Diabetic Retinopathy Clinical Research Network Comparative Effectiveness Study

Diabetic Retinopathy Clinical Research Network Comparative Effectiveness Study of Aflibercept, Bevacizumab, or Ranibizumab for DME Supported through a cooperative agreement from the National Eye Institute; National Institute of Diabetes and Digestive and Kidney Diseases; National Institutes of Health, Department of Health and Human Services EY 14231, EY 14229, EY 018817 1

Disclosure Ø Funding/Support: Cooperative Agreement with NEI and NIDDK of NIH, U. S. Department of Health and Human Services. Ø Additional Contributions: Regeneron Pharmaceuticals, Inc. provided the aflibercept; Genentech Inc. provided the ranibizumab. Genentech Inc. also provided funding for blood pressure cuffs and collection of plasma and urine that are not part of the main study reported herein. Ø A complete list of all DRCR. net investigator financial disclosures can be found at www. drcr. net. 2

Background Ø Diabetic macular edema (DME) affects ~750, 000 people in the U. S. Ø Intravitreous anti-vascular endothelial growth factor (anti-VEGF) injections of either aflibercept (EYLEA), bevacizumab (Avastin), or ranibizumab (Lucentis) are effective in treating DME. Ø The relative efficacy and safety of these agents within a head-to-head study were unknown prior to the results of this trial 3

Background Ø Aflibercept and ranibizumab are FDA approved for DME treatment. Ø Bevacizumab is not FDA approved for intraocular use, § used “off-label” for DME treatment and § repackaged into aliquots ~1/500 of systemic dose in cancer treatments. Ø Medicare allowable charges: § Aflibercept (2. 0 mg): $1961 § Bevacizumab (repackaged 1. 25 mg): $67 § Ranibizumab (0. 3 mg): $1189 4

Primary Objective Ø For eyes with center involved DME with decreased visual acuity, compare one year efficacy and safety of, 1. intravitreous aflibercept (EYLEA®), 2. intravitreous bevacizumab (Avastin®) and 3. intravitreous ranibizumab (Lucentis®)

Study Design Randomized, multi-center clinical trial (N = 89 Sites) Participants meeting all of the following criteria: • At least 18 years old • Type 1 or type 2 diabetes Study eye meeting all of the following criteria: • ~Snellen equivalent visual acuity 20/32 or worse and 20/320 or better • Central-involved DME on clinical exam • Central subfield (CSF) thickness ≥ protocol-defined gender and optical coherence tomography (OCT) machine-specific thresholds • No history of an anti-VEGF treatment for DME in the past 12 months or any other DME treatment in the past 4 months Primary Outcome: Change in visual acuity at one year adjusted for baseline visual acuity using the intent-to-treat principle 6

Pre-Planned Subgroup Analyses Ø Preplanned subgroup analyses included only: • • Baseline visual acuity Baseline OCT Central Subfield Thickness Any Prior Anti-VEGF treatment Lens Status 7
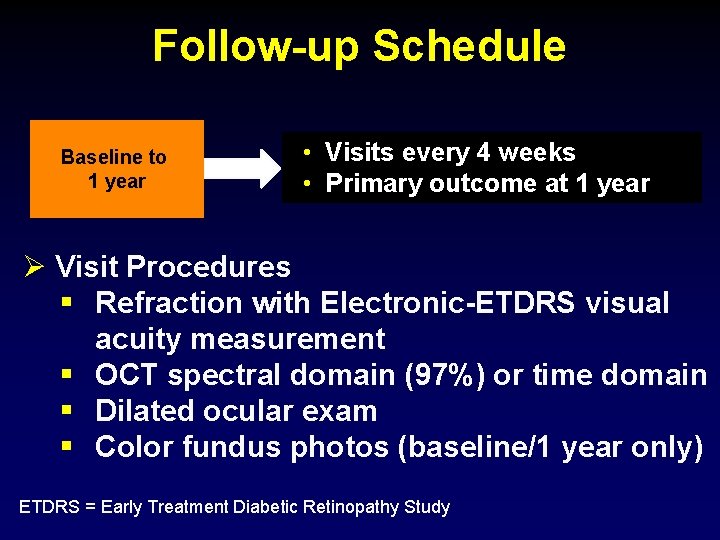
Follow-up Schedule Baseline to 1 year • Visits every 4 weeks • Primary outcome at 1 year Ø Visit Procedures § Refraction with Electronic-ETDRS visual acuity measurement § OCT spectral domain (97%) or time domain § Dilated ocular exam § Color fundus photos (baseline/1 year only) ETDRS = Early Treatment Diabetic Retinopathy Study

Treatment Schedule Through 1 Year (q 4 weeks) Ø Repeat injections at every 4 -week visit if eye “improved”* or “worsened”* Ø Otherwise, defer injections if either: § Visual acuity 20/20 or better and OCT CST “normal” or, § At or after 24 weeks, visual acuity and OCT stable after 2 consecutive injections Ø Resume injections if VA or OCT worsened* *Improved/worsened defined as: ≥ 5 letter change (~1 Snellen line) from last injection, or, ≥ 10% CST change on OCT from last injection

Treatment Schedule Through 1 Year Ø Focal/grid laser: initiated at or after 24 weeks only if persistent DME not improving after at least 2 injections

Randomization Randomly Assigned Eyes (one per participant): N = 660 Baseline Aflibercept (2. 0 mg) N = 224 Bevacizumab (1. 25 mg) N = 218 Ranibizumab (0. 3 mg) N = 218 One Year N = 208 (93%) N = 206 (94%) One Year Excluding Deaths 94% 97% 96% 11
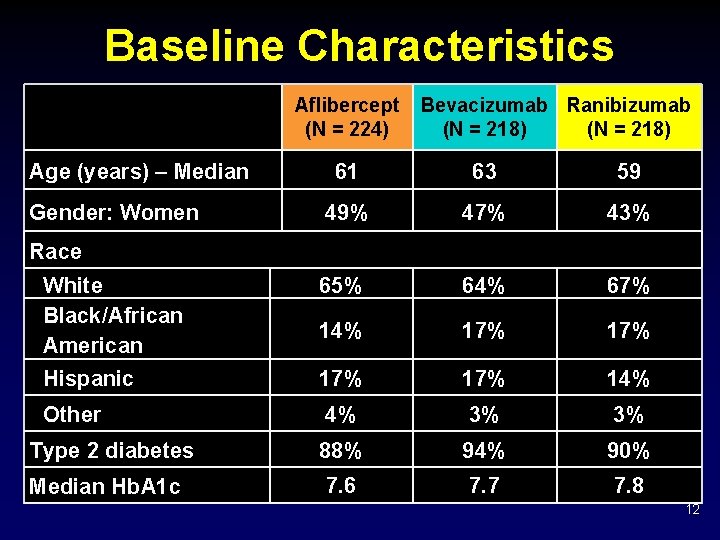
Baseline Characteristics Aflibercept (N = 224) Age (years) – Median Bevacizumab Ranibizumab (N = 218) 61 63 59 49% 47% 43% White Black/African American 65% 64% 67% 14% 17% Hispanic 17% 14% Other 4% 3% 3% Type 2 diabetes 88% 94% 90% Median Hb. A 1 c 7. 6 7. 7 7. 8 Gender: Women Race 12

