Diabetic kidney disease EDUCATIONAL SLIDE MODULE Date of

Diabetic kidney disease EDUCATIONAL SLIDE MODULE Date of preparation: July 2020 Version 3. 0 SC-CRP-05133

Contents Burden of kidney disease in type 2 diabetes Pathophysiology of diabetic kidney disease Prevention and management of diabetic kidney disease New treatments available and under evaluation

Definition of kidney disease Chronic kidney disease (CKD): abnormalities of kidney structure or function, present for >3 months, with implications for health 1 Diabetic kidney disease (DKD): kidney disease that is specific to diabetes 2 • DKD is not to be confused with diabetic nephropathy (DN). DN is the presence of a single, well‑defined, identifiable kidney disease identified by progressive glomerular nephropathy directly related to diabetes 3 • DKD is a clinical diagnosis based on the measurement of e. GFR and albuminuria along with clinical characteristics of diabetes increasing the likelihood of kidney involvement, such as diabetes duration and presence of diabetic retinopathy 4 • In patients with T 2 D, other causes of CKD should be considered in cases with an absence of diabetic retinopathy, rapidly decreasing e. GFR, rapidly increasing albuminuria, presence of an abnormal urinary sediment or signs/symptoms of other systemic disease 2 CKD, chronic kidney disease; DKD, diabetic kidney disease; DN, diabetic nephropathy; e. GFR, estimated glomerular filtration rate; T 2 D, type 2 diabetes 1. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. Kidney Int Suppl 2013; 3: 1; 2. Tuttle KR et al. Am J Kidney Dis 2014; 64: 510; 3. Piccoli GB et al. Rev Diabet Stud 2015; 12: 87; 4. Alicic RZ et al. Clin J Am Soc Nephrol 2017; 12: 2032 3

Guidelines recommend that kidney function is assessed based on glomerular filtration rate and albuminuria 1 Glomerular filtration rate usually declines to levels below the reference only upon extensive kidney damage and is a measure of kidney function. Because it is difficult to measure directly, an estimated value (e. GFR) is routinely calculated using e. GFR-estimating equations Albuminuria indicates increased glomerular permeability and is a measure of kidney damage. It can be quantified by measuring the urine albumin-tocreatinine ratio (UACR)1– 3 UACR = albumin concentration (mg) creatinine concentration (g) • UACR is the preferred method for albuminuria evaluation 1, 4* • Measured using a single spot urine test (morning sample preferred). Albuminuria can also be quantified by collecting 24 -hour urine 1, 5, 6 *See slide notes for the rationale for the preferential use of UACR; >30 mg/g or ≥ 3 mg/mmol creatinine is considered pathologic UACR 1 e. GFR, estimated glomerular filtration rate; UACR, urine albumin-to-creatinine ratio 1. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. Kidney Int Suppl 2013; 3; 2. Polkinghorne KR et al. Clin Biochem Rev 2014; 35: 67; 3. Zhang A & Huang S. Int J Nephrol 2012: 313251; 4. NICE Clinical Guidance on Chronic kidney disease in adults: assessment and management [CC 182], June 2014. https: //www. nice. org. uk/guidance/cg 182/chapter/1 -Recommendations (accessed Jun 2020); 5. National Kidney Foundation 2019, https: //www. kidney. org/kidneydisease/siemens_hcp_acr (accessed Jun 2020); 6. Tuttle KR et al. Am J Kidney Dis 2014; 64: 510 4
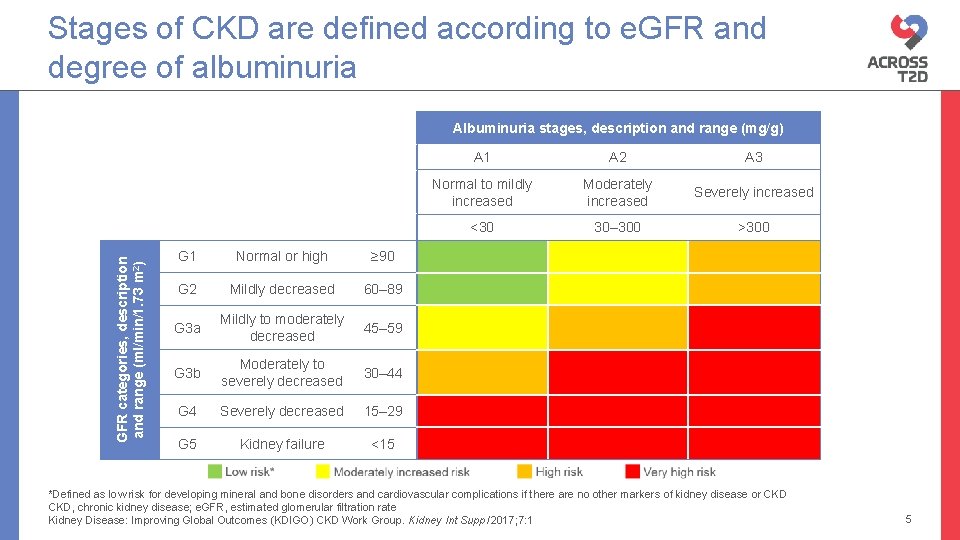
Stages of CKD are defined according to e. GFR and degree of albuminuria GFR categories, description and range (ml/min/1. 73 m 2) Albuminuria stages, description and range (mg/g) G 1 Normal or high ≥ 90 G 2 Mildly decreased 60– 89 G 3 a Mildly to moderately decreased 45– 59 G 3 b Moderately to severely decreased 30– 44 G 4 Severely decreased 15– 29 G 5 Kidney failure <15 A 1 A 2 A 3 Normal to mildly increased Moderately increased Severely increased <30 30– 300 >300 *Defined as low risk for developing mineral and bone disorders and cardiovascular complications if there are no other markers of kidney disease or CKD, chronic kidney disease; e. GFR, estimated glomerular filtration rate Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. Kidney Int Suppl 2017; 7: 1 5

