Diabetic Retinopathy Clinical Research Network Aflibercept Bevacizumab or

Diabetic Retinopathy Clinical Research Network Aflibercept, Bevacizumab, or Ranibizumab for DME: Two-year Results Supported through a cooperative agreement from the National Eye Institute; National Institute of Diabetes and Digestive and Kidney Diseases; National Institutes of Health, Department of Health and Human Services EY 14231, EY 14229, EY 018817 1

Disclosure Ø Funding/Support: Cooperative Agreement with NEI and NIDDK of NIH, U. S. Department of Health and Human Services. Ø Additional Contributions: Regeneron Pharmaceuticals, Inc. provided the aflibercept; Genentech Inc. provided the ranibizumab. Genentech Inc. also provided funding for blood pressure cuffs and collection of plasma and urine that are not part of the main study reported herein. 2

Background Ø DRCR. net trial: Comparative effectiveness of aflibercept, bevacizumab, and ranibizumab for center-involved diabetic macular edema (DME) associated with vision impairment. § At 1 -year: all agents, on average, improved VA. Relative treatment effect on VA depended on initial visual acuity or OCT central subfield thickness (CST) • • § When baseline VA impairment or OCT CST was mild, no differences on average were identified At worse levels of baseline VA impairment or thicker OCT CST, aflibercept more effective No statistical differences in pre-specified ocular/systemic safety outcomes 3

Study Design Randomized, multi-center clinical trial (89 Sites) Participants meeting all of the following criteria: • At least 18 years old • Type 1 or type 2 diabetes Study eye meeting all of the following criteria: • ~Snellen equivalent visual acuity 20/32 or worse and 20/320 or better • Central-involved DME on clinical exam • Central subfield (CSF) thickness ≥ protocol-defined gender and optical coherence tomography (OCT) machine-specific thresholds • No history of an anti-VEGF treatment for DME in the past 12 months or any other DME treatment in the past 4 months 4
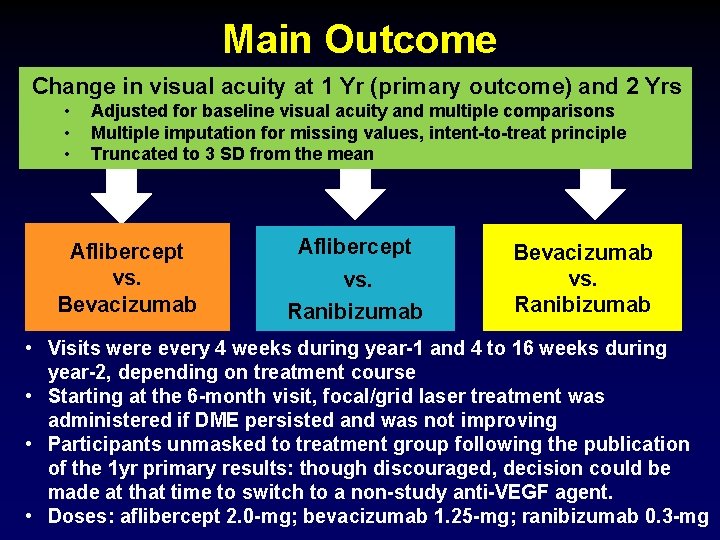
Main Outcome Change in visual acuity at 1 Yr (primary outcome) and 2 Yrs • • • Adjusted for baseline visual acuity and multiple comparisons Multiple imputation for missing values, intent-to-treat principle Truncated to 3 SD from the mean Aflibercept vs. Bevacizumab Aflibercept vs. Ranibizumab Bevacizumab vs. Ranibizumab • Visits were every 4 weeks during year-1 and 4 to 16 weeks during year-2, depending on treatment course • Starting at the 6 -month visit, focal/grid laser treatment was administered if DME persisted and was not improving • Participants unmasked to treatment group following the publication of the 1 yr primary results: though discouraged, decision could be made at that time to switch to a non-study anti-VEGF agent. • Doses: aflibercept 2. 0 -mg; bevacizumab 1. 25 -mg; ranibizumab 0. 3 -mg
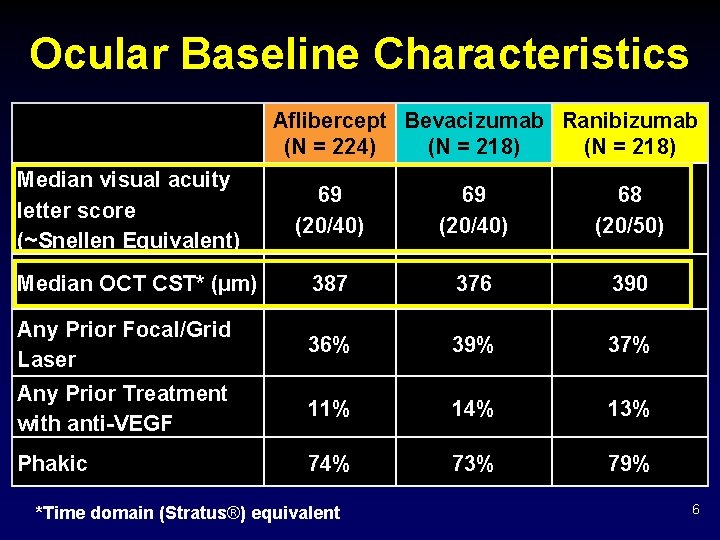
Ocular Baseline Characteristics Aflibercept Bevacizumab Ranibizumab (N = 224) (N = 218) Median visual acuity letter score (~Snellen Equivalent) 69 (20/40) 68 (20/50) Median OCT CST* (µm) 387 376 390 Any Prior Focal/Grid Laser 36% 39% 37% Any Prior Treatment with anti-VEGF 11% 14% 13% Phakic 74% 73% 79% *Time domain (Stratus®) equivalent 6

Randomization Randomly Assigned Eyes (one per participant): N = 660 Aflibercept Bevacizumab Ranibizumab (2. 0 mg/0. 05 m. L) (1. 25 mg/0. 05 m. L) (0. 3 mg/0. 05 m. L) N = 224 N = 218 1 -year (excluding deaths) 95% 97% 96% 2 -Years 90% 85% 88% 2 -Years (excluding deaths) 91% 90% 91% Baseline 7

Visits and Treatment for Diabetic Macular Edema 8
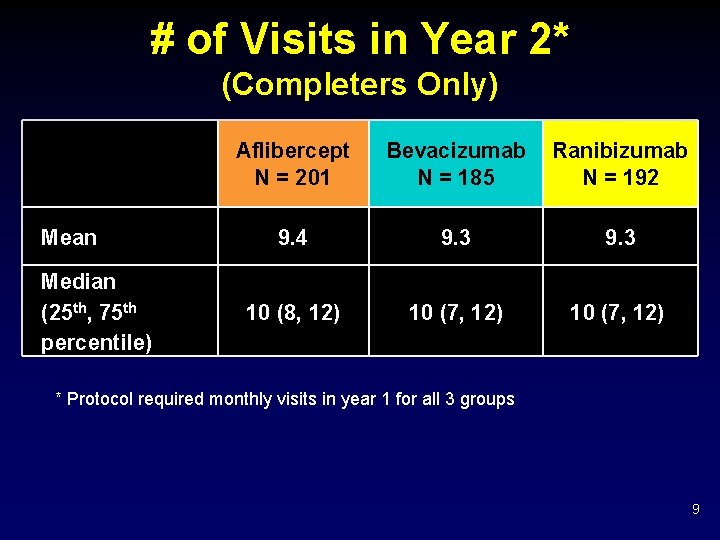
# of Visits in Year 2* (Completers Only) Mean Median (25 th, 75 th percentile) Aflibercept N = 201 Bevacizumab N = 185 Ranibizumab N = 192 9. 4 9. 3 10 (8, 12) 10 (7, 12) * Protocol required monthly visits in year 1 for all 3 groups 9
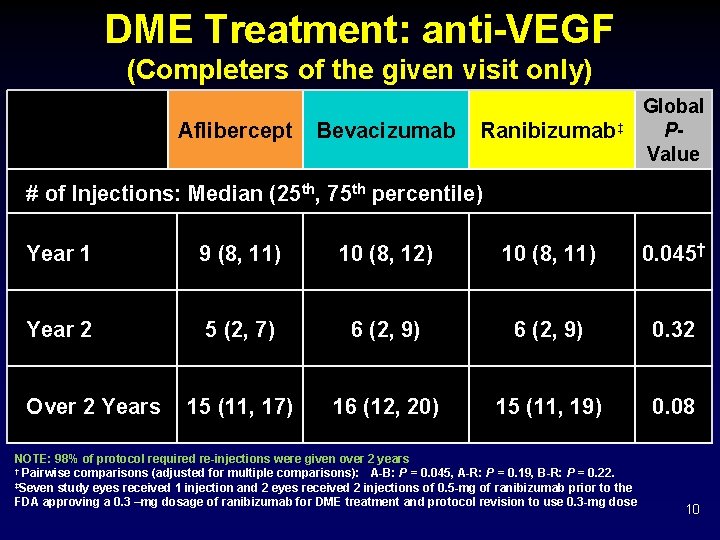
DME Treatment: anti-VEGF (Completers of the given visit only) Aflibercept Bevacizumab Ranibizumab‡ Global PValue # of Injections: Median (25 th, 75 th percentile) Year 1 9 (8, 11) 10 (8, 12) 10 (8, 11) 0. 045† Year 2 5 (2, 7) 6 (2, 9) 0. 32 15 (11, 17) 16 (12, 20) 15 (11, 19) 0. 08 Over 2 Years NOTE: 98% of protocol required re-injections were given over 2 years † Pairwise comparisons (adjusted for multiple comparisons): A-B: P = 0. 045, A-R: P = 0. 19, B-R: P = 0. 22. ‡Seven study eyes received 1 injection and 2 eyes received 2 injections of 0. 5 -mg of ranibizumab prior to the FDA approving a 0. 3 –mg dosage of ranibizumab for DME treatment and protocol revision to use 0. 3 -mg dose 10
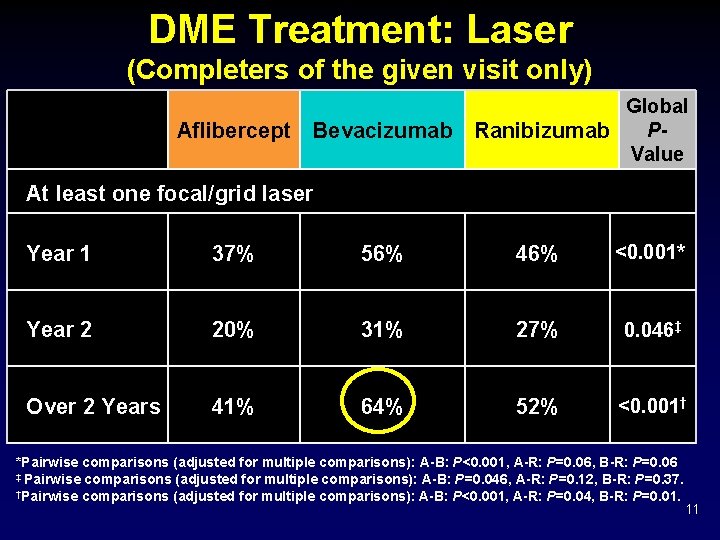
DME Treatment: Laser (Completers of the given visit only) Aflibercept Bevacizumab Global PRanibizumab Value At least one focal/grid laser Year 1 37% 56% 46% <0. 001* Year 2 20% 31% 27% 0. 046‡ Over 2 Years 41% 64% 52% <0. 001† *Pairwise comparisons (adjusted for multiple comparisons): A-B: P<0. 001, A-R: P=0. 06, B-R: P=0. 06 ‡ Pairwise comparisons (adjusted for multiple comparisons): A-B: P=0. 046, A-R: P=0. 12, B-R: P=0. 37. †Pairwise comparisons (adjusted for multiple comparisons): A-B: P<0. 001, A-R: P=0. 04, B-R: P=0. 01. 11