Ocular Baseline Characteristics Aflibercept Bevacizumab Ranibizumab (N = 224) (N = 218) Mean visual acuity letter score (~Snellen Equivalent) Mean OCT CST (µm) Any Prior Focal/Grid Laser Any Prior Treatment with anti-VEGF Phakic 56 (20/80) 57 (20/80) 387 376 390 36% 39% 37% 11% 14% 13% 74% 73% 79% 13

Ocular Baseline Characteristics Aflibercept Bevacizumab Ranibizumab (N = 224) (N = 218) Diabetic Retinopathy Severity (ETDRS Level)* Absent or minimal NPDR (Level 10 -20) Mild to moderately severe NPDR (Level 35, 43, 47) Severe NPDR (Level 53) Prior PRP; without current PDR (Level 60) Mild to moderate PDR (Level 31 and 65) High Risk PDR (Level 71 and 75) 3% 3% 2% 68% 62% 67% 8% 7% 10% 7% 13% 15% 11% 1% 3% 9% *Missing: aflibercept (3), bevacizumab (7), and ranibizumab (2) 14

Treatment for Diabetic Macular Edema 15
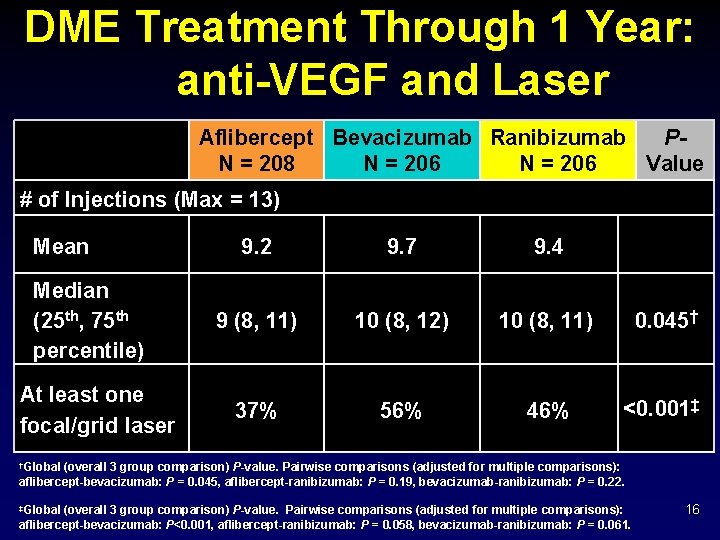
DME Treatment Through 1 Year: anti-VEGF and Laser Aflibercept Bevacizumab Ranibizumab PN = 208 N = 206 Value # of Injections (Max = 13) Mean Median (25 th, 75 th percentile) At least one focal/grid laser 9. 2 9. 7 9. 4 9 (8, 11) 10 (8, 12) 10 (8, 11) 0. 045† 37% 56% 46% <0. 001‡ †Global (overall 3 group comparison) P-value. Pairwise comparisons (adjusted for multiple comparisons): aflibercept-bevacizumab: P = 0. 045, aflibercept-ranibizumab: P = 0. 19, bevacizumab-ranibizumab: P = 0. 22. ‡Global (overall 3 group comparison) P-value. Pairwise comparisons (adjusted for multiple comparisons): aflibercept-bevacizumab: P<0. 001, aflibercept-ranibizumab: P = 0. 058, bevacizumab-ranibizumab: P = 0. 061. 16

Efficacy 17
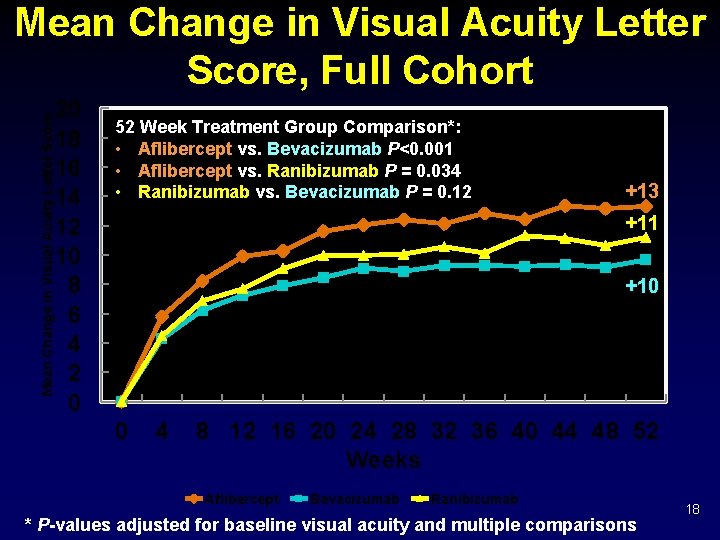
Mean Change in Visual Acuity Letter Score, Full Cohort 20 18 16 14 12 10 8 6 4 2 0 52 Week Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P<0. 001 • Aflibercept vs. Ranibizumab P = 0. 034 • Ranibizumab vs. Bevacizumab P = 0. 12 +13 +11 +10 0 4 8 12 16 20 24 28 32 36 40 44 48 52 Weeks Aflibercept Bevacizumab Ranibizumab * P-values adjusted for baseline visual acuity and multiple comparisons 18