T 2 D constitutes an increasing global burden Number of people with diabetes worldwide and per IDF region in 2019, 2030 and 2045 (20– 79 years) and anticipated percentage increase T 2 D accounts for 90% of diabetes worldwide North America & Caribbean 2045 | 63 million 2030 | 56 million 33% increase 2019 | 48 million Europe 2045 | 68 million 2030 | 66 million 2019 | 59 million South & Central America 2045 | 49 million 2030 | 40 million 55% increase 2019 | 32 million South-East Asia 2045 | 153 million 2030 | 115 million 2019 | 88 million 15% increase 74% increase WORLD 2045 | 700 million 2030 | 578 million 2019 | 463 million 51% increase Africa 2045 | 47 million 2030 | 29 million 2019 | 19 million 143% increase Middle East & North Africa 2045 | 108 million 2030 | 76 million 96% increase 2019 | 55 million T 2 D, type 2 diabetes International Diabetes Federation. IDF Diabetes Atlas. 9 th edn. 2019. www. idf. org/diabetesatlas (accessed Jun 2020) Western Pacific 2045 | 212 million 2030 | 197 million 2019 | 163 million 31% increase 6
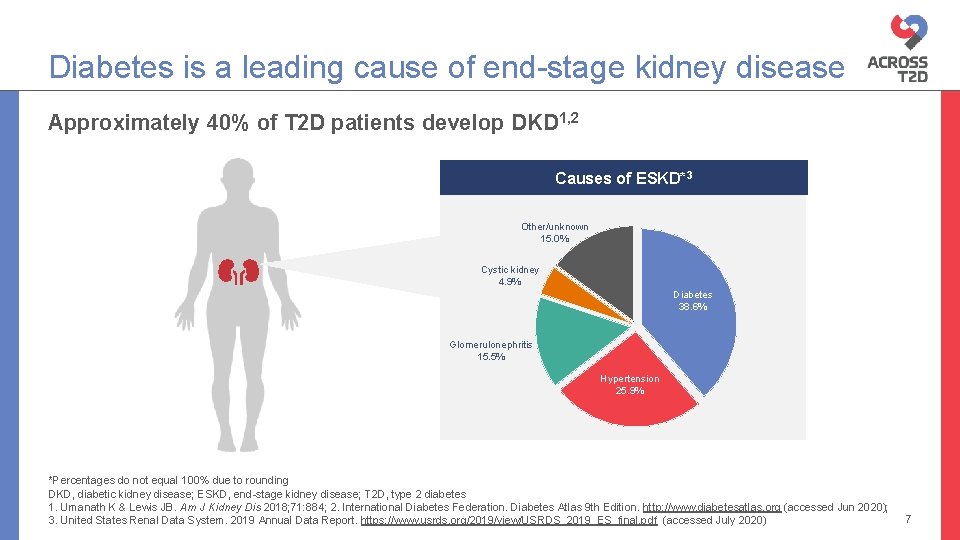
Diabetes is a leading cause of end-stage kidney disease Approximately 40% of T 2 D patients develop DKD 1, 2 Causes of ESKD*3 Other/unknown 15. 0% Cystic kidney 4. 9% Diabetes 38. 6% Diabetes 44% Glomerulonephritis 15. 5% Hypertension 25. 9% Hypertension 28. 4% *Percentages do not equal 100% due to rounding DKD, diabetic kidney disease; ESKD, end-stage kidney disease; T 2 D, type 2 diabetes 1. Umanath K & Lewis JB. Am J Kidney Dis 2018; 71: 884; 2. International Diabetes Federation. Diabetes Atlas 9 th Edition. http: //www. diabetesatlas. org (accessed Jun 2020); 3. United States Renal Data System. 2019 Annual Data Report. https: //www. usrds. org/2019/view/USRDS_2019_ES_final. pdf (accessed July 2020) 7
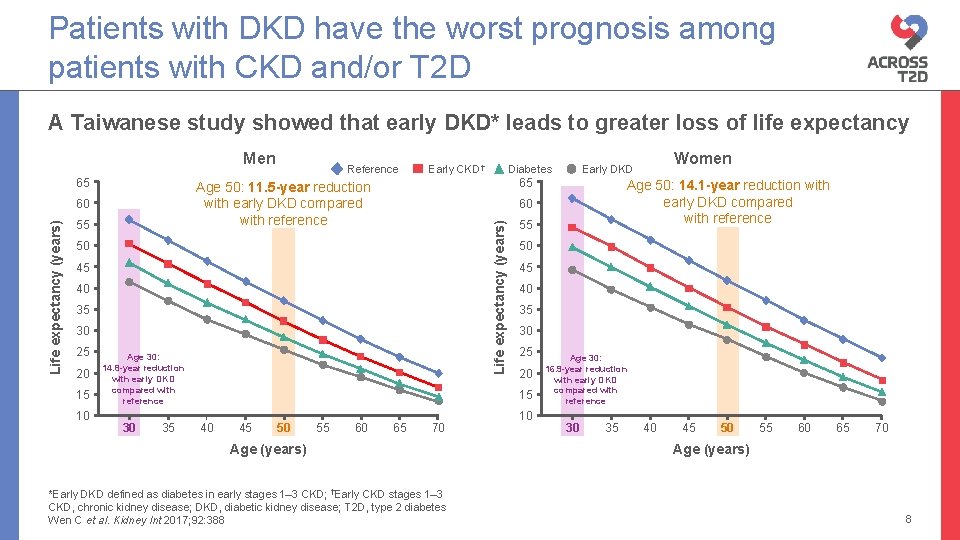
Patients with DKD have the worst prognosis among patients with CKD and/or T 2 D A Taiwanese study showed that early DKD* leads to greater loss of life expectancy Men 65 Early CKD† 50 45 40 35 30 25 20 15 10 Age 30: 14. 8 -year reduction with early DKD compared with reference 30 35 45 50 55 60 Age 50: 14. 1 -year reduction with early DKD compared with reference 55 50 45 40 35 30 25 20 15 40 65 70 Age (years) *Early DKD defined as diabetes in early stages 1– 3 CKD; †Early CKD stages 1– 3 CKD, chronic kidney disease; DKD, diabetic kidney disease; T 2 D, type 2 diabetes Wen C et al. Kidney Int 2017; 92: 388 Women Early DKD 60 Life expectancy (years) 55 Diabetes 65 Age 50: 11. 5 -year reduction with early DKD compared with reference 60 Life expectancy (years) Reference 10 Age 30: 16. 9 -year reduction with early DKD compared with reference 30 35 40 45 50 55 60 65 70 Age (years) 8
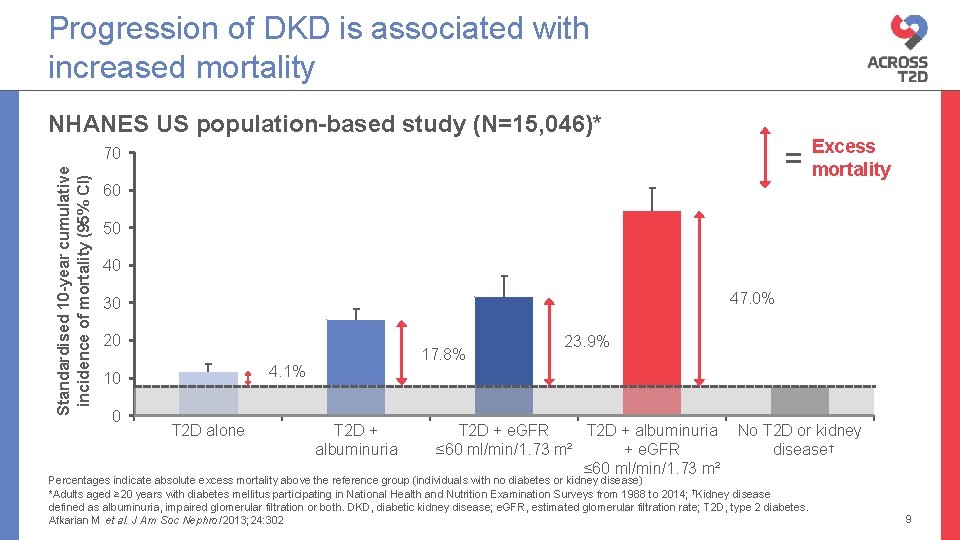
Progression of DKD is associated with increased mortality NHANES US population-based study (N=15, 046)* = Standardised 10 -year cumulative incidence of mortality (95% CI) 70 Excess mortality 60 50 40 47. 0% 30 20 4. 1% 10 0 17. 8% T 2 D alone T 2 D + albuminuria 23. 9% T 2 D + e. GFR T 2 D + albuminuria ≤ 60 ml/min/1. 73 m² + e. GFR ≤ 60 ml/min/1. 73 m² No T 2 D or kidney disease † Percentages indicate absolute excess mortality above the reference group (individuals with no diabetes or kidney disease) *Adults aged ≥ 20 years with diabetes mellitus participating in National Health and Nutrition Examination Surveys from 1988 to 2014; †Kidney disease defined as albuminuria, impaired glomerular filtration or both. DKD, diabetic kidney disease; e. GFR, estimated glomerular filtration rate; T 2 D, type 2 diabetes. Afkarian M et al. J Am Soc Nephrol 2013; 24: 302 9
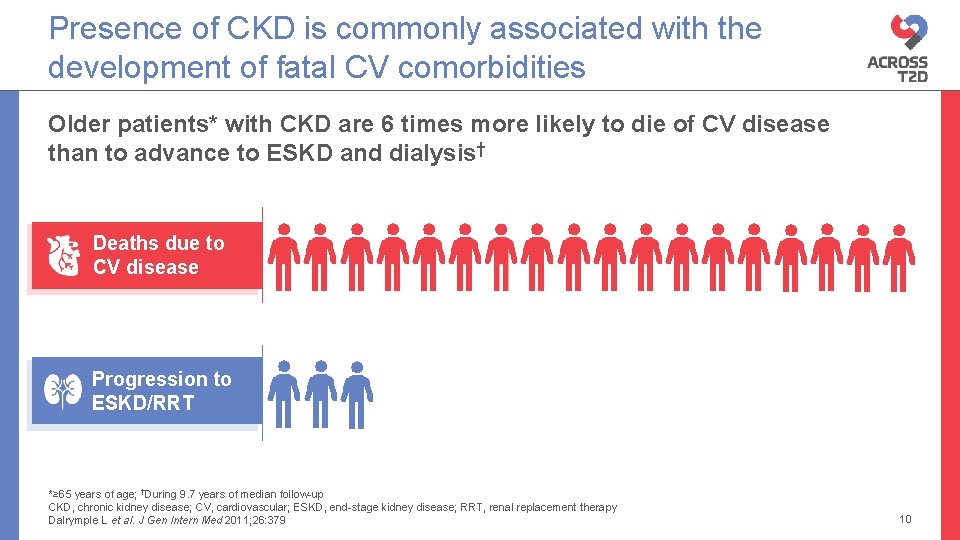
Presence of CKD is commonly associated with the development of fatal CV comorbidities Older patients* with CKD are 6 times more likely to die of CV disease than to advance to ESKD and dialysis† Deaths due to CV disease Progression to ESKD/RRT *≥ 65 years of age; †During 9. 7 years of median follow-up CKD, chronic kidney disease; CV, cardiovascular; ESKD, end-stage kidney disease; RRT, renal replacement therapy Dalrymple L et al. J Gen Intern Med 2011; 26: 379 10
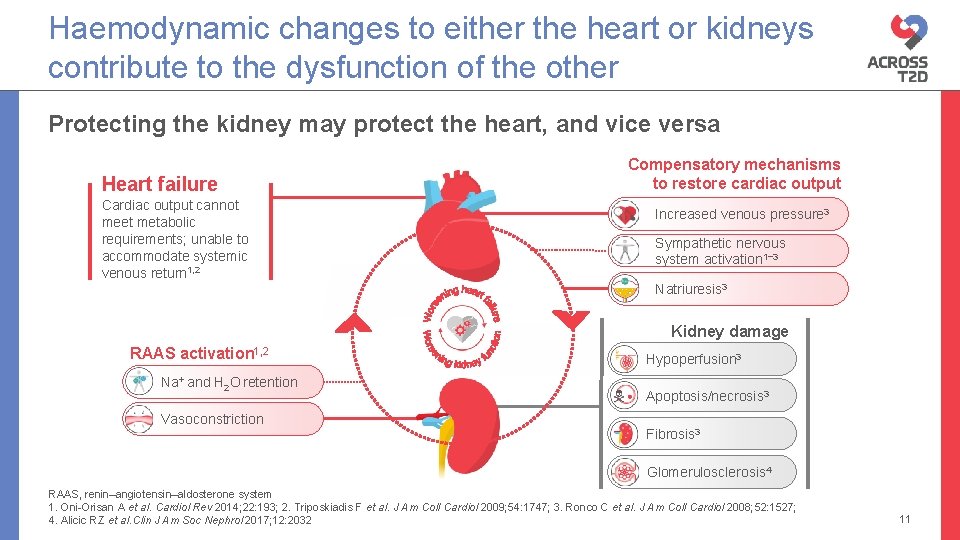
Haemodynamic changes to either the heart or kidneys contribute to the dysfunction of the other Protecting the kidney may protect the heart, and vice versa Heart failure Cardiac output cannot meet metabolic requirements; unable to accommodate systemic venous return 1, 2 Compensatory mechanisms to restore cardiac output Increased venous pressure 3 Sympathetic nervous system activation 1− 3 Natriuresis 3 Kidney damage RAAS activation 1, 2 Na+ and H 2 O retention Vasoconstriction Hypoperfusion 3 Apoptosis/necrosis 3 Fibrosis 3 Glomerulosclerosis 4 RAAS, renin–angiotensin–aldosterone system 1. Oni-Orisan A et al. Cardiol Rev 2014; 22: 193; 2. Triposkiadis F et al. J Am Coll Cardiol 2009; 54: 1747; 3. Ronco C et al. J Am Coll Cardiol 2008; 52: 1527; 4. Alicic RZ et al. Clin J Am Soc Nephrol 2017; 12: 2032 11