Efficacy 12
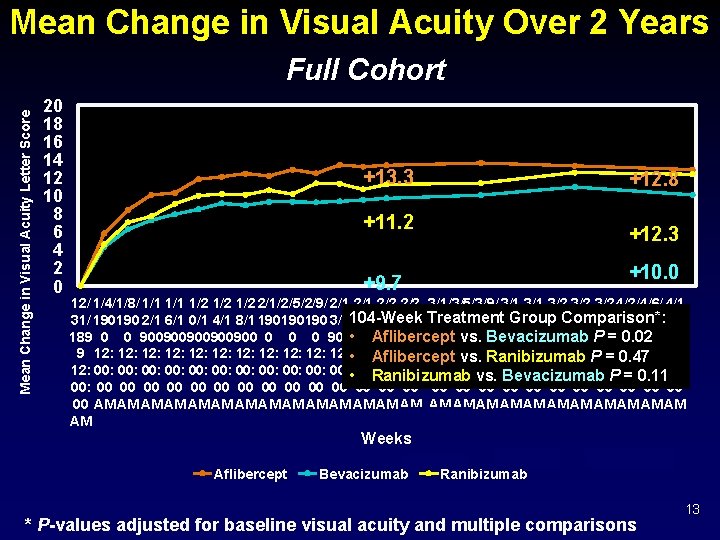
Mean Change in Visual Acuity Over 2 Years Mean Change in Visual Acuity Letter Score Full Cohort 20 18 16 14 12 10 8 6 4 2 0 +13. 3 +12. 8 +11. 2 +12. 3 +10. 0 +9. 7 12/ 1/4/1/8/ 1/1 1/2 1/2 2/1/2/5/2/9/ 2/1 2/2 3/1/3/5/3/9/ 3/1 3/2 3/2 4/2/4/6/ 4/1 Group 31/ 190190 2/1 6/1 0/1 4/1 8/1 190190190 3/1104 -Week 7/1 1/1 5/1 Treatment 190190190 3/1 7/1 1/1 Comparison*: 5/1 9/1 190190 0/1 189 0 0 900900900 0 900 • 900 Aflibercept 900900 0 0 900900900 0 0 900 vs. 0 Bevacizumab P = 0. 02 9 12: 12: 12: • 12: Aflibercept 12: 12: P 12: 12: vs. 12: Ranibizumab = 12: 0. 47 12: 00: 00: 00: • 00: Ranibizumab 00: 00: vs. 00: 00: 00: P 00: 00: Bevacizumab = 0. 11 00: 00 00 00 00 00 00 00 AM AM AM AM AM AM AM Weeks Aflibercept Bevacizumab Ranibizumab * P-values adjusted for baseline visual acuity and multiple comparisons 13
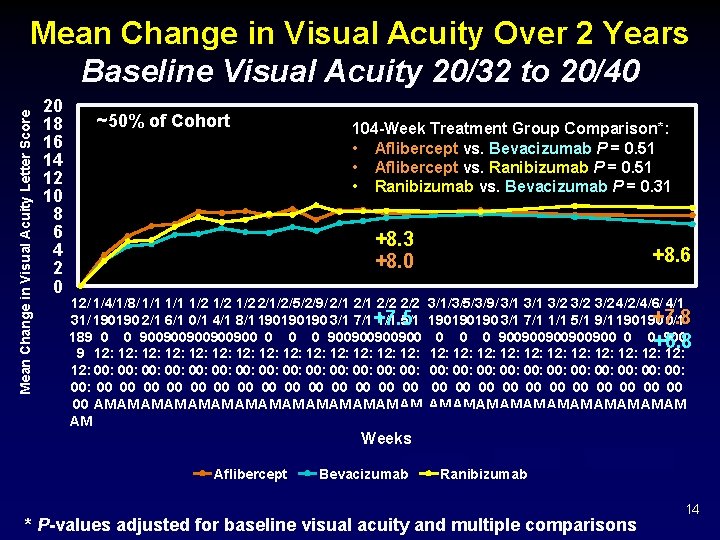
Mean Change in Visual Acuity Letter Score Mean Change in Visual Acuity Over 2 Years Baseline Visual Acuity 20/32 to 20/40 20 18 16 14 12 10 8 6 4 2 0 ~50% of Cohort 104 -Week Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P = 0. 51 • Aflibercept vs. Ranibizumab P = 0. 51 • Ranibizumab vs. Bevacizumab P = 0. 31 +8. 3 +8. 0 12/ 1/4/1/8/ 1/1 1/2 1/2 2/1/2/5/2/9/ 2/1 2/2 31/ 190190 2/1 6/1 0/1 4/1 8/1 190190190 3/1 7/1+7. 5 1/1 5/1 189 0 0 900900900 0 900900 9 12: 12: 12: 12: 00: 00: 00: 00: 00 00 00 00 AM AM AM AM +8. 6 3/1/3/5/3/9/ 3/1 3/2 3/2 4/2/4/6/ 4/1 +7. 8 190190190 3/1 7/1 1/1 5/1 9/1 190190 0/1 0 0 0 900900900 0 0+6. 8 900 12: 12: 12: 00: 00: 00: 00 00 00 AM AM AM Weeks Aflibercept Bevacizumab Ranibizumab * P-values adjusted for baseline visual acuity and multiple comparisons 14
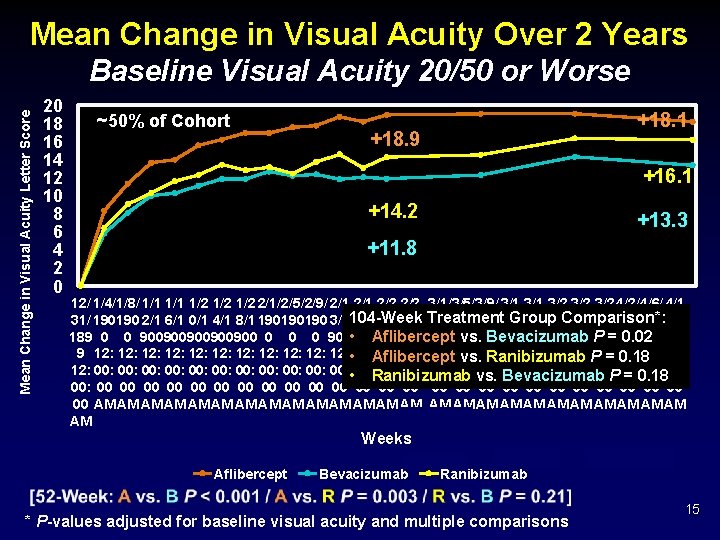
Mean Change in Visual Acuity Over 2 Years Mean Change in Visual Acuity Letter Score Baseline Visual Acuity 20/50 or Worse 20 18 16 14 12 10 8 6 4 2 0 ~50% of Cohort +18. 1 +18. 9 +16. 1 +14. 2 +13. 3 +11. 8 12/ 1/4/1/8/ 1/1 1/2 1/2 2/1/2/5/2/9/ 2/1 2/2 3/1/3/5/3/9/ 3/1 3/2 3/2 4/2/4/6/ 4/1 Group 31/ 190190 2/1 6/1 0/1 4/1 8/1 190190190 3/1104 -Week 7/1 1/1 5/1 Treatment 190190190 3/1 7/1 1/1 Comparison*: 5/1 9/1 190190 0/1 189 0 0 900900900 0 900 • 900 Aflibercept 900900 0 0 900900900 0 0 900 vs. 0 Bevacizumab P = 0. 02 9 12: 12: 12: • 12: Aflibercept 12: 12: P 12: 12: vs. 12: Ranibizumab = 12: 0. 18 12: 00: 00: 00: • 00: Ranibizumab 00: 00: vs. 00: 00: 00: P 00: 00: Bevacizumab = 0. 18 00: 00 00 00 00 00 00 00 AM AM AM AM AM AM AM Weeks Aflibercept Bevacizumab Ranibizumab * P-values adjusted for baseline visual acuity and multiple comparisons 15