Mean Change in Visual Acuity Letter Score Baseline Visual Acuity 20/32 to 20/40 20 18 16 14 12 10 8 6 4 2 0 ~50% of Cohort ~+8 0 4 8 12 16 20 24 28 32 36 40 44 48 52 Weeks Aflibercept Bevacizumab Ranibizumab 19
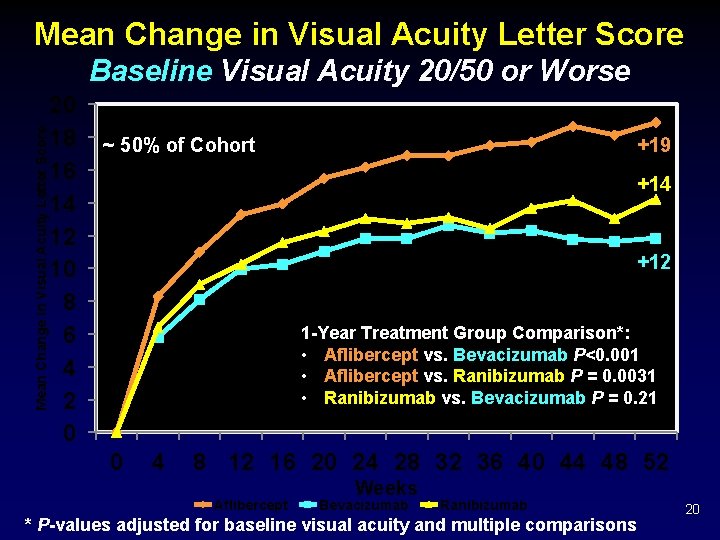
Mean Change in Visual Acuity Letter Score Baseline Visual Acuity 20/50 or Worse 20 18 16 14 12 10 8 6 4 2 0 ~ 50% of Cohort +19 +14 +12 1 -Year Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P<0. 001 • Aflibercept vs. Ranibizumab P = 0. 0031 • Ranibizumab vs. Bevacizumab P = 0. 21 0 4 8 12 16 20 24 28 32 36 40 44 48 52 Aflibercept Weeks Bevacizumab Ranibizumab * P-values adjusted for baseline visual acuity and multiple comparisons 20
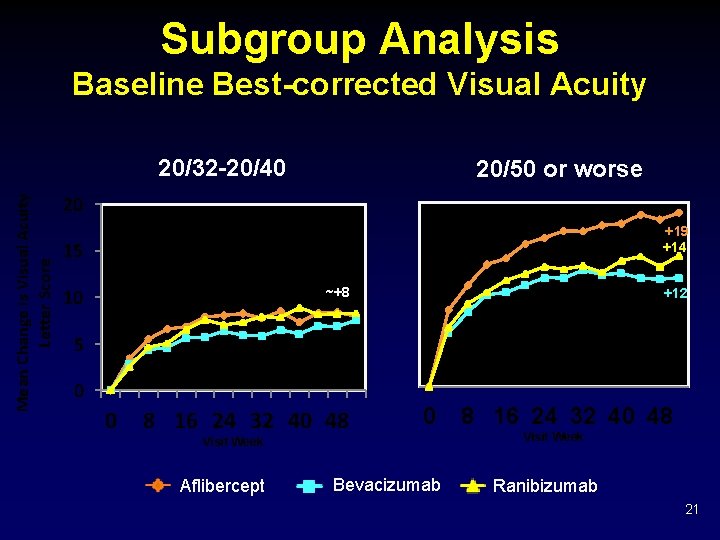
Subgroup Analysis Baseline Best-corrected Visual Acuity Mean Change is Visual Acuity Letter Score 20/32 -20/40 20/50 or worse 20 +19 +14 15 ~+8 10 +12 5 0 0 8 16 24 32 40 48 0 Visit Week Aflibercept Bevacizumab 8 16 24 32 40 48 Visit Week Ranibizumab 21
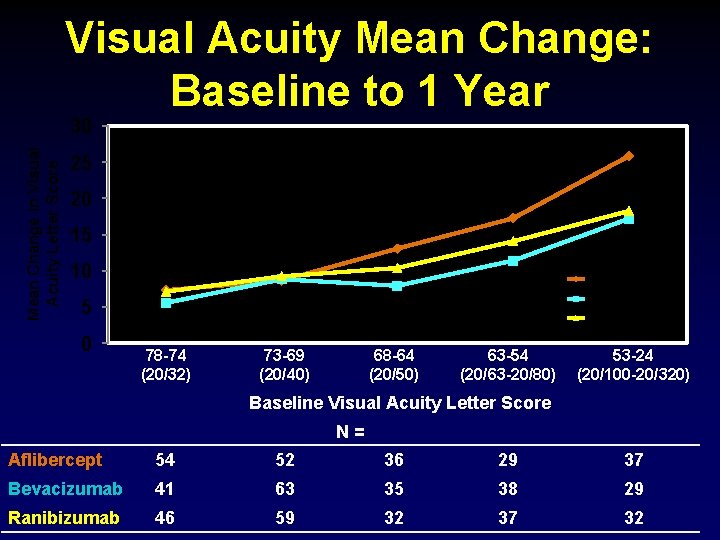
Visual Acuity Mean Change: Baseline to 1 Year Mean Change in Visual Acuity Letter Score 30 25 20 15 10 Aflibercept Bevacizumab 5 0 Ranibizumab 78 -74 (20/32) 73 -69 (20/40) 68 -64 (20/50) 63 -54 (20/63 -20/80) 53 -24 (20/100 -20/320) Baseline Visual Acuity Letter Score N = Aflibercept 54 52 36 29 37 Bevacizumab 41 63 35 38 29 Ranibizumab 46 59 32 37 32
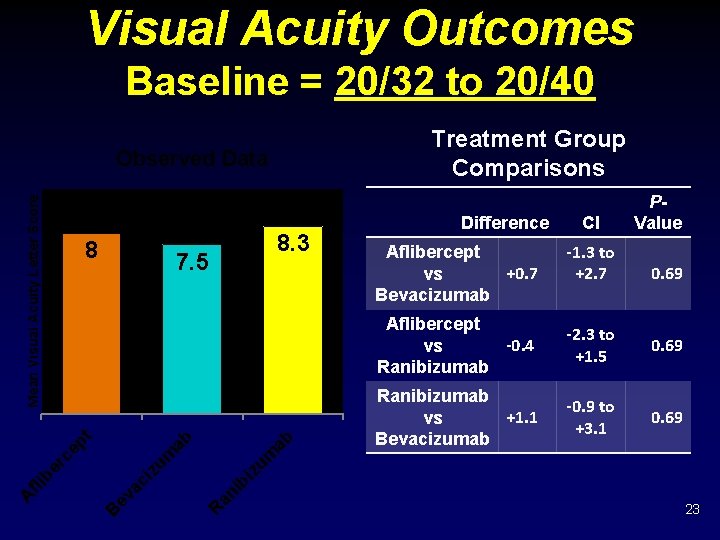
Visual Acuity Outcomes Baseline = 20/32 to 20/40 Treatment Group Comparisons 8 ab ab Aflibercept +0. 7 vs Bevacizumab CI -1. 3 to +2. 7 0. 69 Aflibercept -0. 4 vs Ranibizumab -2. 3 to +1. 5 0. 69 Ranibizumab +1. 1 vs Bevacizumab -0. 9 to +3. 1 0. 69 bi an i ev ac i B Difference P- Value zu m ep t er c fli b A 8. 3 7. 5 R Mean Visual Acuity Letter Score Observed Data 23
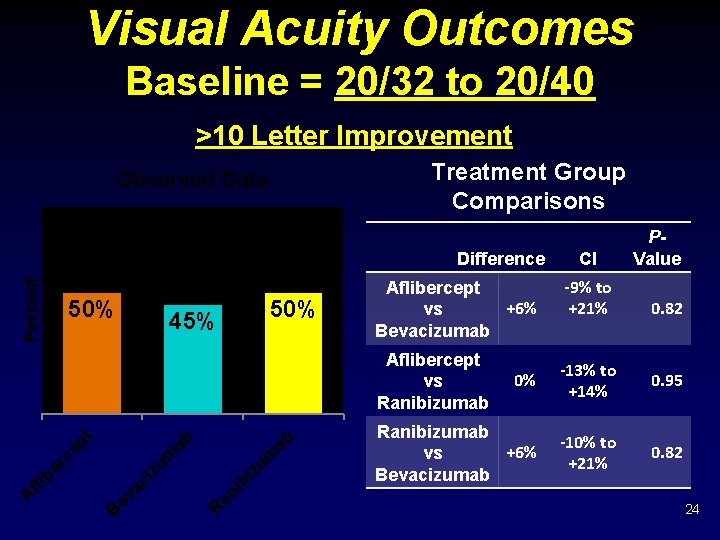
Visual Acuity Outcomes Baseline = 20/32 to 20/40 >10 Letter Improvement Treatment Group Comparisons Observed Data Percent Difference 50% 45% Aflibercept +6% vs Bevacizumab R an i bi zu m ab ab zu m B ev ac i A fli b er ce pt Aflibercept vs Ranibizumab CI -9% to +21% P- Value 0. 82 0% -13% to +14% 0. 95 Ranibizumab +6% vs Bevacizumab -10% to +21% 0. 82 24