Contents Burden of kidney disease in type 2 diabetes Pathophysiology of diabetic kidney disease Prevention and management of diabetic kidney disease New treatments available and under evaluation
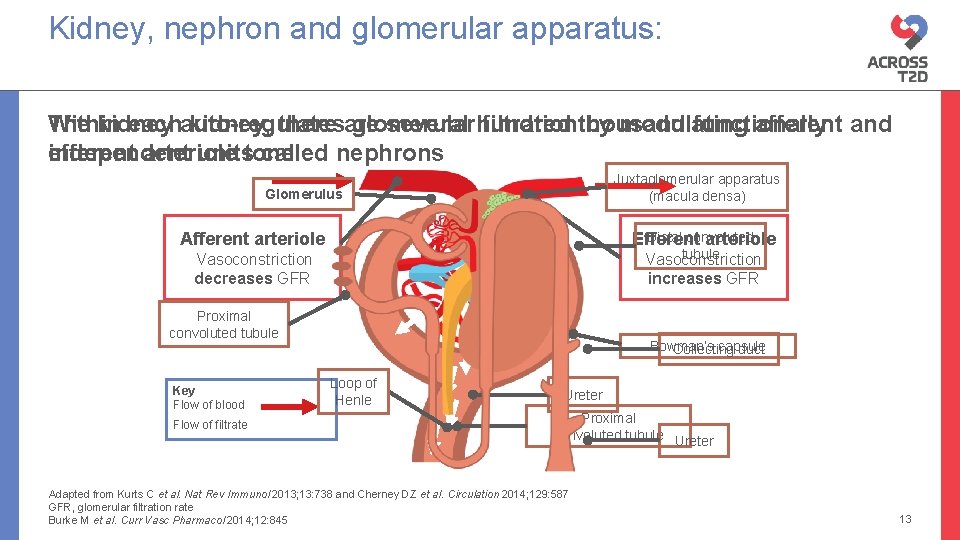
Kidney, nephron and glomerular apparatus: the kidney, the nephron, the glomerulus Within The kidney eachauto-regulates kidney, there are glomerular several hundred filtrationthousand by modulating functionally afferent and efferent arteriole independent unitstone called nephrons Juxtaglomerular apparatus (macula densa) Glomerulus Afferent arteriole Distal convoluted Efferent arteriole tubule Vasoconstriction increases GFR Vasoconstriction decreases GFR Proximal convoluted tubule Key Flow of blood Flow of filtrate Bowman’s capsule Collecting duct Loop of Henle Ureter Proximal convoluted tubule Ureter Adapted from Kurts C et al. Nat Rev Immunol 2013; 13: 738 and Cherney DZ et al. Circulation 2014; 129: 587 GFR, glomerular filtration rate Burke M et al. Curr Vasc Pharmacol 2014; 12: 845 13
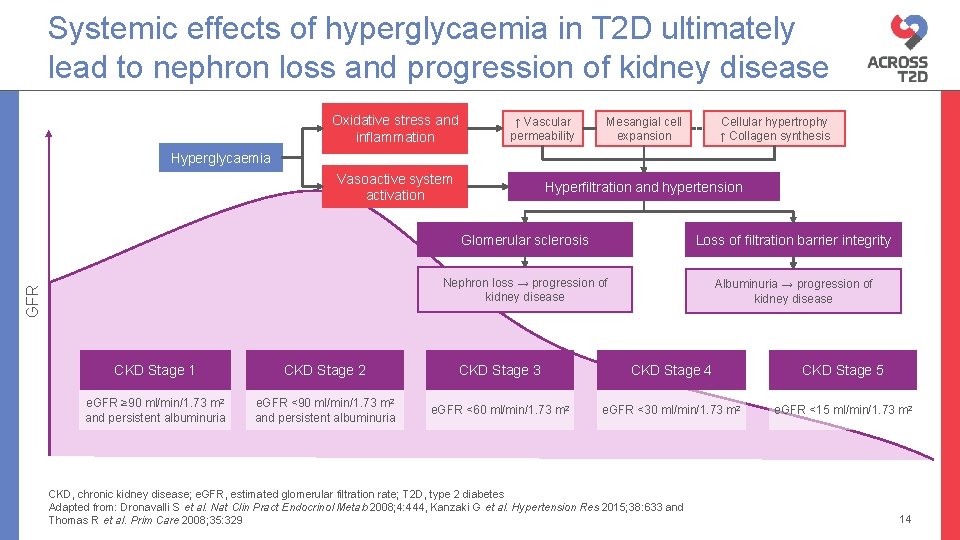
Systemic effects of hyperglycaemia in T 2 D ultimately lead to nephron loss and progression of kidney disease Oxidative stress and inflammation ↑ Vascular permeability Mesangial cell expansion Cellular hypertrophy ↑ Collagen synthesis Hyperglycaemia GFR Vasoactive system activation Hyperfiltration and hypertension Glomerular sclerosis Loss of filtration barrier integrity Nephron loss → progression of kidney disease Albuminuria → progression of kidney disease CKD Stage 1 CKD Stage 2 CKD Stage 3 CKD Stage 4 CKD Stage 5 e. GFR ≥ 90 ml/min/1. 73 m 2 and persistent albuminuria e. GFR <60 ml/min/1. 73 m 2 e. GFR <30 ml/min/1. 73 m 2 e. GFR <15 ml/min/1. 73 m 2 CKD, chronic kidney disease; e. GFR, estimated glomerular filtration rate; T 2 D, type 2 diabetes Adapted from: Dronavalli S et al. Nat Clin Pract Endocrinol Metab 2008; 4: 444, Kanzaki G et al. Hypertension Res 2015; 38: 633 and Thomas R et al. Prim Care 2008; 35: 329 14
Contents Burden of kidney disease in type 2 diabetes Pathophysiology of diabetic kidney disease Prevention and management of diabetic kidney disease New treatments available and under evaluation

ADA consensus recommendations for DKD screening Screening recommendations • “At least once a year, assess UACR and e. GFR in patients with T 1 D with duration of ≥ 5 years and in all patients with T 2 D regardless of treatment” • “Patients with urinary albumin >30 mg/g creatinine and/or an e. GFR <60 ml/min/1. 73 m 2 should be monitored twice annually to guide therapy” ADA, American Diabetes Association; DKD, diabetic kidney disease; e. GFR, estimated glomerular filtration rate; T 1 D, type 1 diabetes; T 2 D, type 2 diabetes; UACR, urine albumin-to-creatinine ratio American Diabetes Association. Diabetes Care 2020; 43: S 1 16

A multifactorial intervention strategy can slow the progression of DKD 1 • Blood glucose target is individualised for each patient, but generally Hb. A 1 c ~7%2 • Multifactorial and intensive glucose control may reduce kidney function decline 3, 4 • Blood pressure target <130/80 mm. Hg 5 • First-line antihypertensive drug treatment should be a once-daily, generic ACEi 5 • RAAS therapy with an ACEi or ARB is suggested 2 when albumin excretion ≥ 30 mg/g • Guidelines recommend that where CKD predominates, SGLT 2 inhibitors should be considered preferentially for patients with T 2 D, independently of Hb. A 1 c 6, 7 • LDL-C lowering recommended to reduce the risk of atherosclerotic events 2; statins should not be initiated in patients with T 2 D who are treated by dialysis 2 ACEi, angiotensin-converting-enzyme inhibitor; ARB, angiotensin receptor blocker; CKD, chronic kidney disease; DKD, diabetic kidney disease; Hb. A 1 c, glycated haemoglobin; LDL-C, low-density lipoprotein cholesterol; RAAS, renin–angiotensin–aldosterone system; SGLT 2, sodium-glucose co-transporter-2; T 2 D, type 2 diabetes 1. Oellgaard J et al. Kidney Int 2017; 91: 982; 2. National Kidney Foundation. Am J Kidney Dis 2012; 60: 850; 3. Zoungas S et al. Lancet Diabetes Endocrinol 2017; 5: 431; 4. Sarafidis P et al. Nephrol Dial Transplant 2019; 34: 208; 5. NICE. Clinical guideline: Type 2 diabetes in adults (NG 28) Dec 2015, https: //www. nice. org. uk/guidance/ng 28 (accessed Jun 2020); 6. Buse J et al. Diabetes Care 2020; 43: 487; 7. American Diabetes Association. Diabetes Care 2020; 43: S 1 17