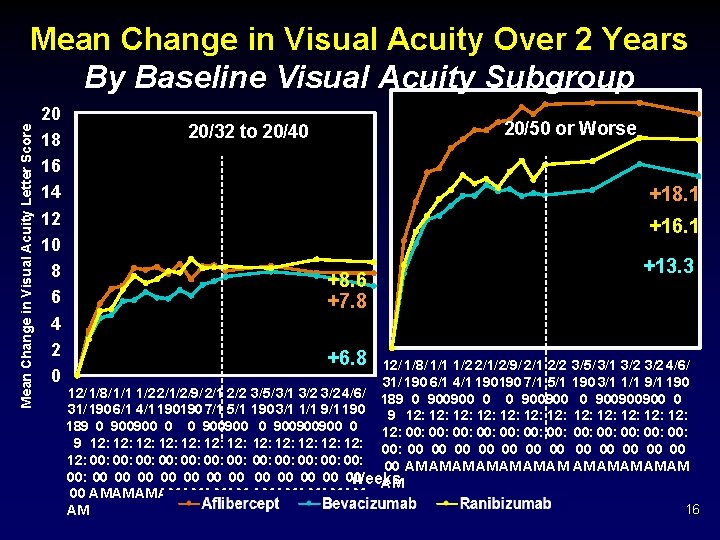
Mean Change in Visual Acuity Letter Score Mean Change in Visual Acuity Over 2 Years By Baseline Visual Acuity Subgroup 20 18 16 14 12 10 8 6 4 2 0 20/50 or Worse 20/32 to 20/40 +18. 1 +16. 1 +8. 6 +7. 8 +6. 8 +13. 3 12/ 1/8/ 1/1 1/2 2/1/2/9/ 2/1 2/2 3/5/ 3/1 3/2 4/6/ 31/ 190 6/1 4/1 190190 7/1 5/1 190 3/1 1/1 9/1 190 12/ 1/8/ 1/1 1/2 2/1/2/9/ 2/1 2/2 3/5/ 3/1 3/2 4/6/ 189 0 900900900 0 31/ 190 6/1 4/1 190190 7/1 5/1 190 3/1 1/1 9/1 190 9 12: 12: 12: 189 0 900900900 0 12: 00: 00: 00: 9 12: 12: 12: 00: 00 00 00 12: 00: 00: 00: 00 AM AM AM 00: 00 00 00 Weeks AM 00 AM AM AM 16 AM
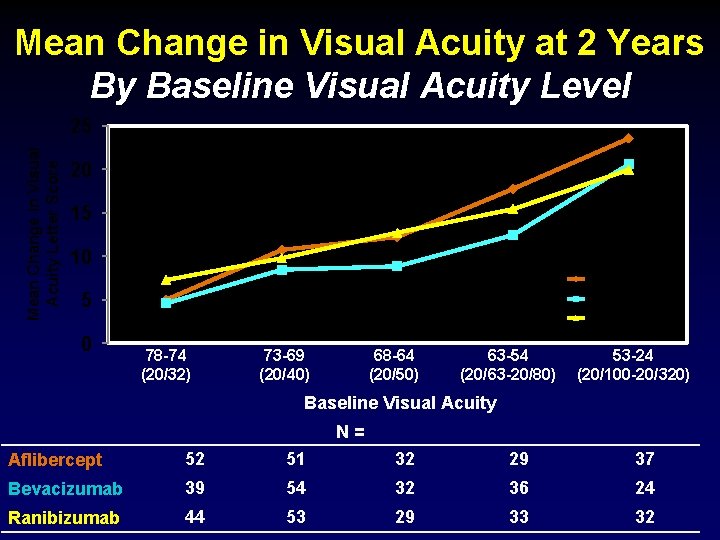
Mean Change in Visual Acuity at 2 Years By Baseline Visual Acuity Level Mean Change in Visual Acuity Letter Score 25 20 15 10 Aflibercept 5 Bevacizumab Ranibizumab 0 78 -74 (20/32) 73 -69 (20/40) 68 -64 (20/50) 63 -54 (20/63 -20/80) 53 -24 (20/100 -20/320) Baseline Visual Acuity N= Aflibercept 52 51 32 29 37 Bevacizumab 39 54 32 36 24 Ranibizumab 44 53 29 33 32
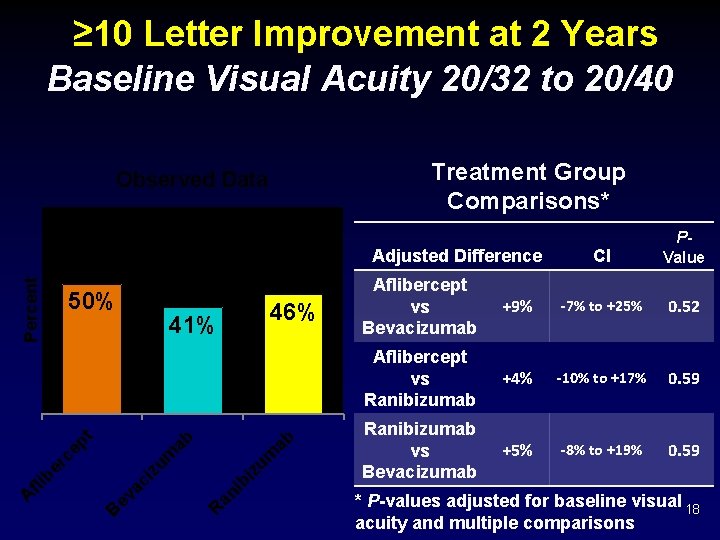
≥ 10 Letter Improvement at 2 Years Baseline Visual Acuity 20/32 to 20/40 Treatment Group Comparisons* Observed Data 50% 46% ab zu m bi R an i zu m ab 41% B ev ac i A fli b er ce pt Percent Adjusted Difference CI PValue Aflibercept vs Bevacizumab +9% -7% to +25% 0. 52 Aflibercept vs Ranibizumab +4% -10% to +17% 0. 59 Ranibizumab vs Bevacizumab +5% -8% to +19% 0. 59 * P-values adjusted for baseline visual 18 acuity and multiple comparisons
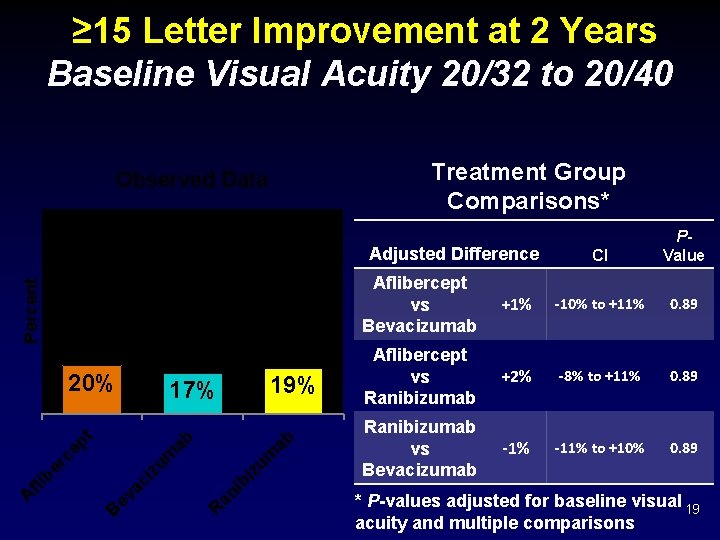
≥ 15 Letter Improvement at 2 Years Baseline Visual Acuity 20/32 to 20/40 Treatment Group Comparisons* Observed Data Percent Adjusted Difference 19% ab zu m bi R an i zu m ab 17% B ev ac i A fli b er c ep t 20% CI PValue Aflibercept vs Bevacizumab +1% -10% to +11% 0. 89 Aflibercept vs Ranibizumab +2% -8% to +11% 0. 89 Ranibizumab vs Bevacizumab -1% -11% to +10% 0. 89 * P-values adjusted for baseline visual 19 acuity and multiple comparisons
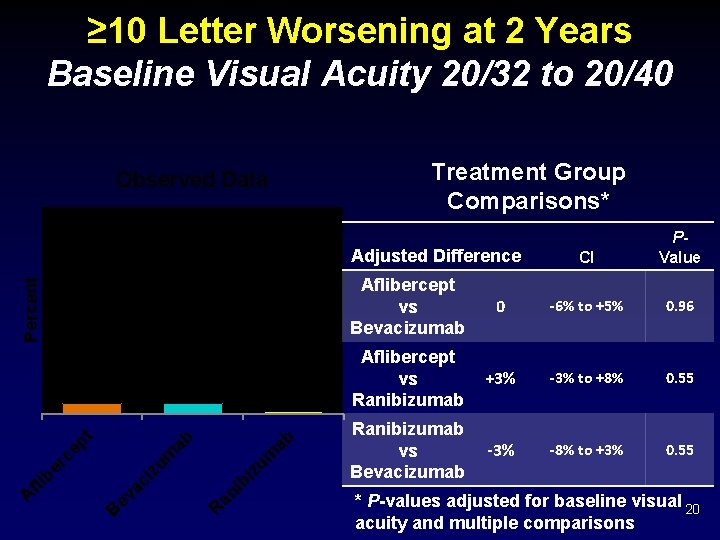
≥ 10 Letter Worsening at 2 Years Baseline Visual Acuity 20/32 to 20/40 Treatment Group Comparisons* Observed Data Percent Adjusted Difference 4% 1% R an i bi zu m ab ab zu m B ev ac i A fli b er ce pt 4% CI PValue Aflibercept vs Bevacizumab 0 -6% to +5% 0. 96 Aflibercept vs Ranibizumab +3% -3% to +8% 0. 55 Ranibizumab vs Bevacizumab -3% -8% to +3% 0. 55 * P-values adjusted for baseline visual 20 acuity and multiple comparisons
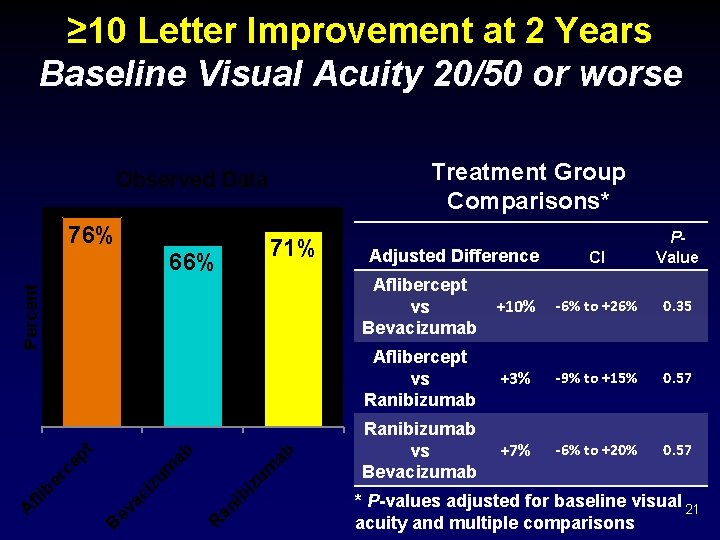