Visual Acuity Outcomes Baseline = 20/32 to 20/40 >15 Letter Improvement Treatment Group Comparisons Observed Data Difference Percent Aflibercept +2% vs Bevacizumab 16% 15% R an i bi zu m ab ab zu m B ev ac i A fli b er c ep t 18% Aflibercept vs Ranibizumab +4% Ranibizumab -2% vs Bevacizumab CI -7% to +11% P- Value 0. 73 -5% to +12% 0. 73 -10% to +7% 0. 73 25
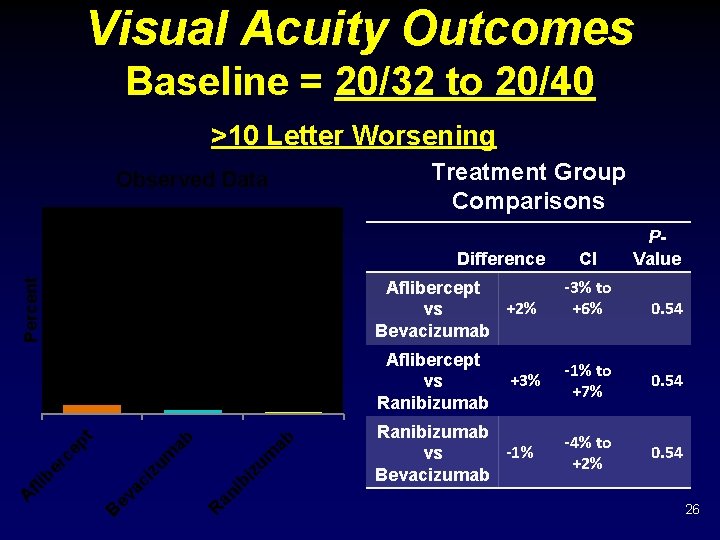
Visual Acuity Outcomes Baseline = 20/32 to 20/40 >10 Letter Worsening Treatment Group Comparisons Observed Data Difference Percent Aflibercept +2% vs Bevacizumab 2% 1% R an i bi zu m ab ab zu m B ev ac i A fli b er ce pt 4% Aflibercept vs Ranibizumab +3% Ranibizumab -1% vs Bevacizumab CI -3% to +6% P- Value 0. 54 -1% to +7% 0. 54 -4% to +2% 0. 54 26