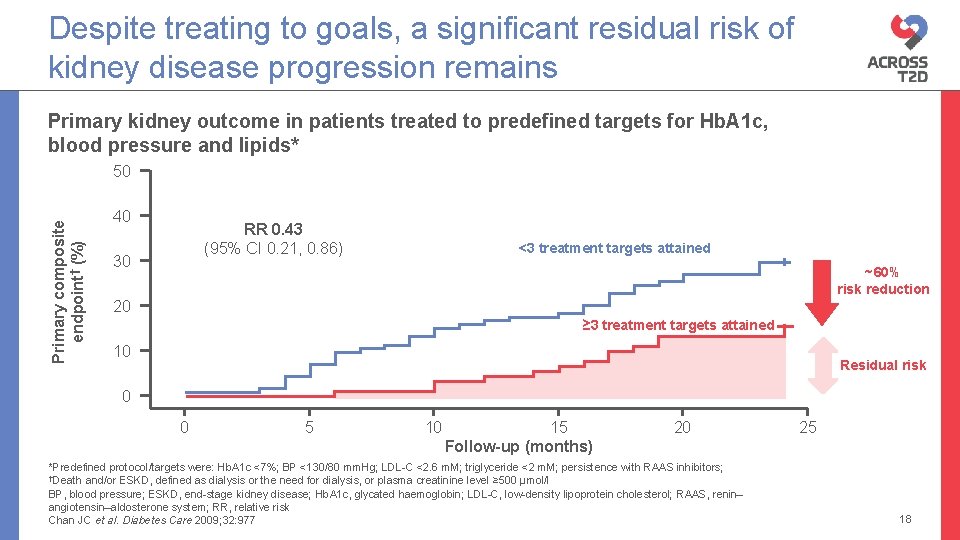
Despite treating to goals, a significant residual risk of kidney disease progression remains Primary kidney outcome in patients treated to predefined targets for Hb. A 1 c, blood pressure and lipids* Primary composite endpoint† (%) 50 40 RR 0. 43 (95% CI 0. 21, 0. 86) 30 <3 treatment targets attained ~60% risk reduction 20 ≥ 3 treatment targets attained 10 Residual risk 0 0 5 10 15 Follow-up (months) 20 *Predefined protocol/targets were: Hb. A 1 c <7%; BP <130/80 mm. Hg; LDL-C <2. 6 m. M; triglyceride <2 m. M; persistence with RAAS inhibitors; †Death and/or ESKD, defined as dialysis or the need for dialysis, or plasma creatinine level ≥ 500 µmol/l BP, blood pressure; ESKD, end-stage kidney disease; Hb. A 1 c, glycated haemoglobin; LDL-C, low-density lipoprotein cholesterol; RAAS, renin– angiotensin–aldosterone system; RR, relative risk Chan JC et al. Diabetes Care 2009; 32: 977 25 18
Contents Burden of kidney disease in type 2 diabetes Pathophysiology of diabetic kidney disease Prevention and management of diabetic kidney disease New treatments available and under evaluation

For two decades, no new treatments were identified for the treatment of DKD 1980 1985 High BP identified as DKD risk factor 1990 1995 2000 Captopril 3 Ramipril 4 (T 1 D) (CKD) 2005 2010 2015 2020 Irbesartan 5, 6 β-blockers 1 Hydralazine 2 (T 2 D) Losartan 7 (T 2 D) See notes page for abbreviations 1. Mogensen CE et al. Br Med J (Clin Res Ed) 1982; 285: 685; 2. Parving H-H et al. Lancet 1983; 1: 1175; 3. Lewis EJ et al. N Engl J Med 1993; 329: 1456; 4. The GISEN Group. Lancet 1997; 349: 1857; 5. Lewis EJ et al. N Engl J Med 2001; 345: 851; 6. Parving H-H et al. N Engl J Med 2001; 345: 870; 7. Brenner BM et al. N Engl J Med 2001; 345: 861 20
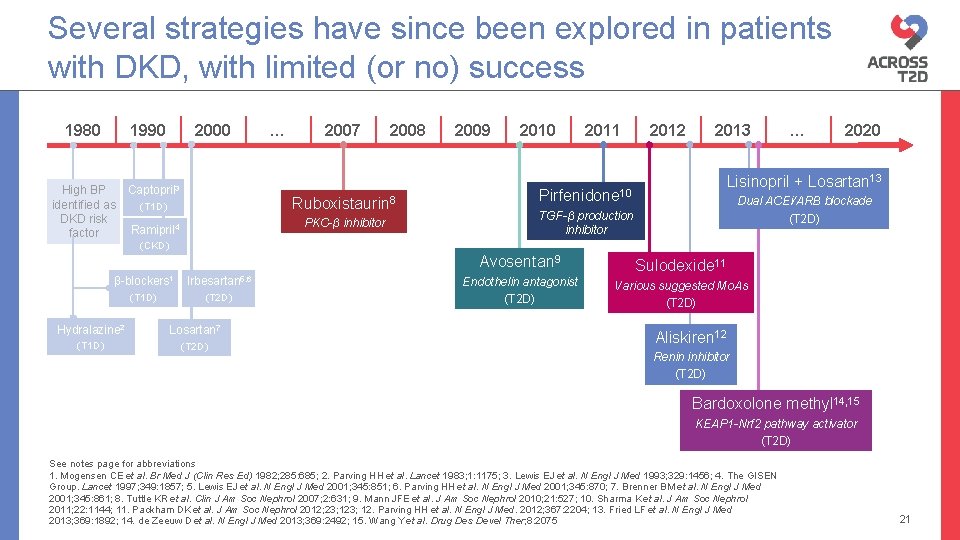
Several strategies have since been explored in patients with DKD, with limited (or no) success 1980 1990 2000 Captopril 3 High BP identified as (T 1 D) DKD risk Ramipril 4 factor … 2007 2008 Ruboxistaurin 8 PKC-β inhibitor 2009 2010 2011 2012 2013 … 2020 Lisinopril + Losartan 13 Pirfenidone 10 Dual ACEi/ARB blockade (T 2 D) TGF-β production inhibitor (CKD) β-blockers 1 Irbesartan 5, 6 (T 1 D) (T 2 D) Hydralazine 2 Losartan 7 (T 1 D) (T 2 D) Avosentan 9 Sulodexide 11 Endothelin antagonist (T 2 D) Various suggested Mo. As (T 2 D) Aliskiren 12 Renin inhibitor (T 2 D) Bardoxolone methyl 14, 15 KEAP 1 -Nrf 2 pathway activator (T 2 D) See notes page for abbreviations 1. Mogensen CE et al. Br Med J (Clin Res Ed) 1982; 285: 685; 2. Parving HH et al. Lancet 1983; 1: 1175; 3. Lewis EJ et al. N Engl J Med 1993; 329: 1456; 4. The GISEN Group. Lancet 1997; 349: 1857; 5. Lewis EJ et al. N Engl J Med 2001; 345: 851; 6. Parving HH et al. N Engl J Med 2001; 345: 870; 7. Brenner BM et al. N Engl J Med 2001; 345: 861; 8. Tuttle KR et al. Clin J Am Soc Nephrol 2007; 2: 631; 9. Mann JFE et al. J Am Soc Nephrol 2010; 21: 527; 10. Sharma K et al. J Am Soc Nephrol 2011; 22: 1144; 11. Packham DK et al. J Am Soc Nephrol 2012; 23; 12. Parving HH et al. N Engl J Med. 2012; 367: 2204; 13. Fried LF et al. N Engl J Med 2013; 369: 1892; 14. de Zeeuw D et al. N Engl J Med 2013; 369: 2492; 15. Wang Y et al. Drug Des Devel Ther; 8: 2075 21
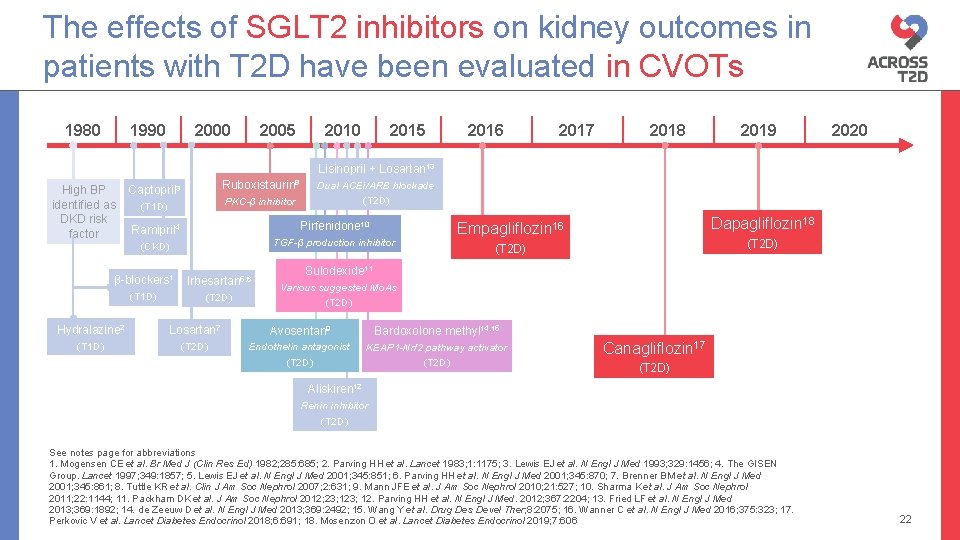
The effects of SGLT 2 inhibitors on kidney outcomes in patients with T 2 D have been evaluated in CVOTs 1980 1990 2005 2010 2015 2016 2017 2018 2019 2020 Lisinopril + Losartan 13 High BP identified as DKD risk factor Ruboxistaurin 8 Captopril 3 PKC-β inhibitor (T 1 D) Dual ACEi/ARB blockade (T 2 D) Ramipril 4 Pirfenidone 10 (CKD) TGF-β production inhibitor β-blockers 1 Irbesartan 5, 6 (T 1 D) (T 2 D) Dapagliflozin 18 Empagliflozin 16 (T 2 D) Sulodexide 11 Various suggested Mo. As (T 2 D) Hydralazine 2 Losartan 7 Avosentan 9 Bardoxolone methyl 14, 15 (T 1 D) (T 2 D) Endothelin antagonist (T 2 D) KEAP 1 -Nrf 2 pathway activator (T 2 D) Canagliflozin 17 (T 2 D) Aliskiren 12 Renin inhibitor (T 2 D) See notes page for abbreviations 1. Mogensen CE et al. Br Med J (Clin Res Ed) 1982; 285: 685; 2. Parving HH et al. Lancet 1983; 1: 1175; 3. Lewis EJ et al. N Engl J Med 1993; 329: 1456; 4. The GISEN Group. Lancet 1997; 349: 1857; 5. Lewis EJ et al. N Engl J Med 2001; 345: 851; 6. Parving HH et al. N Engl J Med 2001; 345: 870; 7. Brenner BM et al. N Engl J Med 2001; 345: 861; 8. Tuttle KR et al. Clin J Am Soc Nephrol 2007; 2: 631; 9. Mann JFE et al. J Am Soc Nephrol 2010; 21: 527; 10. Sharma K et al. J Am Soc Nephrol 2011; 22: 1144; 11. Packham DK et al. J Am Soc Nephrol 2012; 23; 12. Parving HH et al. N Engl J Med. 2012; 367: 2204; 13. Fried LF et al. N Engl J Med 2013; 369: 1892; 14. de Zeeuw D et al. N Engl J Med 2013; 369: 2492; 15. Wang Y et al. Drug Des Devel Ther; 8: 2075; 16. Wanner C et al. N Engl J Med 2016; 375: 323; 17. Perkovic V et al. Lancet Diabetes Endocrinol 2018; 6: 691; 18. Mosenzon O et al. Lancet Diabetes Endocrinol 2019; 7: 606 22
