST at 2 -Yr 2 Younger Age Other*/Black Race/Ethnicity Lower Hb. A 1 C No Prior PRP/and any level of DR (excluding 60) Subretinal Fluid present Race: Other* No interactions with drug assignment 1 Adjusted for baseline VA, treatment, & 2 Adjusted for baseline VA, CST, treatment and their interaction, *88% Hispanic interaction of CST and treatment 30
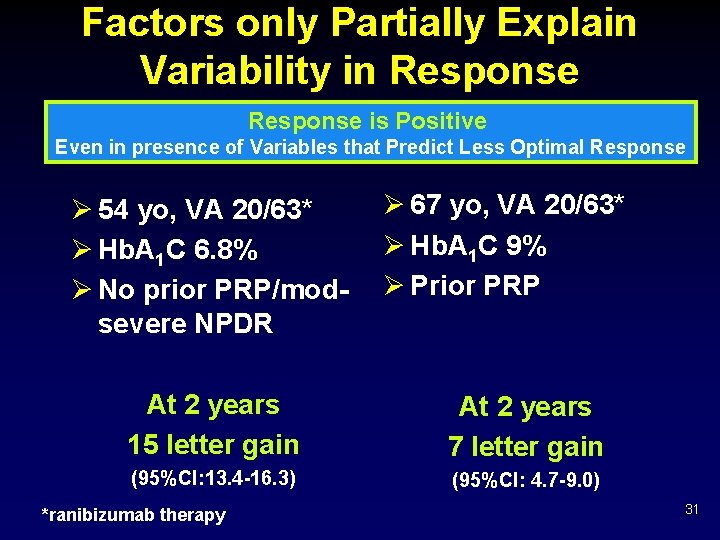
Factors only Partially Explain Variability in Response is Positive Even in presence of Variables that Predict Less Optimal Response Ø 54 yo, VA 20/63* Ø Hb. A 1 C 6. 8% Ø No prior PRP/modsevere NPDR Ø 67 yo, VA 20/63* Ø Hb. A 1 C 9% Ø Prior PRP At 2 years 15 letter gain At 2 years 7 letter gain (95%CI: 13. 4 -16. 3) (95%CI: 4. 7 -9. 0) *ranibizumab therapy 31

Strengths and Limitations of this Analysis Strengths: Ø Prospective, large sample size, standardized measurements at baseline and outcomes Limitations: Ø High number of statistical comparisons increases likelihood of false detection associations Ø As with all post hoc analyses- confidence in the validity of these findings would be enhanced by replication outside of DRCR. net 32
- Slides: 32