Visual Acuity Outcomes Baseline = 20/50 or Worse Treatment Group Comparisons 18. 9 14. 2 ab ab Aflibercept +6. 5 vs Bevacizumab CI P- Value +2. 9 to +10. 1 <0. 001 Aflibercept +4. 7 vs Ranibizumab +1. 4 to 0. 0031 +8. 0 Ranibizumab +1. 8 vs Bevacizumab -1. 1 to +4. 8 0. 21 R an i bi zu m ev ac i B fli b A Difference 11. 8 er ce pt Mean Visual Acuity Letter Score Observed Data 27
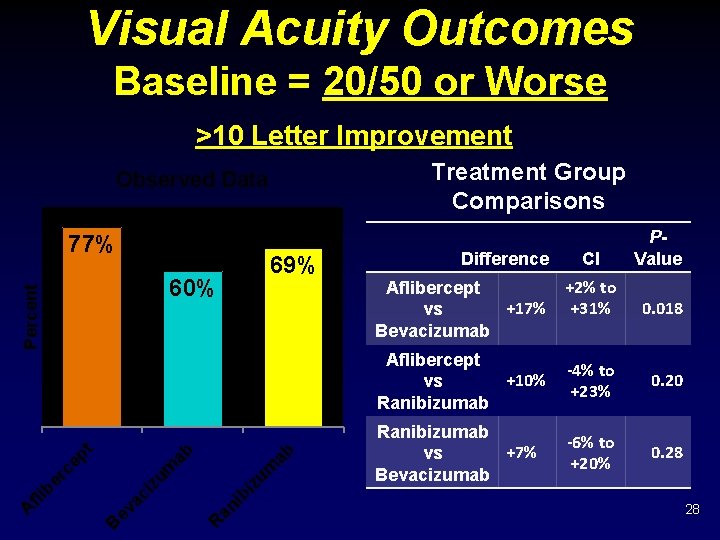
Visual Acuity Outcomes Baseline = 20/50 or Worse >10 Letter Improvement Treatment Group Comparisons Observed Data 77% 60% ab iz um ib an R B ev ac i er c zu m ep t ab Percent fli b A 69% Difference Aflibercept +17% vs Bevacizumab CI P- Value +2% to +31% 0. 018 Aflibercept +10% vs Ranibizumab -4% to +23% 0. 20 Ranibizumab +7% vs Bevacizumab -6% to +20% 0. 28 28
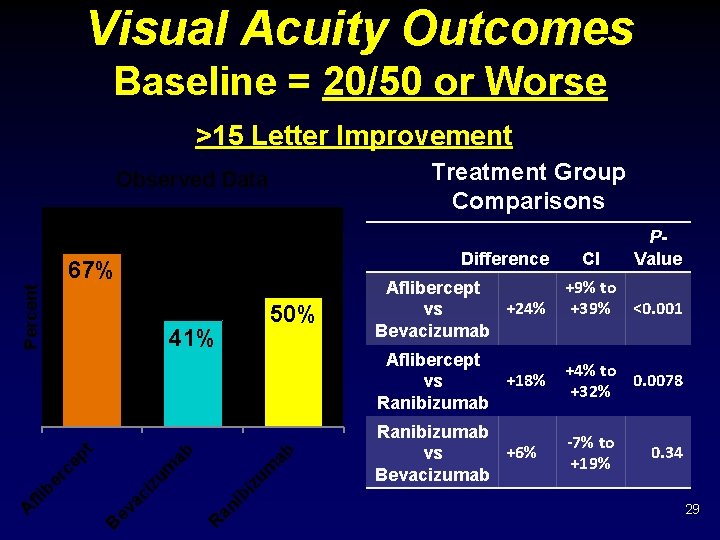
Visual Acuity Outcomes Baseline = 20/50 or Worse >15 Letter Improvement Treatment Group Comparisons Observed Data Difference Percent 67% ab ib an R ev ac i B iz um zu m ep t ab 41% er c fli b A 50% Aflibercept +24% vs Bevacizumab CI P- Value +9% to +39% <0. 001 Aflibercept +18% vs Ranibizumab +4% to 0. 0078 +32% Ranibizumab +6% vs Bevacizumab -7% to +19% 0. 34 29
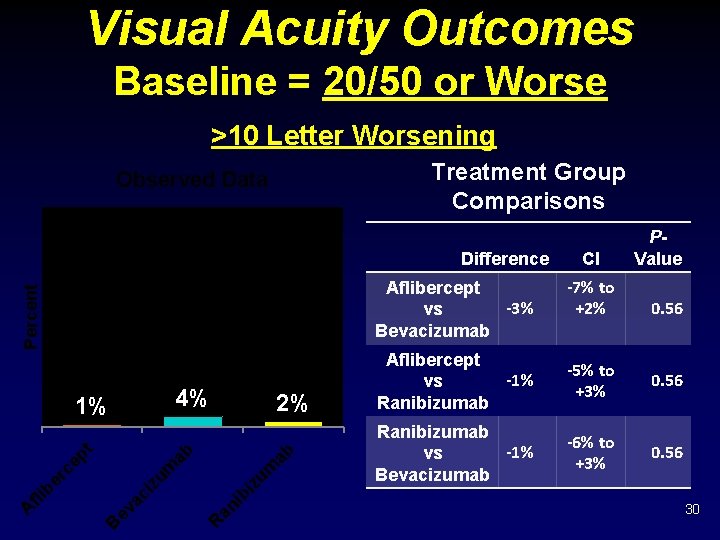
Visual Acuity Outcomes Baseline = 20/50 or Worse >10 Letter Worsening Treatment Group Comparisons Observed Data Difference Percent Aflibercept -3% vs Bevacizumab 4% ab ib an R ev ac i B iz um zu m ep t er c fli b A 2% ab 1% CI -7% to +2% P- Value 0. 56 Aflibercept -1% vs Ranibizumab -5% to +3% 0. 56 Ranibizumab -1% vs Bevacizumab -6% to +3% 0. 56 30
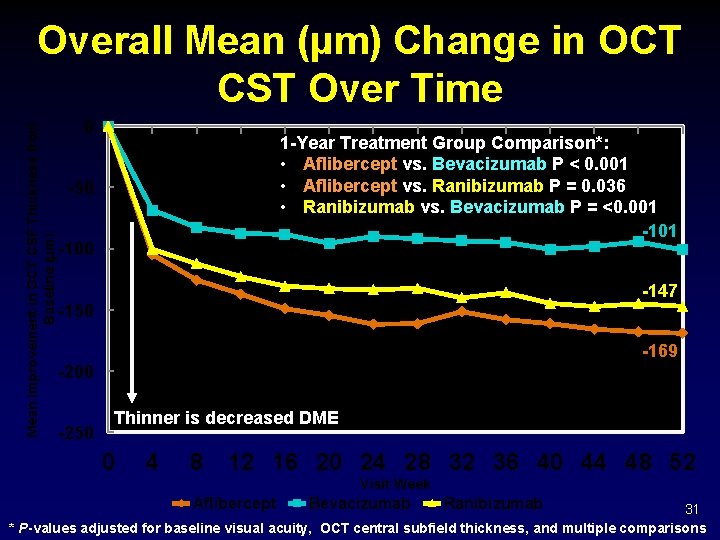
Mean Improvement in OCT CSF Thickness from Baseline (µm) Overall Mean (µm) Change in OCT CST Over Time 0 1 -Year Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P < 0. 001 • Aflibercept vs. Ranibizumab P = 0. 036 • Ranibizumab vs. Bevacizumab P = <0. 001 -101 -50 -100 -147 -150 -169 -200 -250 Thinner is decreased DME 0 4 8 12 16 20 24 28 32 36 40 44 48 52 Visit Week Aflibercept Bevacizumab Ranibizumab 31 * P-values adjusted for baseline visual acuity, OCT central subfield thickness, and multiple comparisons
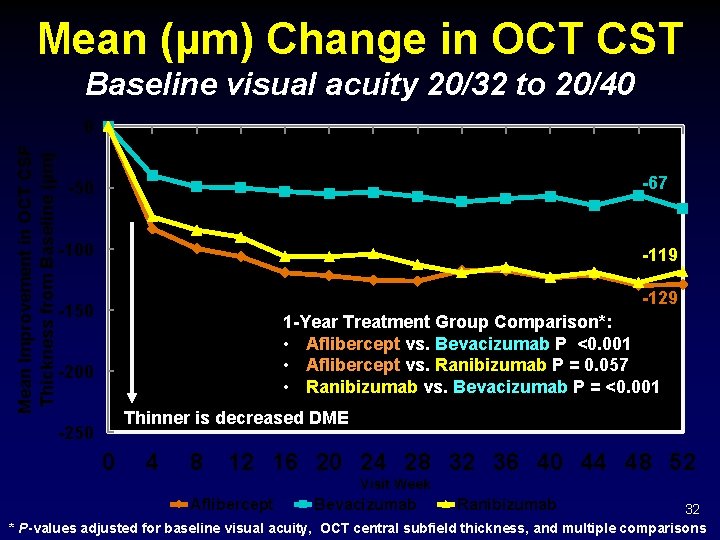
Mean (µm) Change in OCT CST Baseline visual acuity 20/32 to 20/40 Mean Improvement in OCT CSF Thickness from Baseline (µm) 0 -67 -50 -100 -119 -129 -150 1 -Year Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P <0. 001 • Aflibercept vs. Ranibizumab P = 0. 057 • Ranibizumab vs. Bevacizumab P = <0. 001 -200 Thinner is decreased DME -250 0 4 8 12 16 20 24 28 32 36 40 44 48 52 Visit Week Aflibercept Bevacizumab Ranibizumab 32 * P-values adjusted for baseline visual acuity, OCT central subfield thickness, and multiple comparisons
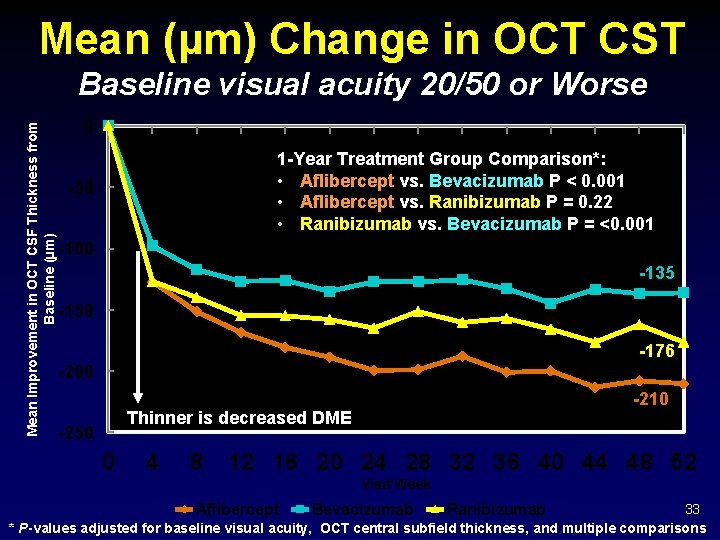
Mean (µm) Change in OCT CST Mean Improvement in OCT CSF Thickness from Baseline (µm) Baseline visual acuity 20/50 or Worse 0 1 -Year Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P < 0. 001 • Aflibercept vs. Ranibizumab P = 0. 22 • Ranibizumab vs. Bevacizumab P = <0. 001 -50 -100 -135 -150 -176 -200 -210 Thinner is decreased DME -250 0 4 8 12 16 20 24 28 32 36 40 44 48 52 Visit Week Aflibercept Bevacizumab Ranibizumab 33 * P-values adjusted for baseline visual acuity, OCT central subfield thickness, and multiple comparisons