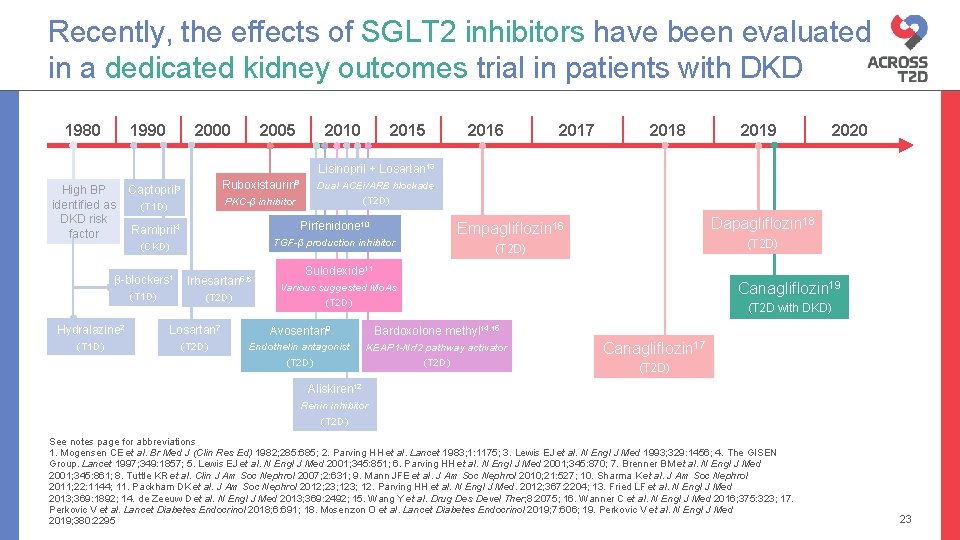
Recently, the effects of SGLT 2 inhibitors have been evaluated in a dedicated kidney outcomes trial in patients with DKD 1980 1990 2005 2010 2015 2016 2017 2018 2019 2020 Lisinopril + Losartan 13 High BP identified as DKD risk factor Ruboxistaurin 8 Captopril 3 PKC-β inhibitor (T 1 D) Dual ACEi/ARB blockade (T 2 D) Ramipril 4 Pirfenidone 10 (CKD) TGF-β production inhibitor β-blockers 1 Irbesartan 5, 6 (T 1 D) (T 2 D) Dapagliflozin 18 Empagliflozin 16 (T 2 D) Sulodexide 11 Canagliflozin 19 Various suggested Mo. As (T 2 D) Hydralazine 2 Losartan 7 Avosentan 9 Bardoxolone methyl 14, 15 (T 1 D) (T 2 D) Endothelin antagonist (T 2 D) KEAP 1 -Nrf 2 pathway activator (T 2 D) (T 2 D with DKD) Canagliflozin 17 (T 2 D) Aliskiren 12 Renin inhibitor (T 2 D) See notes page for abbreviations 1. Mogensen CE et al. Br Med J (Clin Res Ed) 1982; 285: 685; 2. Parving HH et al. Lancet 1983; 1: 1175; 3. Lewis EJ et al. N Engl J Med 1993; 329: 1456; 4. The GISEN Group. Lancet 1997; 349: 1857; 5. Lewis EJ et al. N Engl J Med 2001; 345: 851; 6. Parving HH et al. N Engl J Med 2001; 345: 870; 7. Brenner BM et al. N Engl J Med 2001; 345: 861; 8. Tuttle KR et al. Clin J Am Soc Nephrol 2007; 2: 631; 9. Mann JFE et al. J Am Soc Nephrol 2010; 21: 527; 10. Sharma K et al. J Am Soc Nephrol 2011; 22: 1144; 11. Packham DK et al. J Am Soc Nephrol 2012; 23; 12. Parving HH et al. N Engl J Med. 2012; 367: 2204; 13. Fried LF et al. N Engl J Med 2013; 369: 1892; 14. de Zeeuw D et al. N Engl J Med 2013; 369: 2492; 15. Wang Y et al. Drug Des Devel Ther; 8: 2075; 16. Wanner C et al. N Engl J Med 2016; 375: 323; 17. Perkovic V et al. Lancet Diabetes Endocrinol 2018; 6: 691; 18. Mosenzon O et al. Lancet Diabetes Endocrinol 2019; 7: 606; 19. Perkovic V et al. N Engl J Med 2019; 380: 2295 23
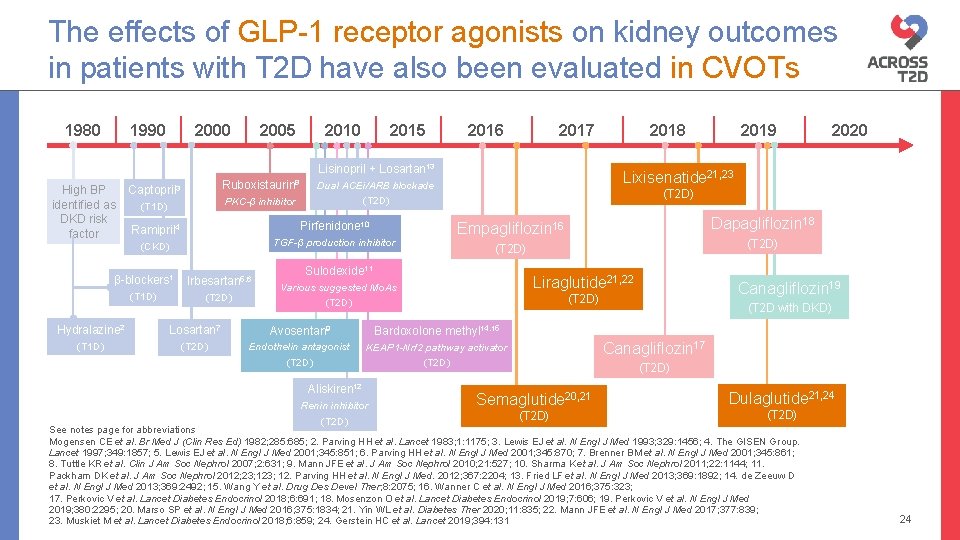
The effects of GLP-1 receptor agonists on kidney outcomes in patients with T 2 D have also been evaluated in CVOTs 1980 1990 2005 2010 2015 2016 2017 Lisinopril + Losartan 13 High BP identified as DKD risk factor Ruboxistaurin 8 Captopril 3 PKC-β inhibitor (T 1 D) Pirfenidone 10 (CKD) TGF-β production inhibitor β-blockers 1 Irbesartan 5, 6 (T 1 D) (T 2 D) Dapagliflozin 18 Empagliflozin 16 (T 2 D) Liraglutide 21, 22 Various suggested Mo. As (T 2 D) Canagliflozin 19 (T 2 D) Hydralazine 2 Losartan 7 Avosentan 9 Bardoxolone methyl 14, 15 (T 1 D) (T 2 D) Endothelin antagonist (T 2 D) KEAP 1 -Nrf 2 pathway activator (T 2 D) Renin inhibitor (T 2 D) 2020 (T 2 D) Sulodexide 11 Aliskiren 12 2019 Lixisenatide 21, 23 Dual ACEi/ARB blockade (T 2 D) Ramipril 4 2018 (T 2 D with DKD) Canagliflozin 17 (T 2 D) Semaglutide 20, 21 Dulaglutide 21, 24 (T 2 D) See notes page for abbreviations Mogensen CE et al. Br Med J (Clin Res Ed) 1982; 285: 685; 2. Parving HH et al. Lancet 1983; 1: 1175; 3. Lewis EJ et al. N Engl J Med 1993; 329: 1456; 4. The GISEN Group. Lancet 1997; 349: 1857; 5. Lewis EJ et al. N Engl J Med 2001; 345: 851; 6. Parving HH et al. N Engl J Med 2001; 345: 870; 7. Brenner BM et al. N Engl J Med 2001; 345: 861; 8. Tuttle KR et al. Clin J Am Soc Nephrol 2007; 2: 631; 9. Mann JFE et al. J Am Soc Nephrol 2010; 21: 527; 10. Sharma K et al. J Am Soc Nephrol 2011; 22: 1144; 11. Packham DK et al. J Am Soc Nephrol 2012; 23; 12. Parving HH et al. N Engl J Med. 2012; 367: 2204; 13. Fried LF et al. N Engl J Med 2013; 369: 1892; 14. de Zeeuw D et al. N Engl J Med 2013; 369: 2492; 15. Wang Y et al. Drug Des Devel Ther; 8: 2075; 16. Wanner C et al. N Engl J Med 2016; 375: 323; 17. Perkovic V et al. Lancet Diabetes Endocrinol 2018; 6: 691; 18. Mosenzon O et al. Lancet Diabetes Endocrinol 2019; 7: 606; 19. Perkovic V et al. N Engl J Med 2019; 380: 2295; 20. Marso SP et al. N Engl J Med 2016; 375: 1834; 21. Yin WL et al. Diabetes Ther 2020; 11: 835; 22. Mann JFE et al. N Engl J Med 2017; 377: 839; 23. Muskiet M et al. Lancet Diabetes Endocrinol 2018; 6: 859; 24. Gerstein HC et al. Lancet 2019; 394: 131 24