≥ 10 Letter Improvement at 2 Years Baseline Visual Acuity 20/50 or worse Treatment Group Comparisons* Observed Data 76% 71% ab iz um ib an R ev ac i B A fli b er c zu m ep t ab Percent 66% Adjusted Difference CI PValue Aflibercept vs Bevacizumab +10% -6% to +26% 0. 35 Aflibercept vs Ranibizumab +3% -9% to +15% 0. 57 Ranibizumab vs Bevacizumab +7% -6% to +20% 0. 57 * P-values adjusted for baseline visual 21 acuity and multiple comparisons
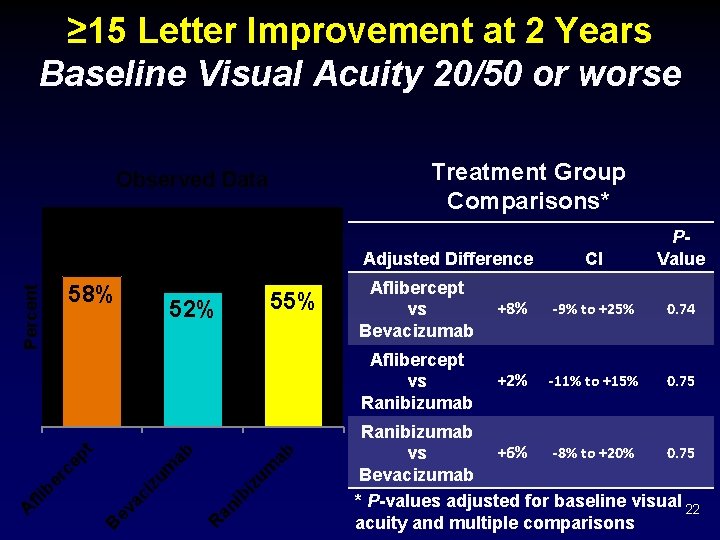
≥ 15 Letter Improvement at 2 Years Baseline Visual Acuity 20/50 or worse Treatment Group Comparisons* Observed Data 58% ab ab iz um ib an R B ev ac i er c fli b A 55% 52% zu m ep t Percent Adjusted Difference CI PValue Aflibercept vs Bevacizumab +8% -9% to +25% 0. 74 Aflibercept vs Ranibizumab +2% -11% to +15% 0. 75 Ranibizumab 0. 75 +6% -8% to +20% vs Bevacizumab * P-values adjusted for baseline visual 22 acuity and multiple comparisons
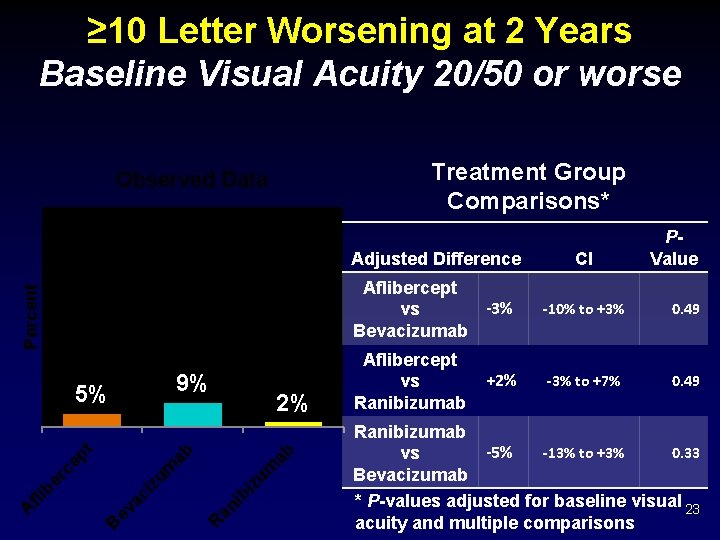
≥ 10 Letter Worsening at 2 Years Baseline Visual Acuity 20/50 or worse Treatment Group Comparisons* Percent Observed Data 9% ab ib an R ev ac i B iz um zu m ep t er c fli b A 2% ab 5% PValue Adjusted Difference CI Aflibercept -3% vs Bevacizumab -10% to +3% 0. 49 Aflibercept vs Ranibizumab -3% to +7% 0. 49 +2% Ranibizumab -13% to +3% 0. 33 -5% vs Bevacizumab * P-values adjusted for baseline visual 23 acuity and multiple comparisons
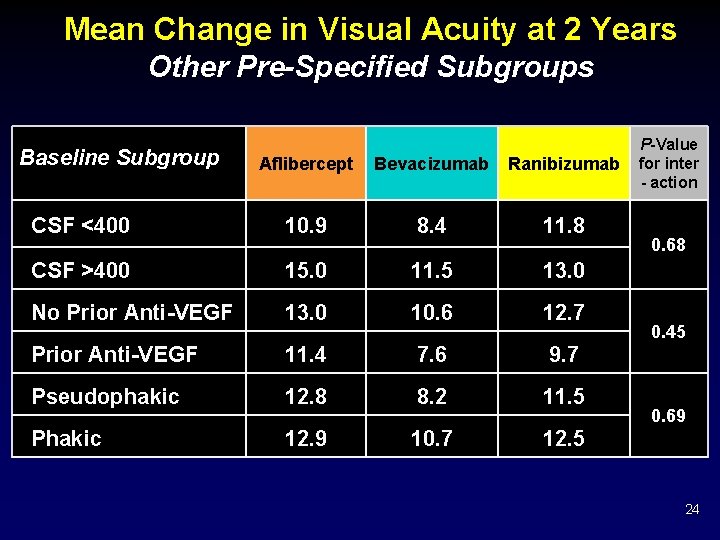
Mean Change in Visual Acuity at 2 Years Other Pre-Specified Subgroups Baseline Subgroup Aflibercept Bevacizumab Ranibizumab CSF <400 10. 9 8. 4 11. 8 CSF >400 15. 0 11. 5 13. 0 No Prior Anti-VEGF 13. 0 10. 6 12. 7 Prior Anti-VEGF 11. 4 7. 6 9. 7 Pseudophakic 12. 8 8. 2 11. 5 Phakic 12. 9 10. 7 12. 5 P-Value for inter - action 0. 68 0. 45 0. 69 24
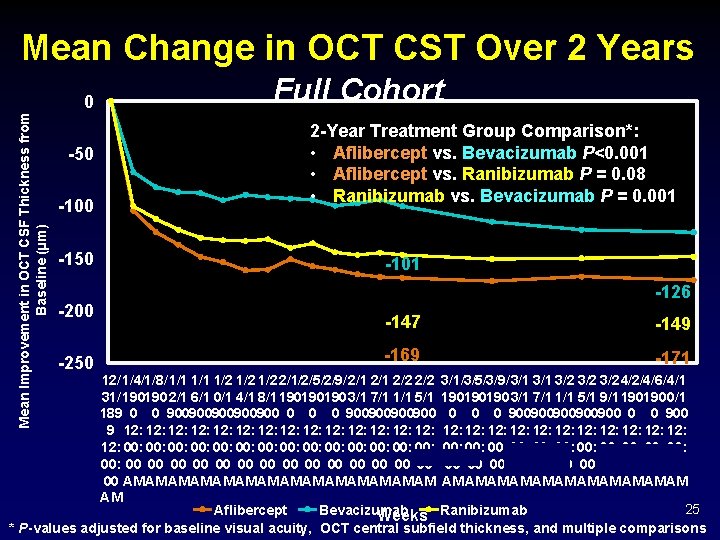
Mean Change in OCT CST Over 2 Years Mean Improvement in OCT CSF Thickness from Baseline (µm) 0 -50 -100 -150 -200 -250 Full Cohort 2 -Year Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P<0. 001 • Aflibercept vs. Ranibizumab P = 0. 08 • Ranibizumab vs. Bevacizumab P = 0. 001 -126 -147 -149 -169 -171 12/ 1/4/1/8/ 1/1 1/2 1/2 2/1/2/5/2/9/ 2/1 2/2 3/1/3/5/3/9/ 3/1 3/2 3/2 4/2/4/6/ 4/1 31/ 190190 2/1 6/1 0/1 4/1 8/1 190190190 3/1 7/1 1/1 5/1 9/1 190190 0/1 189 0 0 900900900 0 0 0 900900900900900 0 0 900 9 12: 12: 12: 12: 12: 12: 12: 00: 00: 00: 00: 00: 00: 00: 00 00 00 00 00 00 00 AM AM AM AM AM AM AM 25 Aflibercept Bevacizumab Weeks Ranibizumab * P-values adjusted for baseline visual acuity, OCT central subfield thickness, and multiple comparisons
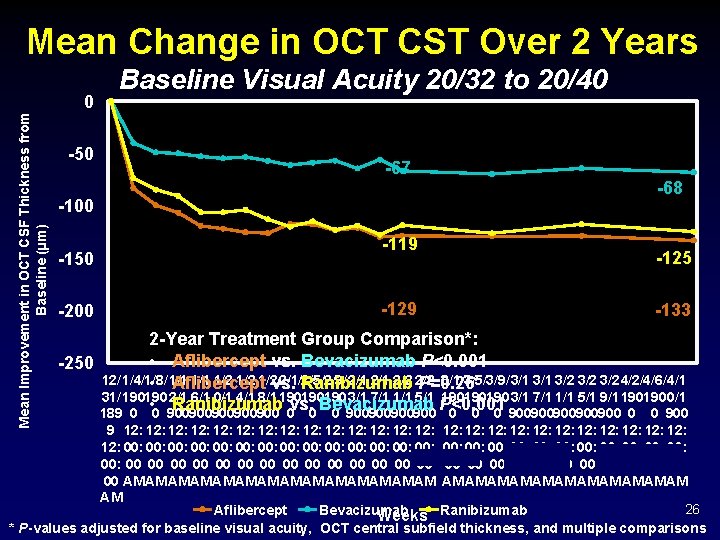