OCT CSF Outcomes Baseline = 20/32 -20/40 -119 Aflibercept -55. 8 vs Bevacizumab CI -78. 3 to -32. 9 P- Value <0. 001 -36. 9 to +0. 6 0. 057 Ranibizumab -37. 7 vs Bevacizumab -59. 3 to -16. 1 <0. 001 bi Aflibercept -18. 2 vs Ranibizumab an i R ev ac i B zu m ab ab -129 er c fli b A Difference -67 ep t Mean OCT CSF Change Observed Data Treatment Group Comparisons 34
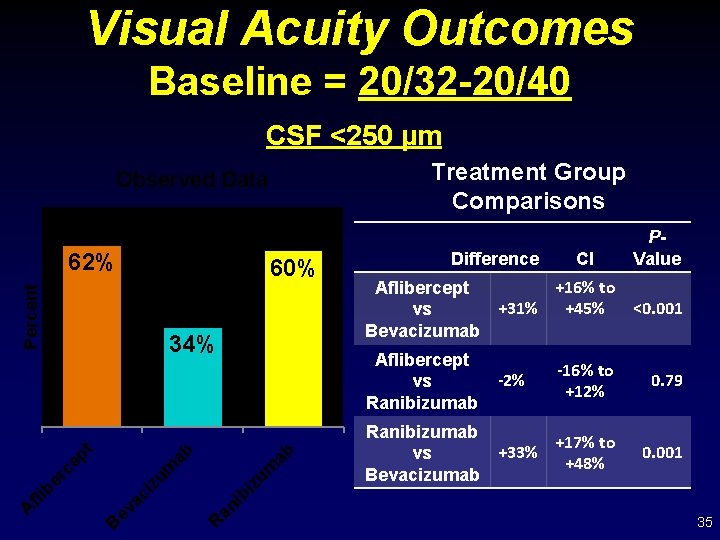
Visual Acuity Outcomes Baseline = 20/32 -20/40 CSF <250 µm Treatment Group Comparisons Observed Data 62% Percent 60% 34% ab iz um -16% to +12% 0. 79 Ranibizumab +17% to +33% vs +48% Bevacizumab 0. 001 -2% ib an R ev ac i B CI +16% to Aflibercept +31% +45% <0. 001 vs Bevacizumab Aflibercept vs Ranibizumab ab zu m ep t er c fli b A Difference P- Value 35

OCT CSF Outcomes Baseline = 20/50 or Worse Difference -135 -176 CI P- Value -121. 9 to 49. 7 <0. 001 Aflibercept vs Bevacizumab -85. 8 Aflibercept vs Ranibizumab -18. 5 -48. 2 to +11. 2 0. 22 Ranibizumab vs Bevacizumab -67. 3 -101. 4 to -33. 1 <0. 001 R an i bi zu m B ev ac i er c fli b A ab ab -210 ep t Mean OCT CSF Change Observed Data Treatment Group Comparisons 36
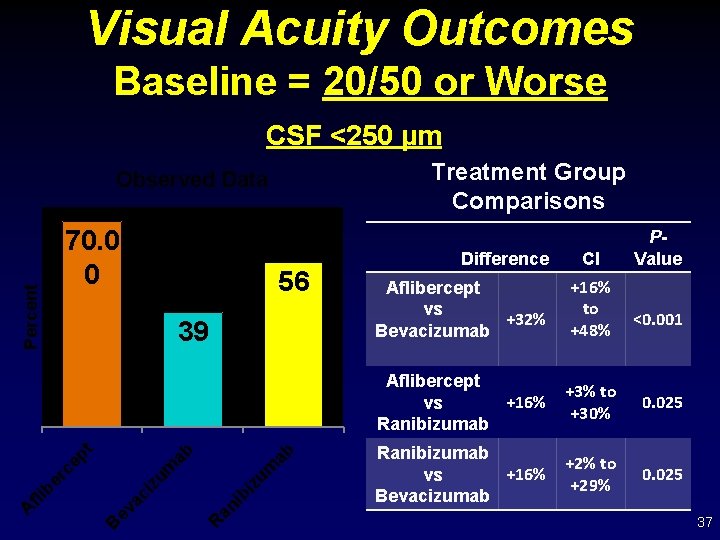
Visual Acuity Outcomes Baseline = 20/50 or Worse CSF <250 µm Treatment Group Comparisons 70. 0 0 ab ab R an ib iz um zu m B ev ac i er c fli b A 56 39 ep t Percent Observed Data Difference CI P- Value Aflibercept vs +32% Bevacizumab +16% to +48% Aflibercept +16% vs Ranibizumab +3% to +30% 0. 025 Ranibizumab +16% vs Bevacizumab +2% to +29% 0. 025 <0. 001 37