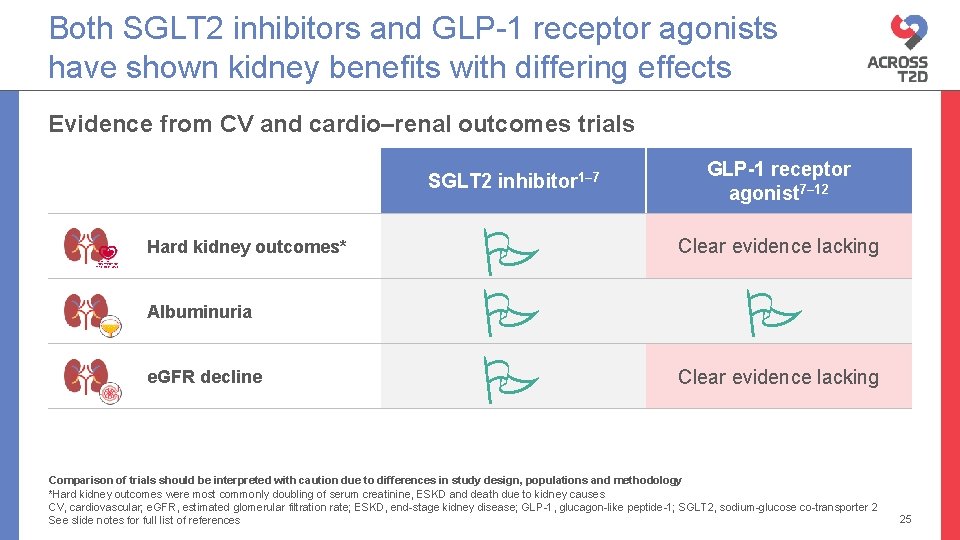
Both SGLT 2 inhibitors and GLP-1 receptor agonists have shown kidney benefits with differing effects Evidence from CV and cardio–renal outcomes trials SGLT 2 inhibitor 1– 7 Hard kidney outcomes* Albuminuria e. GFR decline GLP-1 receptor agonist 7– 12 Clear evidence lacking Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology *Hard kidney outcomes were most commonly doubling of serum creatinine, ESKD and death due to kidney causes CV, cardiovascular; e. GFR, estimated glomerular filtration rate; ESKD, end-stage kidney disease; GLP-1, glucagon-like peptide-1; SGLT 2, sodium-glucose co-transporter 2 See slide notes for full list of references 25
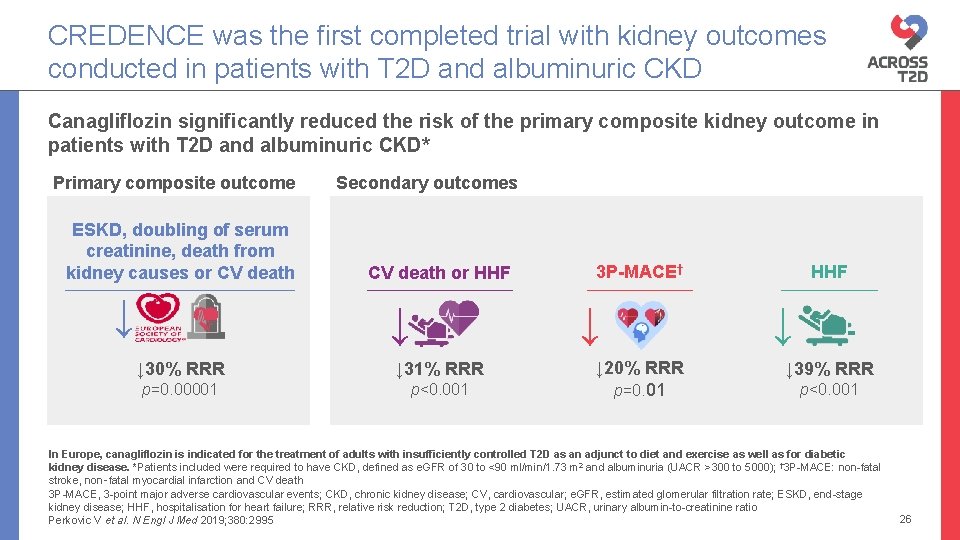
CREDENCE was the first completed trial with kidney outcomes conducted in patients with T 2 D and albuminuric CKD Canagliflozin significantly reduced the risk of the primary composite kidney outcome in patients with T 2 D and albuminuric CKD* Primary composite outcome ESKD, doubling of serum creatinine, death from kidney causes or CV death ↓ Secondary outcomes CV death or HHF ↓ ↓ ↓ 30% RRR ↓ 31% RRR p=0. 00001 p<0. 001 HHF 3 P-MACE† ↓ 20% RRR p=0. 01 ↓ ↓ 39% RRR p<0. 001 In Europe, canagliflozin is indicated for the treatment of adults with insufficiently controlled T 2 D as an adjunct to diet and exercise as well as for diabetic kidney disease. *Patients included were required to have CKD, defined as e. GFR of 30 to <90 ml/min/1. 73 m 2 and albuminuria (UACR >300 to 5000); † 3 P-MACE: non-fatal stroke, non‑fatal myocardial infarction and CV death 3 P-MACE, 3 -point major adverse cardiovascular events; CKD, chronic kidney disease; CV, cardiovascular; e. GFR, estimated glomerular filtration rate; ESKD, end-stage kidney disease; HHF, hospitalisation for heart failure; RRR, relative risk reduction; T 2 D, type 2 diabetes; UACR, urinary albumin-to-creatinine ratio Perkovic V et al. N Engl J Med 2019; 380: 2995 26

Global clinical guidelines and societies now recommend the preferential use of SGLT 2 inhibitors in patients with CKD • For patients with T 2 D and CKD, SGLT 2 inhibitors have been shown to reduce the risk of CKD progression and are recommended*1– 4 • If the SGLT 2 inhibitor is not tolerated or e. GFR is not adequate, use a GLP-1 receptor agonist with a proven CVD benefit 1 In Europe, canagliflozin is indicated for the treatment of adults with insufficiently controlled T 2 D as an adjunct to diet and exercise as well as for diabetic kidney disease. *Provided the e. GFR of the patient is within the licenced range. CKD, chronic kidney disease; CV, cardiovascular; CVD, cardiovascular disease; e. GFR, estimated glomerular filtration rate; GLP-1, glucagon-like peptide-1; HHF, hospitalisation for heart failure; SGLT 2, sodium-glucose co-transporter-2; T 2 D, type 2 diabetes 1. American Diabetes Association. Diabetes Care 2020; 43: S 1; 2. Buse JB et al. Diabetes Care 2020; 43: 487; 3. Sarafidis P et al. Nephrol Dial Transplant 2019; 34: 208; 4. Cosentino F et al. Eur Heart J 2020; 41: 255 27
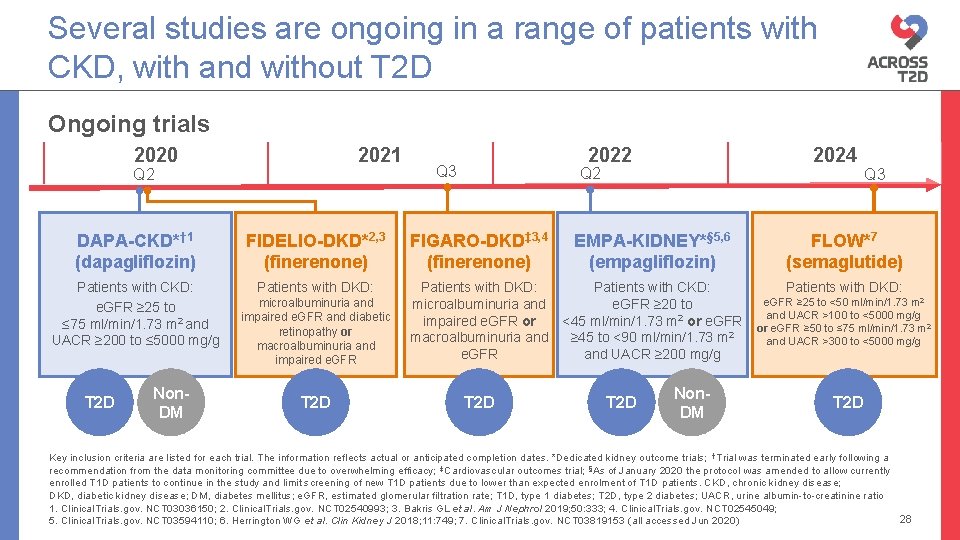
Several studies are ongoing in a range of patients with CKD, with and without T 2 D Ongoing trials 2020 2021 Q 2 2022 Q 3 2024 Q 2 Q 3 DAPA-CKD*† 1 (dapagliflozin) FIDELIO-DKD*2, 3 (finerenone) FIGARO-DKD‡ 3, 4 (finerenone) EMPA-KIDNEY*§ 5, 6 (empagliflozin) FLOW*7 (semaglutide) Patients with CKD: e. GFR ≥ 25 to ≤ 75 ml/min/1. 73 m 2 and UACR ≥ 200 to ≤ 5000 mg/g Patients with DKD: Patients with CKD: e. GFR ≥ 20 to <45 ml/min/1. 73 m 2 or e. GFR ≥ 45 to <90 ml/min/1. 73 m 2 and UACR ≥ 200 mg/g Patients with DKD: microalbuminuria and impaired e. GFR and diabetic retinopathy or macroalbuminuria and impaired e. GFR Patients with DKD: microalbuminuria and impaired e. GFR or macroalbuminuria and e. GFR T 2 D Non. DM e. GFR ≥ 25 to <50 ml/min/1. 73 m 2 and UACR >100 to <5000 mg/g or e. GFR ≥ 50 to ≤ 75 ml/min/1. 73 m 2 and UACR >300 to <5000 mg/g T 2 D Key inclusion criteria are listed for each trial. The information reflects actual or anticipated completion dates. *Dedicated kidney outcome trials; †Trial was terminated early following a recommendation from the data monitoring committee due to overwhelming efficacy; ‡Cardiovascular outcomes trial; §As of January 2020 the protocol was amended to allow currently enrolled T 1 D patients to continue in the study and limit screening of new T 1 D patients due to lower than expected enrolment of T 1 D patients. CKD, chronic kidney disease; DKD, diabetic kidney disease; DM, diabetes mellitus; e. GFR, estimated glomerular filtration rate; T 1 D, type 1 diabetes; T 2 D, type 2 diabetes; UACR, urine albumin-to-creatinine ratio 1. Clinical. Trials. gov. NCT 03036150; 2. Clinical. Trials. gov. NCT 02540993; 3. Bakris GL et al. Am J Nephrol 2019; 50: 333; 4. Clinical. Trials. gov. NCT 02545049; 28 5. Clinical. Trials. gov. NCT 03594110; 6. Herrington WG et al. Clin Kidney J 2018; 11: 749; 7. Clinical. Trials. gov. NCT 03819153 (all accessed Jun 2020)