Mean Change in OCT CST Over 2 Years Mean Improvement in OCT CSF Thickness from Baseline (µm) 0 -50 Baseline Visual Acuity 20/32 to 20/40 -67 -100 -150 -200 -119 -129 -68 -125 -133 2 -Year Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P<0. 001 -250 12/ 1/4/1/8/ 1/1 1/2 1/2 2/1/Ranibizumab 2/5/2/9/ 2/1 2/2 3/1/3/5/3/9/ 3/1 3/2 3/2 4/2/4/6/ 4/1 • 1/1 Aflibercept vs. P=0. 26 31/ 190190 2/1 6/1 0/1 4/1 8/1 190190190 3/1 7/1 1/1 5/1 9/1 190190 0/1 • Ranibizumab vs. Bevacizumab P<0. 001 189 0 0 900900900 0 0 0 900900900900900 0 0 900 9 12: 12: 12: 12: 12: 12: 12: 00: 00: 00: 00: 00: 00: 00: 00 00 00 00 00 00 00 AM AM AM AM AM AM AM 26 Aflibercept Bevacizumab Weeks Ranibizumab * P-values adjusted for baseline visual acuity, OCT central subfield thickness, and multiple comparisons
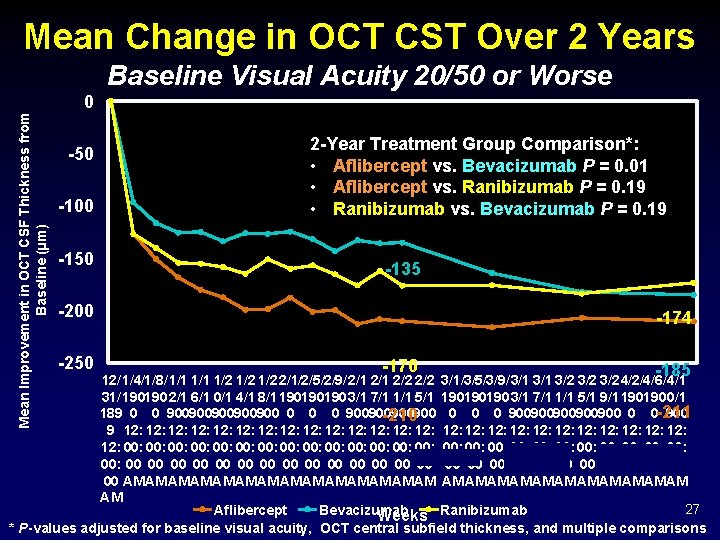
Mean Change in OCT CST Over 2 Years Baseline Visual Acuity 20/50 or Worse Mean Improvement in OCT CSF Thickness from Baseline (µm) 0 -50 -100 -150 2 -Year Treatment Group Comparison*: • Aflibercept vs. Bevacizumab P = 0. 01 • Aflibercept vs. Ranibizumab P = 0. 19 • Ranibizumab vs. Bevacizumab P = 0. 19 -135 -200 -250 -174 -176 -185 12/ 1/4/1/8/ 1/1 1/2 1/2 2/1/2/5/2/9/ 2/1 2/2 3/1/3/5/3/9/ 3/1 3/2 3/2 4/2/4/6/ 4/1 31/ 190190 2/1 6/1 0/1 4/1 8/1 190190190 3/1 7/1 1/1 5/1 9/1 190190 0/1 189 0 0 900900900 0 0 0 900900900 0 0 -211 900 -210 9 12: 12: 12: 12: 12: 12: 12: 00: 00: 00: 00: 00: 00: 00: 00 00 00 00 00 00 00 AM AM AM AM AM AM AM 27 Aflibercept Bevacizumab Weeks Ranibizumab * P-values adjusted for baseline visual acuity, OCT central subfield thickness, and multiple comparisons
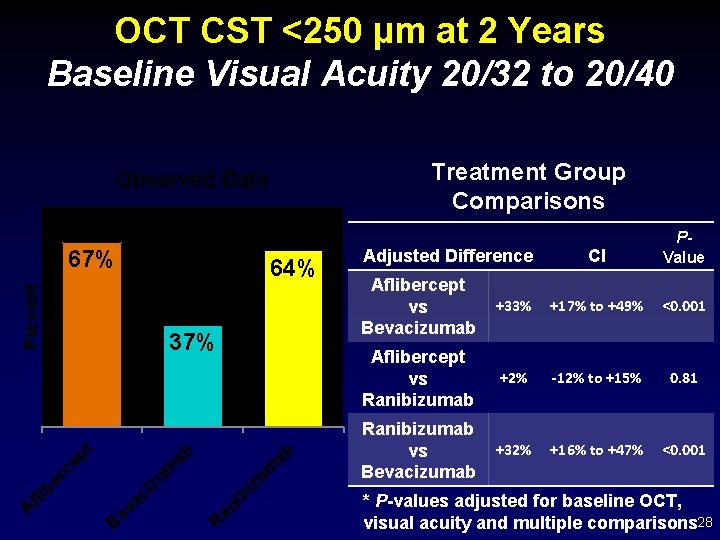
OCT CST <250 µm at 2 Years Baseline Visual Acuity 20/32 to 20/40 Treatment Group Comparisons Observed Data 67% Percent 64% ab iz um ib an R ev ac i B A fli b er c zu m ep t ab 37% Adjusted Difference CI PValue Aflibercept vs Bevacizumab +33% +17% to +49% <0. 001 Aflibercept vs Ranibizumab +2% -12% to +15% 0. 81 Ranibizumab vs Bevacizumab +32% +16% to +47% <0. 001 * P-values adjusted for baseline OCT, visual acuity and multiple comparisons 28
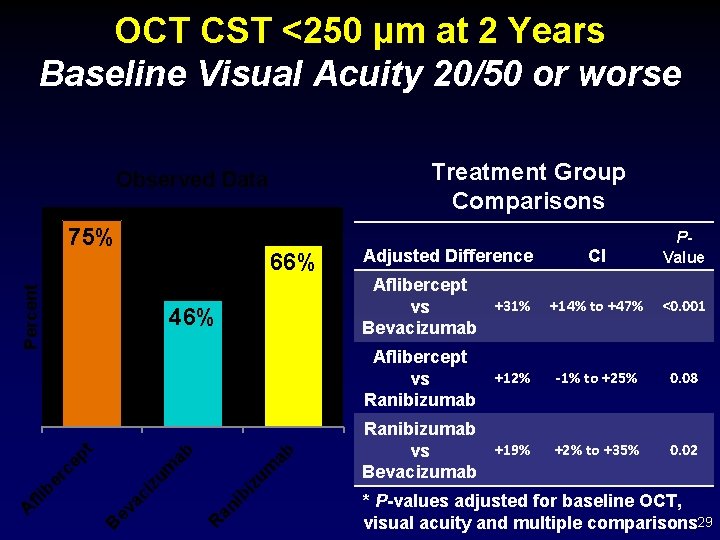
OCT CST <250 µm at 2 Years Baseline Visual Acuity 20/50 or worse Treatment Group Comparisons Observed Data 75% Percent 66% 46% CI Aflibercept +31% vs Bevacizumab +14% to +47% <0. 001 +12% -1% to +25% 0. 08 Ranibizumab +19% vs Bevacizumab +2% to +35% 0. 02 ab ib an R ev ac i B iz um zu m ep t ab Aflibercept vs Ranibizumab er c fli b A Adjusted Difference PValue * P-values adjusted for baseline OCT, visual acuity and multiple comparisons 29