Safety 38
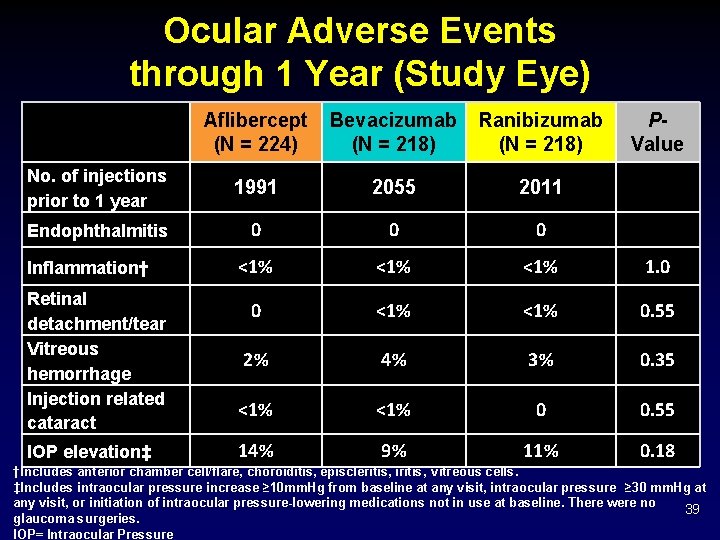
Ocular Adverse Events through 1 Year (Study Eye) Aflibercept (N = 224) Bevacizumab (N = 218) Ranibizumab (N = 218) No. of injections prior to 1 year 1991 2055 2011 Endophthalmitis 0 0 0 <1% <1% 1. 0 0 <1% 0. 55 2% 4% 3% 0. 35 <1% 0 0. 55 14% 9% 11% 0. 18 Inflammation† Retinal detachment/tear Vitreous hemorrhage Injection related cataract IOP elevation‡ PValue †Includes anterior chamber cell/flare, choroiditis, episcleritis, iritis, vitreous cells. ‡Includes intraocular pressure increase ≥ 10 mm. Hg from baseline at any visit, intraocular pressure ≥ 30 mm. Hg at any visit, or initiation of intraocular pressure-lowering medications not in use at baseline. There were no 39 glaucoma surgeries. IOP= Intraocular Pressure
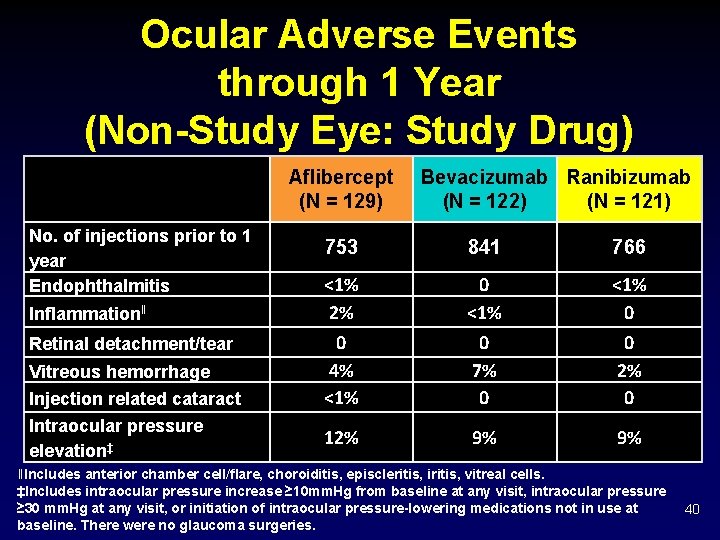
Ocular Adverse Events through 1 Year (Non-Study Eye: Study Drug) Aflibercept (N = 129) No. of injections prior to 1 year Endophthalmitis Inflammationǁ Retinal detachment/tear Vitreous hemorrhage Injection related cataract Intraocular pressure elevation‡ Bevacizumab Ranibizumab (N = 122) (N = 121) 753 841 766 <1% 2% 0 <1% 0 0 4% <1% 0 7% 0 0 2% 0 12% 9% 9% ǁIncludes anterior chamber cell/flare, choroiditis, episcleritis, iritis, vitreal cells. ‡Includes intraocular pressure increase ≥ 10 mm. Hg from baseline at any visit, intraocular pressure ≥ 30 mm. Hg at any visit, or initiation of intraocular pressure-lowering medications not in use at baseline. There were no glaucoma surgeries. 40
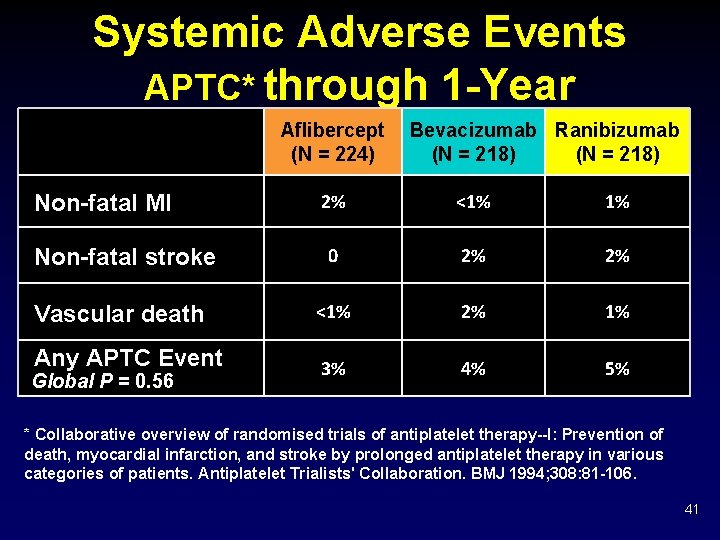
Systemic Adverse Events APTC* through 1 -Year Aflibercept (N = 224) Bevacizumab Ranibizumab (N = 218) 2% <1% 1% Non-fatal stroke 0 2% 2% Vascular death <1% 2% 1% Any APTC Event 3% 4% 5% Non-fatal MI Global P = 0. 56 * Collaborative overview of randomised trials of antiplatelet therapy--I: Prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients. Antiplatelet Trialists' Collaboration. BMJ 1994; 308: 81 -106. 41
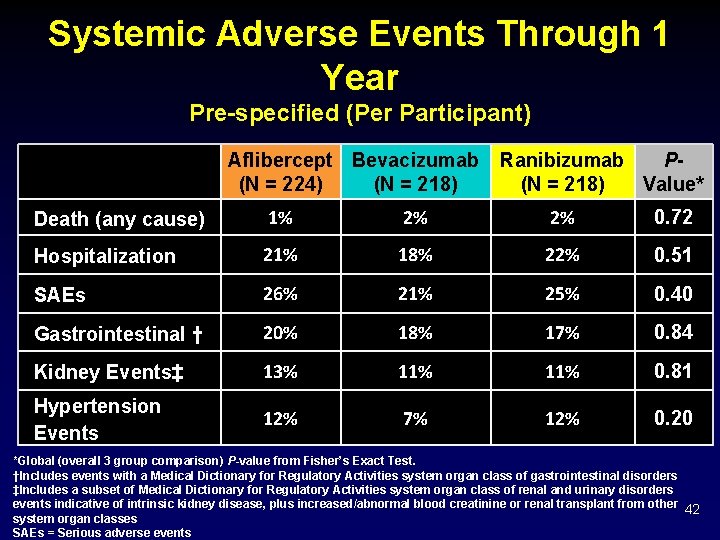
Systemic Adverse Events Through 1 Year Pre-specified (Per Participant) Aflibercept Bevacizumab (N = 224) (N = 218) Ranibizumab P(N = 218) Value* Death (any cause) 1% 2% 2% 0. 72 Hospitalization 21% 18% 22% 0. 51 SAEs 26% 21% 25% 0. 40 Gastrointestinal † 20% 18% 17% 0. 84 Kidney Events‡ 13% 11% 0. 81 Hypertension Events 12% 7% 12% 0. 20 *Global (overall 3 group comparison) P-value from Fisher’s Exact Test. †Includes events with a Medical Dictionary for Regulatory Activities system organ class of gastrointestinal disorders ‡Includes a subset of Medical Dictionary for Regulatory Activities system organ class of renal and urinary disorders events indicative of intrinsic kidney disease, plus increased/abnormal blood creatinine or renal transplant from other 42 system organ classes SAEs = Serious adverse events
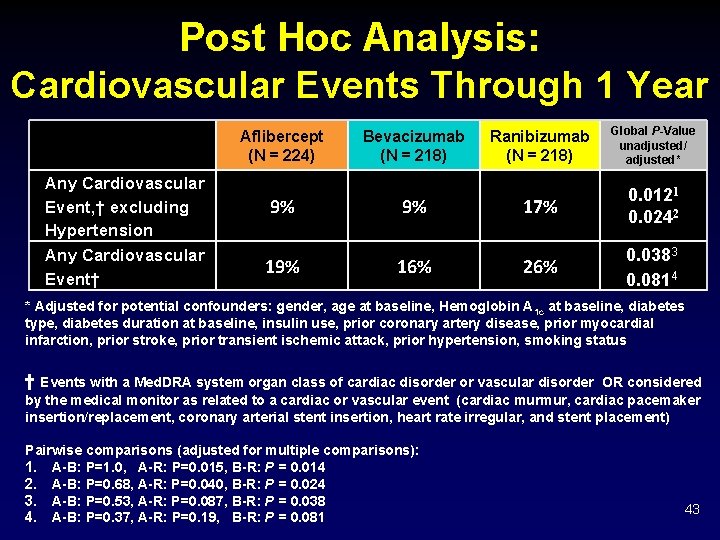
Post Hoc Analysis: Cardiovascular Events Through 1 Year Aflibercept (N = 224) Any Cardiovascular Event, † excluding Hypertension Any Cardiovascular Event† 9% 19% Bevacizumab (N = 218) 9% 16% Ranibizumab (N = 218) Global P-Value unadjusted/ adjusted* 17% 0. 0121 0. 0242 26% 0. 0383 0. 0814 * Adjusted for potential confounders: gender, age at baseline, Hemoglobin A 1 c at baseline, diabetes type, diabetes duration at baseline, insulin use, prior coronary artery disease, prior myocardial infarction, prior stroke, prior transient ischemic attack, prior hypertension, smoking status † Events with a Med. DRA system organ class of cardiac disorder or vascular disorder OR considered by the medical monitor as related to a cardiac or vascular event (cardiac murmur, cardiac pacemaker insertion/replacement, coronary arterial stent insertion, heart rate irregular, and stent placement) Pairwise comparisons (adjusted for multiple comparisons): 1. A-B: P=1. 0, A-R: P=0. 015, B-R: P = 0. 014 2. A-B: P=0. 68, A-R: P=0. 040, B-R: P = 0. 024 3. A-B: P=0. 53, A-R: P=0. 087, B-R: P = 0. 038 4. A-B: P=0. 37, A-R: P=0. 19, B-R: P = 0. 081 43
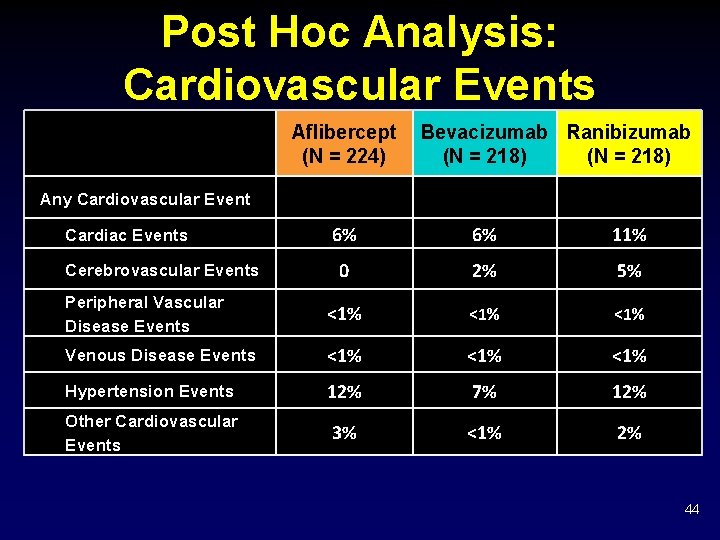
Post Hoc Analysis: Cardiovascular Events Aflibercept (N = 224) Bevacizumab Ranibizumab (N = 218) Any Cardiovascular Event 6% 6% 11% 0 2% 5% Peripheral Vascular Disease Events <1% <1% Venous Disease Events <1% <1% Hypertension Events 12% 7% 12% Other Cardiovascular Events 3% <1% 2% Cardiac Events Cerebrovascular Events 44