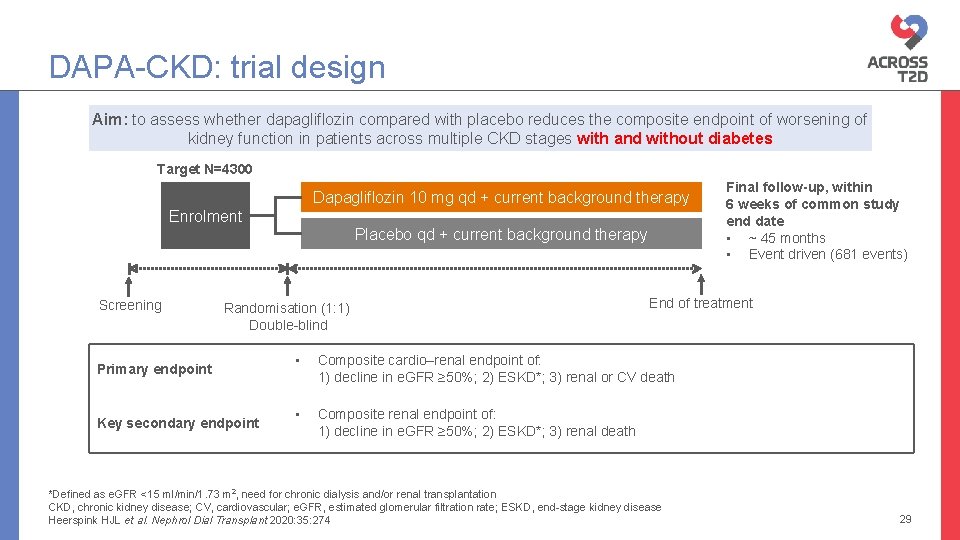
DAPA-CKD: trial design Aim: to assess whether dapagliflozin compared with placebo reduces the composite endpoint of worsening of kidney function in patients across multiple CKD stages with and without diabetes Target N=4300 Dapagliflozin 10 mg qd + current background therapy Enrolment Placebo qd + current background therapy Screening Randomisation (1: 1) Double-blind Primary endpoint Key secondary endpoint Final follow-up, within 6 weeks of common study end date • ~ 45 months • Event driven (681 events) End of treatment • Composite cardio–renal endpoint of: 1) decline in e. GFR ≥ 50%; 2) ESKD*; 3) renal or CV death • Composite renal endpoint of: 1) decline in e. GFR ≥ 50%; 2) ESKD*; 3) renal death *Defined as e. GFR <15 ml/min/1. 73 m 2, need for chronic dialysis and/or renal transplantation CKD, chronic kidney disease; CV, cardiovascular; e. GFR, estimated glomerular filtration rate; ESKD, end-stage kidney disease Heerspink HJL et al. Nephrol Dial Transplant 2020: 35: 274 29

DAPA-CKD: key inclusion and exclusion criteria Key inclusion criteria Key exclusion criteria* • Aged ≥ 18 years • T 1 D • e. GFR ≥ 25 but ≤ 75 ml/min/1. 73 m 2 at screening • UACR ≥ 200 but ≤ 5000 mg/g at screening • Autosomal dominant or autosomal recessive polycystic kidney disease, lupus nephritis or ANCA-associated vasculitis • Stable and, for the patient, maximum tolerated labelled dose of an ACE inhibitor or ARB for ≥ 4 weeks before screening, if not medically contraindicated • Receiving cytotoxic therapy, immunosuppressive therapy or other immunotherapy for primary or secondary kidney disease within 6 months prior to enrolment • Note: patients with e. GFR 60– 75 ml/min/1. 73 m 2 capped at 10% of total cohort; at least 30% of patients had to have diabetes vs non-DKD • NYHA class IV congestive heart failure *See slide notes for further exclusion criteria ACE, angiotensin-converting enzyme; ANCA, antineutrophil cytoplasmic antibodies; ARB, angiotensin receptor blocker; DKD, diabetic kidney disease; e. GFR, estimated glomerular filtration rate; NYHA, New York Heart Association; T 1 D, type 1 diabetes; UACR, urine albumin-to-creatinine ratio Heerspink HJL et al. Nephrol Dial Transplant 2020: 35: 274 30
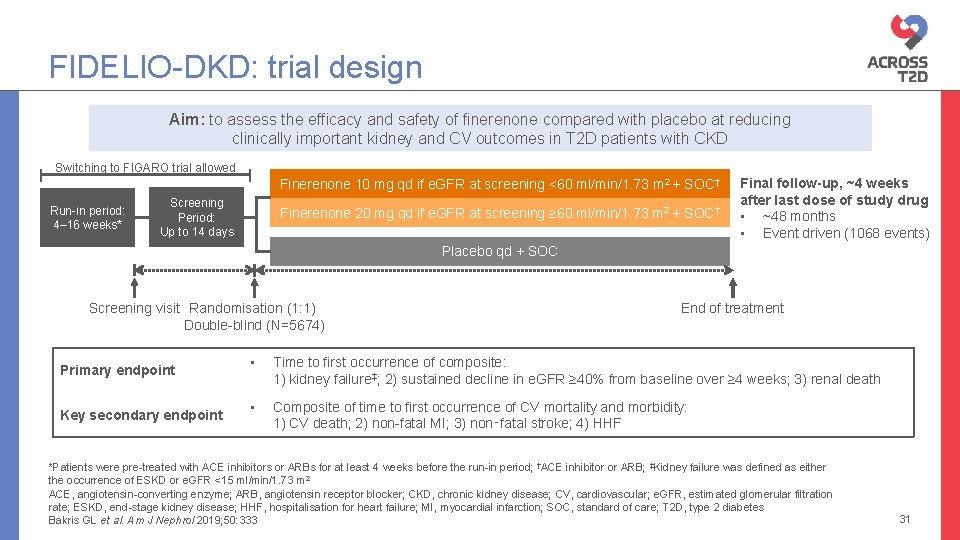
FIDELIO-DKD: trial design Aim: to assess the efficacy and safety of finerenone compared with placebo at reducing clinically important kidney and CV outcomes in T 2 D patients with CKD Switching to FIGARO trial allowed Finerenone 10 mg qd if e. GFR at screening <60 ml/min/1. 73 m 2 + SOC† Run-in period: 4– 16 weeks* Screening Period: Up to 14 days Finerenone 20 mg qd if e. GFR at screening ≥ 60 ml/min/1. 73 m 2 + SOC† Final follow-up, ~4 weeks after last dose of study drug • ~48 months • Event driven (1068 events) Placebo qd + SOC Screening visit Randomisation (1: 1) Double-blind (N=5674) Primary endpoint Key secondary endpoint End of treatment • Time to first occurrence of composite: 1) kidney failure‡; 2) sustained decline in e. GFR ≥ 40% from baseline over ≥ 4 weeks; 3) renal death • Composite of time to first occurrence of CV mortality and morbidity: 1) CV death; 2) non-fatal MI; 3) non‑fatal stroke; 4) HHF *Patients were pre-treated with ACE inhibitors or ARBs for at least 4 weeks before the run-in period; †ACE inhibitor or ARB; ‡Kidney failure was defined as either the occurrence of ESKD or e. GFR <15 ml/min/1. 73 m 2 ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CKD, chronic kidney disease; CV, cardiovascular; e. GFR, estimated glomerular filtration rate; ESKD, end-stage kidney disease; HHF, hospitalisation for heart failure; MI, myocardial infarction; SOC, standard of care; T 2 D, type 2 diabetes Bakris GL et al. Am J Nephrol 2019; 50: 333 31