Safety 30
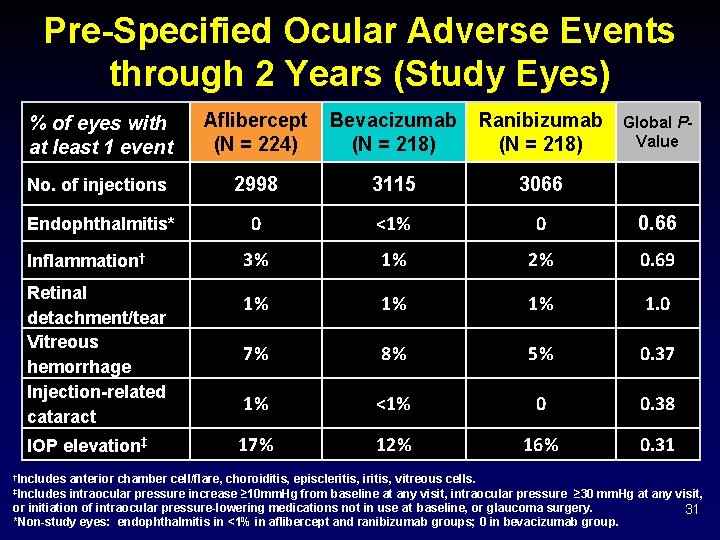
Pre-Specified Ocular Adverse Events through 2 Years (Study Eyes) % of eyes with at least 1 event Aflibercept (N = 224) Bevacizumab (N = 218) Ranibizumab (N = 218) No. of injections 2998 3115 3066 Endophthalmitis* 0 <1% 0 0. 66 Inflammation† 3% 1% 2% 0. 69 Retinal detachment/tear Vitreous hemorrhage Injection-related cataract 1% 1% 1% 1. 0 7% 8% 5% 0. 37 1% <1% 0 0. 38 IOP elevation‡ 17% 12% 16% 0. 31 †Includes Global PValue anterior chamber cell/flare, choroiditis, episcleritis, iritis, vitreous cells. intraocular pressure increase ≥ 10 mm. Hg from baseline at any visit, intraocular pressure ≥ 30 mm. Hg at any visit, or initiation of intraocular pressure-lowering medications not in use at baseline, or glaucoma surgery. 31 *Non-study eyes: endophthalmitis in <1% in aflibercept and ranibizumab groups; 0 in bevacizumab group. ‡Includes
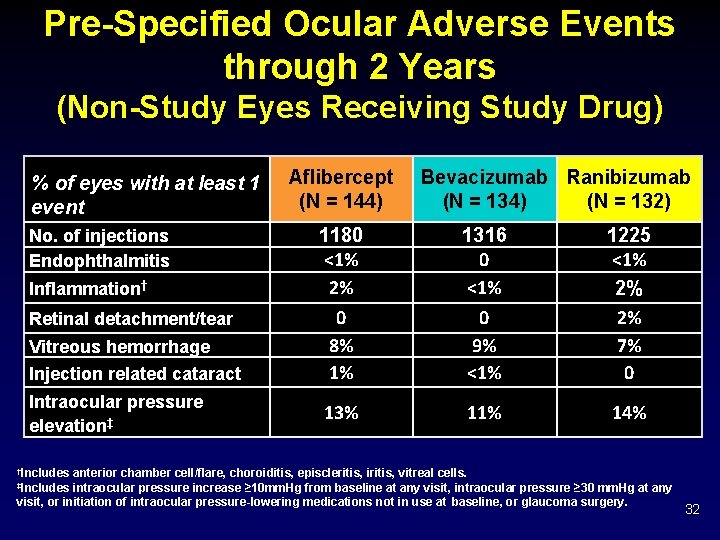
Pre-Specified Ocular Adverse Events through 2 Years (Non-Study Eyes Receiving Study Drug) % of eyes with at least 1 event No. of injections Endophthalmitis Inflammation† Retinal detachment/tear Vitreous hemorrhage Injection related cataract Intraocular pressure elevation‡ Aflibercept (N = 144) Bevacizumab Ranibizumab (N = 134) (N = 132) 1180 <1% 2% 1316 0 <1% 0 8% 1% 0 9% <1% 1225 <1% 2% 2% 7% 0 13% 11% 14% †Includes anterior chamber cell/flare, choroiditis, episcleritis, iritis, vitreal cells. intraocular pressure increase ≥ 10 mm. Hg from baseline at any visit, intraocular pressure ≥ 30 mm. Hg at any visit, or initiation of intraocular pressure-lowering medications not in use at baseline, or glaucoma surgery. ‡Includes 32
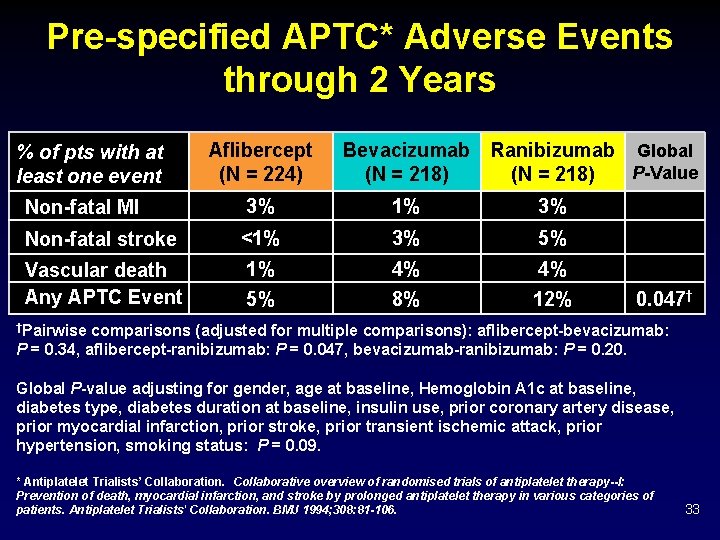
Pre-specified APTC* Adverse Events through 2 Years Aflibercept (N = 224) Bevacizumab (N = 218) Non-fatal MI 3% 1% 3% Non-fatal stroke <1% 3% 5% Vascular death Any APTC Event 1% 5% 4% 8% 4% 12% % of pts with at least one event Ranibizumab Global P-Value (N = 218) 0. 047† †Pairwise comparisons (adjusted for multiple comparisons): aflibercept-bevacizumab: P = 0. 34, aflibercept-ranibizumab: P = 0. 047, bevacizumab-ranibizumab: P = 0. 20. Global P-value adjusting for gender, age at baseline, Hemoglobin A 1 c at baseline, diabetes type, diabetes duration at baseline, insulin use, prior coronary artery disease, prior myocardial infarction, prior stroke, prior transient ischemic attack, prior hypertension, smoking status: P = 0. 09. * Antiplatelet Trialists’ Collaboration. Collaborative overview of randomised trials of antiplatelet therapy--I: Prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients. Antiplatelet Trialists' Collaboration. BMJ 1994; 308: 81 -106. 33
Post Hoc Analysis of APTC Adverse Events Stratified by Prior MI/Stroke % of pts with at least one event No Prior MI/Stroke Non-fatal MI Aflibercept Bevacizumab Ranibizumab N = 203 N = 193 N = 21 N = 25 Non-fatal stroke Vascular death Any APTC Event Prior MI/Stroke Non-fatal MI Non-fatal stroke Vascular death Any APTC Event 34
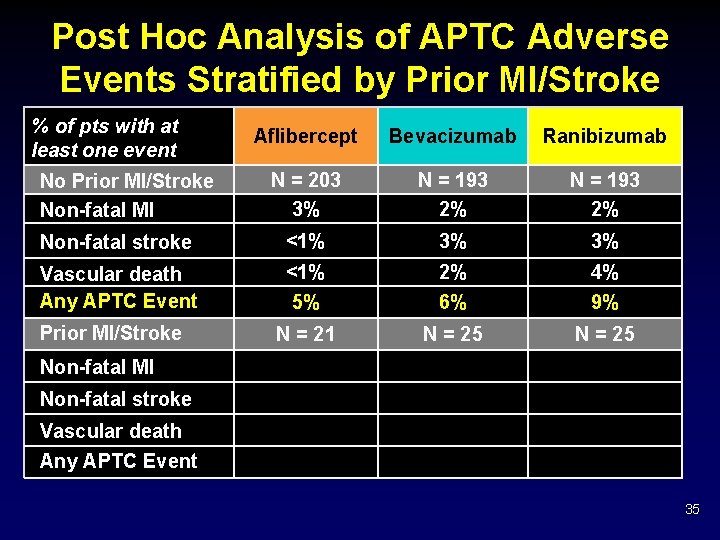
Post Hoc Analysis of APTC Adverse Events Stratified by Prior MI/Stroke % of pts with at least one event Aflibercept Bevacizumab Ranibizumab N = 203 3% N = 193 2% Non-fatal stroke <1% 3% 3% Vascular death Any APTC Event <1% 5% 2% 6% 4% 9% N = 21 N = 25 No Prior MI/Stroke Non-fatal MI Non-fatal stroke Vascular death Any APTC Event 35
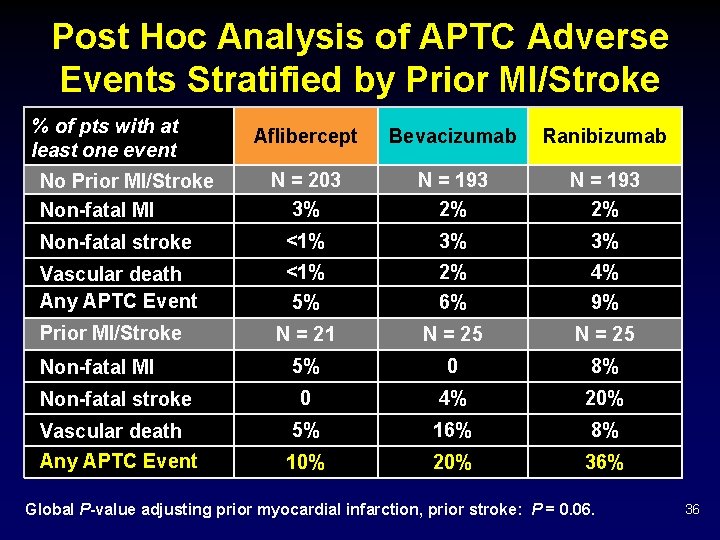
Post Hoc Analysis of APTC Adverse Events Stratified by Prior MI/Stroke % of pts with at least one event Aflibercept Bevacizumab Ranibizumab N = 203 3% N = 193 2% Non-fatal stroke <1% 3% 3% Vascular death Any APTC Event <1% 5% 2% 6% 4% 9% N = 21 N = 25 5% 0 8% Non-fatal stroke 0 4% 20% Vascular death Any APTC Event 5% 16% 8% 10% 20% 36% No Prior MI/Stroke Non-fatal MI Global P-value adjusting prior myocardial infarction, prior stroke: P = 0. 06. 36
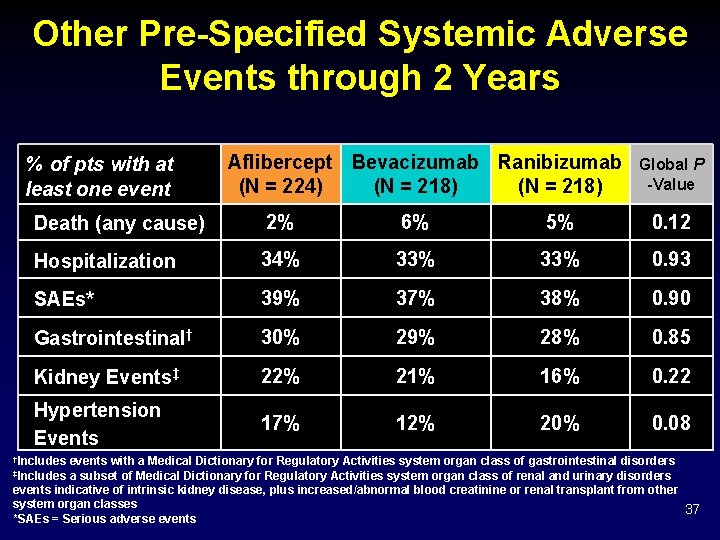
Other Pre-Specified Systemic Adverse Events through 2 Years % of pts with at least one event Aflibercept Bevacizumab Ranibizumab (N = 224) (N = 218) Global P -Value Death (any cause) 2% 6% 5% 0. 12 Hospitalization 34% 33% 0. 93 SAEs* 39% 37% 38% 0. 90 Gastrointestinal† 30% 29% 28% 0. 85 Kidney Events‡ 22% 21% 16% 0. 22 Hypertension Events 17% 12% 20% 0. 08 †Includes events with a Medical Dictionary for Regulatory Activities system organ class of gastrointestinal disorders a subset of Medical Dictionary for Regulatory Activities system organ class of renal and urinary disorders events indicative of intrinsic kidney disease, plus increased/abnormal blood creatinine or renal transplant from other system organ classes 37 *SAEs = Serious adverse events ‡Includes
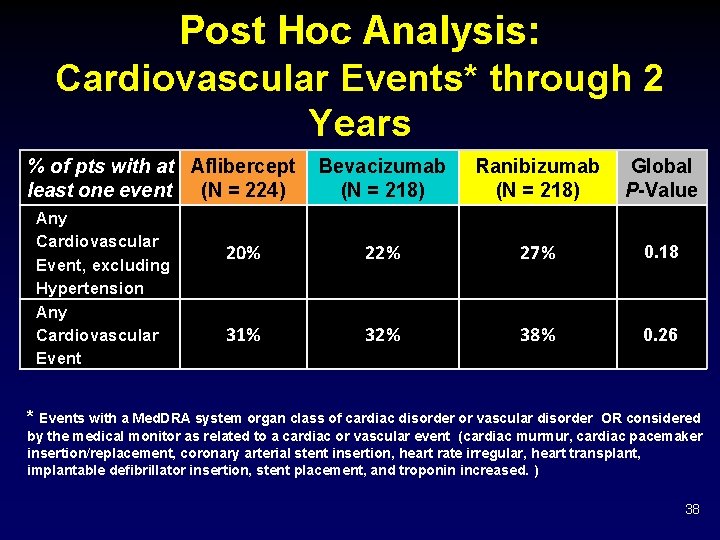
Post Hoc Analysis: Cardiovascular Events* through 2 Years % of pts with at Aflibercept least one event (N = 224) Any Cardiovascular Event, excluding Hypertension Any Cardiovascular Event Bevacizumab (N = 218) Ranibizumab (N = 218) Global P-Value 20% 22% 27% 0. 18 31% 32% 38% 0. 26 * Events with a Med. DRA system organ class of cardiac disorder or vascular disorder OR considered by the medical monitor as related to a cardiac or vascular event (cardiac murmur, cardiac pacemaker insertion/replacement, coronary arterial stent insertion, heart rate irregular, heart transplant, implantable defibrillator insertion, stent placement, and troponin increased. ) 38