Discussion Ø All three anti-VEGF agents, on average, produced substantial visual acuity improvement by 1 month, sustained through 1 year. Ø On average, greater improvement occurred with aflibercept, but relative effect varied by initial visual acuity. § Mild initial vision loss (20/32 -20/40, 50% of study eyes): little difference in mean visual acuity at 1 year § Worse initial vision loss: aflibercept had an advantage over the other agents • Statistically significant: Mean improvement of 18. 9 for aflibercept vs. 11. 8 for bevacizumab (P<0. 001) and vs. 14. 2 with ranibizumab (P = 0. 003) • Clinically meaningful: For example, relative improvement ≥ 15 letters (>3 Snellen lines) in 63% more aflibercept-treated eyes than bevacizumabtreated eyes, and 34% more than ranibizumab-treated eyes 45

Discussion Ø Bevacizumab had a lesser effect on reducing macular edema than the other two agents, regardless of starting acuity. Ø Few eyes in any group had substantial visual acuity loss. Ø Median number of injections: 9 to 10 in all three groups. Ø Fewer eyes in the aflibercept group received focal/grid laser for DME after 24 weeks, presumably because a greater % of eyes in the aflibercept group had resolution of central DME (which drives decision to apply laser). 46

Discussion Ø Serious adverse event, death, and hospitalization rates appeared similar among treatment groups. Ø Significant differences in frequencies of major cardiovascular events were not identified • However, post-hoc analysis combining Med. DRA system organ classes of cardiac and vascular resulted in more participants in the ranibizumab group reporting these adverse events. • This is inconsistent with prior studies and may be due to chance. Ø Endophthalmitis was rare: 0. 02% of injections. Ø No differences in intraocular inflammation. 47

Discussion Ø Bevacizumab: § Note: a central pharmacy repackaged into single use vials • Testing was completed for sterility, purity, and potency, a standard that may not be available in a clinical practice setting ØResults may not apply to eyes with persistent or recurrent DME already receiving anti-VEGF 48

Conclusion Ø All three anti-VEGF agents are effective treatments for DME causing vision impairment. Ø When initial visual acuity loss is mild, on average there is little difference in visual acuity at 1 -year. Ø At worse levels of initial visual acuity aflibercept is more effective at improving vision. 49

Thank You on Behalf of Diabetic Retinopathy Clinical Research Network (DRCR. net) A complete list of all DRCR. net investigator financial disclosures and these slides can be found at www. drcr. net. Full protocol available on clinical. Trials. gov (NCT 01627249) Reference: DOI: 10. 1056/NEJMoa 1414264 Diabetic Retinopathy Clinical Research Network. Aflibercept, ranibizumab, or bevacizumab for diabetic macular edema. NEJM 2015; 372: 1193 -1203. 50
- Slides: 50