Discussion At one year: Ø All three anti-VEGF agents are effective treatments for DME causing vision impairment. Ø When initial visual acuity loss is mild, on average, there is little difference in visual acuity. Ø At worse levels of initial visual acuity, aflibercept is more effective at improving vision. 39

Discussion At 2 years: Ø Vision gains (from baseline) were seen with all three drugs at 2 years, with reduced number of injections and lasers in year 2 Ø When initial visual acuity loss is mild, on average there is still little difference in visual acuity. Ø At worse levels of initial visual acuity aflibercept was more effective at improving visual acuity versus bevacizumab, but not ranibizumab. 40

Discussion Ø Few eyes in any group lost substantial amounts of visual acuity Ø In worse VA subgroup: • At one year, percentage in the aflibercept group gaining 15 or more letters was 63% and 34% greater (relative difference) than that of the bevacizumab and ranibizumab groups, respectively • At 2 years o The percentage in aflibercept group gaining >15 letters was 12% and 5% greater (relative difference), respectively o The percentage in aflibercept group gaining >10 letters was 15% and 7% greater (relative difference), respectively 41

Discussion Ø At year one, bevacizumab was found to reduce retinal thickness less than the other 2 agents. This difference persisted in year 2 in the eyes with better initial visual acuity. Ø In eyes with worse initial visual acuity, the mean change in CST was significantly worse only in the bevacizumab group compared with aflibercept 42

Discussion Ø In year 2, ~half the number of injections were indicated per protocol as in year 1 in each group. Ø Cumulative number of injections was similar across all groups Ø Similar to year 1, laser treatment was indicated per protocol less frequently in the aflibercepttreated eyes in year two. • Since focal/grid laser to persistent DME after the 24 week visit was a protocol-defined part of the treatment regimen, it is not possible to separate the effect of macular laser from the anti-VEGF treatment on the VA and thickness outcomes after the 6 -month visit. 43

Discussion Ø Pre-defined systemic APTC rates were higher in the ranibizumab group • Consisting of more non-fatal strokes and vascular deaths in the ranibizumab group. • Although P-values increased slightly after adjusting for a history of prior stroke or MI and other potential confounders, this did not alter the results substantially. Ø These findings have not been demonstrated consistently in previously reported clinical trials. 44
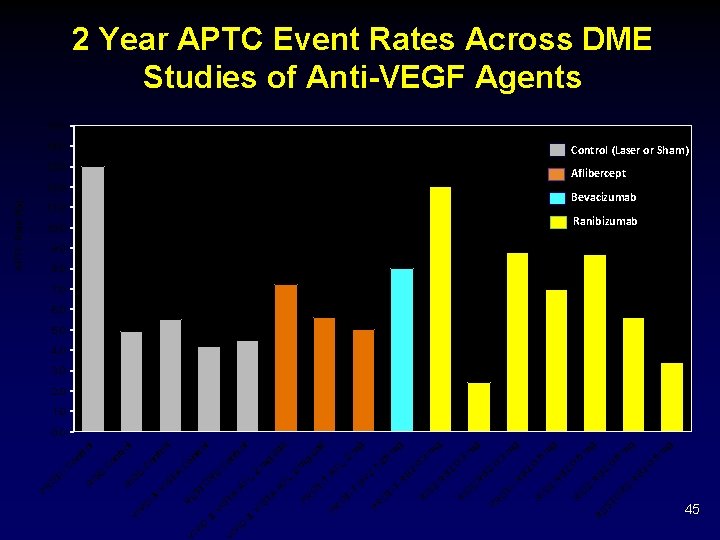
ID IV Z B R 0. 5 g 5. 6 m g m 8. 8 E 0. 5 10. 0 R 11. 0 TO g m g 12. 0 Z B R 0. 5 m g m 12. 0 ES E ID Z B R 5 0. 3 g m 7. 2 R E IS R B Z IR T- O Z B R 0. 3 3 0. g m 3. 0 PR E ID Z B R Z R B 25 1. 2 13. 0 R E IS T B VZ FL 9. 0 R T- O T T- 8 2 q 4. 5 PR O A g m 2 q 4 5. 5 PR T T- O 2 g m 8. 0 PR FL A 2 on tr ol C 4. 2 A FL A E R 4. 9 ST VI A ST VI on tr ol C ol tr on C 13 R & & TO A ST VI ES R & E 5. 0 D tr ol 6. 0 VI R ID C on ol tr 14. 0 VI E IS R on IC T- O PR APTC Rate (%) 2 Year APTC Event Rates Across DME Studies of Anti-VEGF Agents 15. 0 Control (Laser or Sham) Aflibercept Bevacizumab Ranibizumab 8. 7 8. 0 7. 0 5. 6 4. 0 3. 4 2. 0 1. 0 0. 0 45
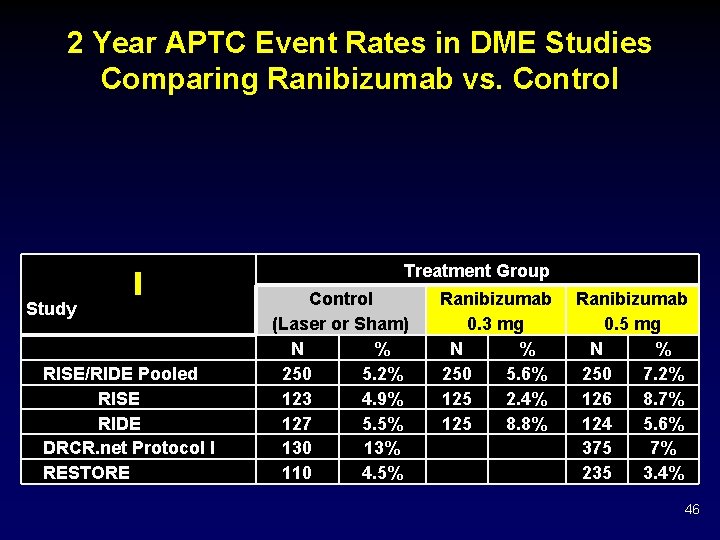
2 Year APTC Event Rates in DME Studies Comparing Ranibizumab vs. Control Treatment Group Study RISE/RIDE Pooled RISE RIDE DRCR. net Protocol I RESTORE Control (Laser or Sham) N % 250 5. 2% 123 4. 9% 127 5. 5% 130 13% 110 4. 5% Ranibizumab 0. 3 mg N % 250 5. 6% 125 2. 4% 125 8. 8% Ranibizumab 0. 5 mg N % 250 7. 2% 126 8. 7% 124 5. 6% 375 7% 235 3. 4% 46
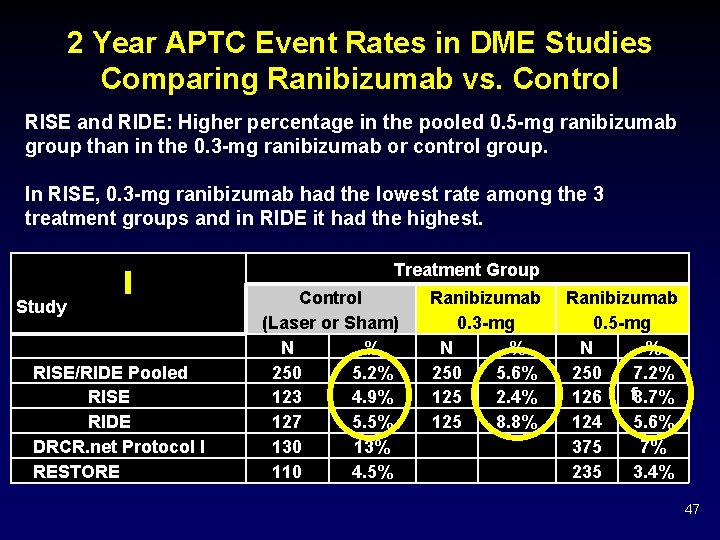
2 Year APTC Event Rates in DME Studies Comparing Ranibizumab vs. Control RISE and RIDE: Higher percentage in the pooled 0. 5 -mg ranibizumab group than in the 0. 3 -mg ranibizumab or control group. In RISE, 0. 3 -mg ranibizumab had the lowest rate among the 3 treatment groups and in RIDE it had the highest. Treatment Group Study RISE/RIDE Pooled RISE RIDE DRCR. net Protocol I RESTORE Control (Laser or Sham) N % 250 5. 2% 123 4. 9% 127 5. 5% 130 13% 110 4. 5% Ranibizumab 0. 3 -mg N % 250 5. 6% 125 2. 4% 125 8. 8% Ranibizumab 0. 5 -mg N % 250 7. 2% f 8. 7% 126 124 5. 6% 375 7% 235 3. 4% 47
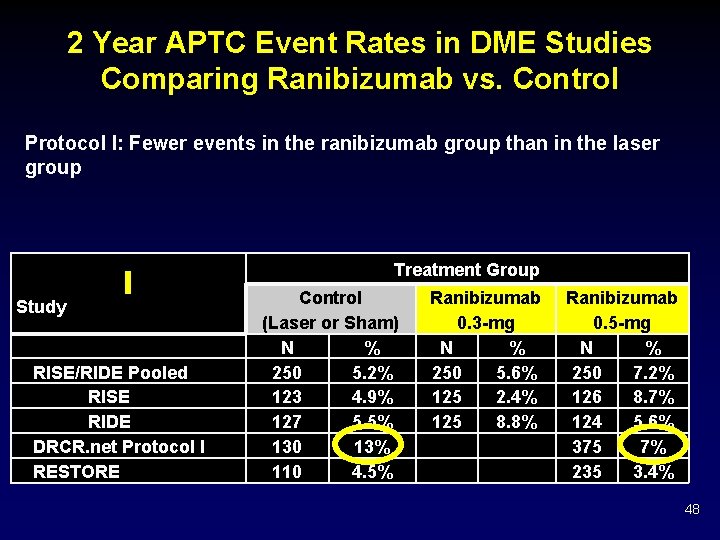
2 Year APTC Event Rates in DME Studies Comparing Ranibizumab vs. Control Protocol I: Fewer events in the ranibizumab group than in the laser group Treatment Group Study RISE/RIDE Pooled RISE RIDE DRCR. net Protocol I RESTORE Control (Laser or Sham) N % 250 5. 2% 123 4. 9% 127 5. 5% 130 13% 110 4. 5% Ranibizumab 0. 3 -mg N % 250 5. 6% 125 2. 4% 125 8. 8% Ranibizumab 0. 5 -mg N % 250 7. 2% 126 8. 7% 124 5. 6% 375 7% 235 3. 4% 48
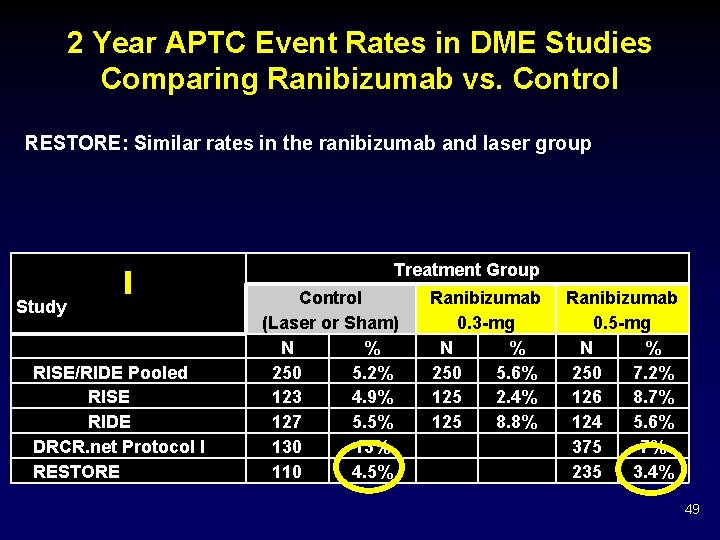
2 Year APTC Event Rates in DME Studies Comparing Ranibizumab vs. Control RESTORE: Similar rates in the ranibizumab and laser group Treatment Group Study RISE/RIDE Pooled RISE RIDE DRCR. net Protocol I RESTORE Control (Laser or Sham) N % 250 5. 2% 123 4. 9% 127 5. 5% 130 13% 110 4. 5% Ranibizumab 0. 3 -mg N % 250 5. 6% 125 2. 4% 125 8. 8% Ranibizumab 0. 5 -mg N % 250 7. 2% 126 8. 7% 124 5. 6% 375 7% 235 3. 4% 49

2 Year APTC Event Rates in AMD Studies Comparing Ranibizumab vs. Other Anti-VEGF Agents CATT and IVAN: Rates were similar between the ranibizumab and bevacizumab groups Treatment Group Study IVAN CATT VIEW Aflibercept 0. 5 -mg N % 601 3. 8% Bevacizumab Ranibizumab 1. 25 -mg 0. 5 -mg N % 296 3% 314 4% 586 5. 0% 599 4. 7% 595 3. 2% 51
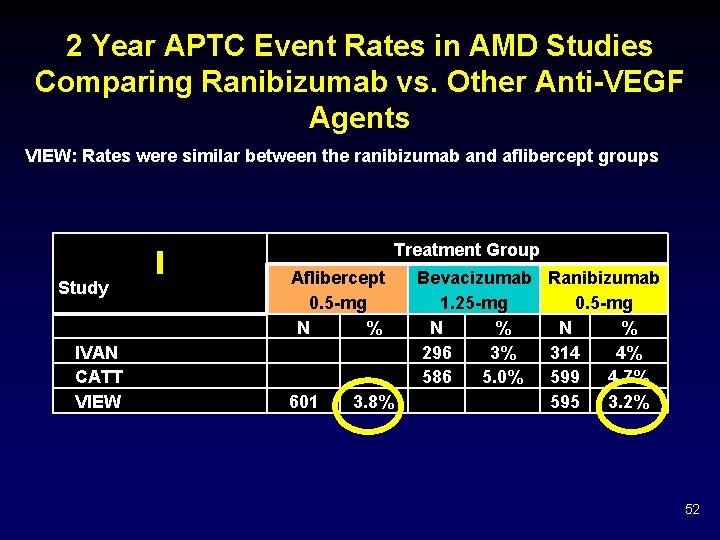
2 Year APTC Event Rates in AMD Studies Comparing Ranibizumab vs. Other Anti-VEGF Agents VIEW: Rates were similar between the ranibizumab and aflibercept groups Treatment Group Study IVAN CATT VIEW Aflibercept 0. 5 -mg N % 601 3. 8% Bevacizumab Ranibizumab 1. 25 -mg 0. 5 -mg N % 296 3% 314 4% 586 5. 0% 599 4. 7% 595 3. 2% 52

Discussion Ø The 12% rate of APTC events in ranibizumab participants in the current study appears to be an outlier relative to other trials, including DRCR. net Protocol I, where the percentage was 7% with high overlap in DRCR. net clinical centers. Ø Additionally, across multiple retinal diseases, a metaanalysis from Thulliez et al did not identify an increased risk of major cardiovascular or hemorrhagic events with ranibizumab compared with control. Ø The inconsistencies in the totality of evidence creates uncertainty as to whethere is a true increased risk of APTC events with ranibizumab at this time. Thulliez M, Angoulvant D, Le Lez ML, et al. Cardiovascular events and bleeding risk associated with intravitreal antivascular endothelial growth factor monoclonal antibodies: systematic review and meta-analysis. JAMA Ophthalmol. 2014; 132(11): 1317 -26. 53

Conclusions Ø Vision gains (from baseline) at 2 years were seen in all 3 groups with ~half the number of injections, slightly decreased frequency of visits, and decreased amounts of laser in the 2 nd year Ø Among eyes with better VA no differences in 2 -year vision outcomes identified Ø Among eyes with worse baseline VA: Ø Aflibercept, on average, had superior 2 -year VA outcomes compared with bevacizumab, although the difference was diminished Ø The VA difference between aflibercept and ranibizumab that was noted at 1 year had decreased at 2 years and was no longer statistically significant. Ø The implication of the increased rate of APTC events with ranibizumab found in the current study is uncertain due to inconsistency with prior trials, warranting 54 continued evaluation

Thank You on Behalf of Diabetic Retinopathy Clinical Research Network (DRCR. net) A complete list of all DRCR. net investigator financial disclosures and these slides can be found at www. drcr. net. Full protocol available on clinical. Trials. gov (NCT 01627249) 55
- Slides: 54