FIDELIO-DKD: key inclusion and exclusion criteria Key inclusion criteria* Key exclusion criteria† • Men or women aged ≥ 18 years • Known significant non-diabetic kidney disease, including clinically relevant renal artery stenosis • Patients with T 2 D as defined by the American Diabetes Association • Patients with a clinical diagnosis of CKD: • UACR >5000 mg/g (>565 mg/mmol) at the run-in or screening visit – Persistent high albuminuria defined as UACR ≥ 30 mg/g (≥ 3. 4 mg/mmol) but <300 mg/g (<33. 9 mg/mmol) and e. GFR ≥ 25 but <60 ml/min/1. 73 m 2 (CKD-EPI) and presence of diabetic retinopathy in the medical history • Concomitant therapy with eplerenone, spironolactone, any renin inhibitor or potassium-sparing diuretic that cannot be discontinued ≥ 4 weeks before the screening visit – Or, persistent very high albuminuria defined as UACR ≥ 300 mg/g (≥ 33. 9 mg/mmol) and e. GFR ≥ 25 but <75 ml/min/1. 73 m 2 (CKD-EPI) • Concomitant therapy with ACE inhibitors and ARBs that cannot be discontinued for the purpose of the studies • Prior treatment with ACE inhibitors and ARBs • Serum potassium ≤ 4. 8 mmol/l at both the run-in and screening visits • Concomitant therapy with potent cytochrome P 450 isoenzyme 3 A 4 inhibitors or inducers (to be stopped at least 7 days before randomisation) *See reference for full inclusion criteria; †See slide notes for further exclusion criteria ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; e. GFR, estimated glomerular filtration rate; T 2 D, type 2 diabetes; UACR, urine albumin-to-creatinine ratio Bakris GL et al. Am J Nephrol 2019; 50: 333 32
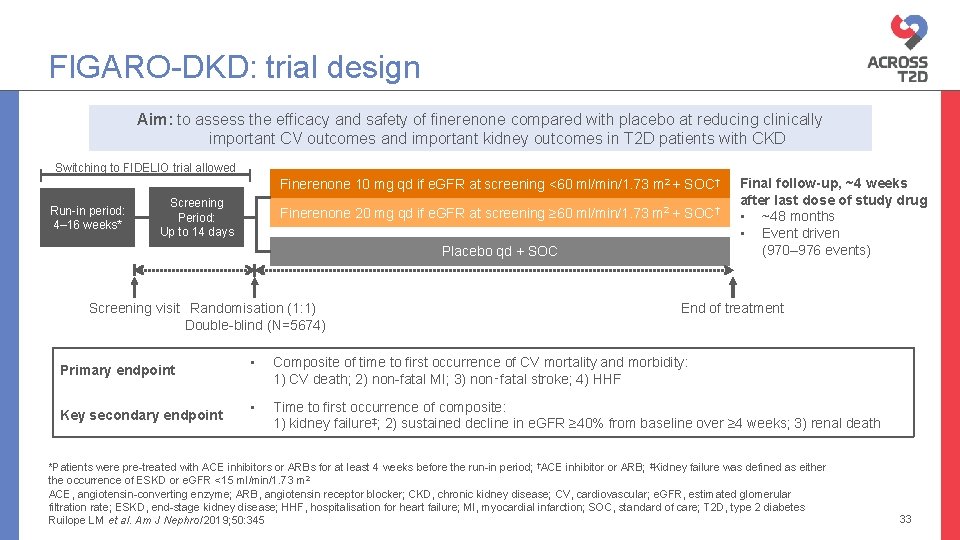
FIGARO-DKD: trial design Aim: to assess the efficacy and safety of finerenone compared with placebo at reducing clinically important CV outcomes and important kidney outcomes in T 2 D patients with CKD Switching to FIDELIO trial allowed Finerenone 10 mg qd if e. GFR at screening <60 ml/min/1. 73 m 2 + SOC† Run-in period: 4– 16 weeks* Screening Period: Up to 14 days Finerenone 20 mg qd if e. GFR at screening ≥ 60 ml/min/1. 73 m 2 + SOC† Placebo qd + SOC Screening visit Randomisation (1: 1) Double-blind (N=5674) Primary endpoint Key secondary endpoint Final follow-up, ~4 weeks after last dose of study drug • ~48 months • Event driven (970– 976 events) End of treatment • Composite of time to first occurrence of CV mortality and morbidity: 1) CV death; 2) non-fatal MI; 3) non‑fatal stroke; 4) HHF • Time to first occurrence of composite: 1) kidney failure‡; 2) sustained decline in e. GFR ≥ 40% from baseline over ≥ 4 weeks; 3) renal death *Patients were pre-treated with ACE inhibitors or ARBs for at least 4 weeks before the run-in period; †ACE inhibitor or ARB; ‡Kidney failure was defined as either the occurrence of ESKD or e. GFR <15 ml/min/1. 73 m 2 ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CKD, chronic kidney disease; CV, cardiovascular; e. GFR, estimated glomerular filtration rate; ESKD, end-stage kidney disease; HHF, hospitalisation for heart failure; MI, myocardial infarction; SOC, standard of care; T 2 D, type 2 diabetes Ruilope LM et al. Am J Nephrol 2019; 50: 345 33

FIGARO-DKD: key inclusion and exclusion criteria Key inclusion criteria* Key exclusion criteria† • Men or women aged ≥ 18 years • Known significant non-diabetic kidney disease, including clinically relevant renal artery stenosis • Patients with T 2 D as defined by the American Diabetes Association • Patients with a clinical diagnosis of CKD: • UACR >5000 mg/g (>565 mg/mmol) at the run-in or screening visit – Persistent high albuminuria defined as UACR ≥ 30 mg/g (≥ 3. 4 mg/mmol) but <300 mg/g (<33. 9 mg/mmol) and e. GFR ≥ 25 but ≤ 90 ml/min/1. 73 m 2 (CKD-EPI) in the medical history • Concomitant therapy with eplerenone, spironolactone, any renin inhibitor or potassium-sparing diuretic that cannot be discontinued ≥ 4 weeks before the screening visit – Or, persistent very high albuminuria defined as UACR ≥ 300 mg/g (≥ 33. 9 mg/mmol) and e. GFR ≥ 60 ml/min/1. 73 m 2 (CKD-EPI) • Concomitant therapy with ACE inhibitors and ARBs that cannot be discontinued for the purpose of the studies • Prior treatment with ACE inhibitors and ARBs • Serum potassium ≤ 4. 8 mmol/l at both the run-in and screening visits • Concomitant therapy with potent cytochrome P 450 isoenzyme 3 A 4 inhibitors or inducers (to be stopped at least 7 days before randomisation) *See reference for full inclusion criteria; †See slide notes for further exclusion criteria ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; e. GFR, estimated glomerular filtration rate; T 2 D, type 2 diabetes; UACR, urine albumin-to-creatinine ratio Ruilope LM et al. Am J Nephrol 2019; 50: 345 34
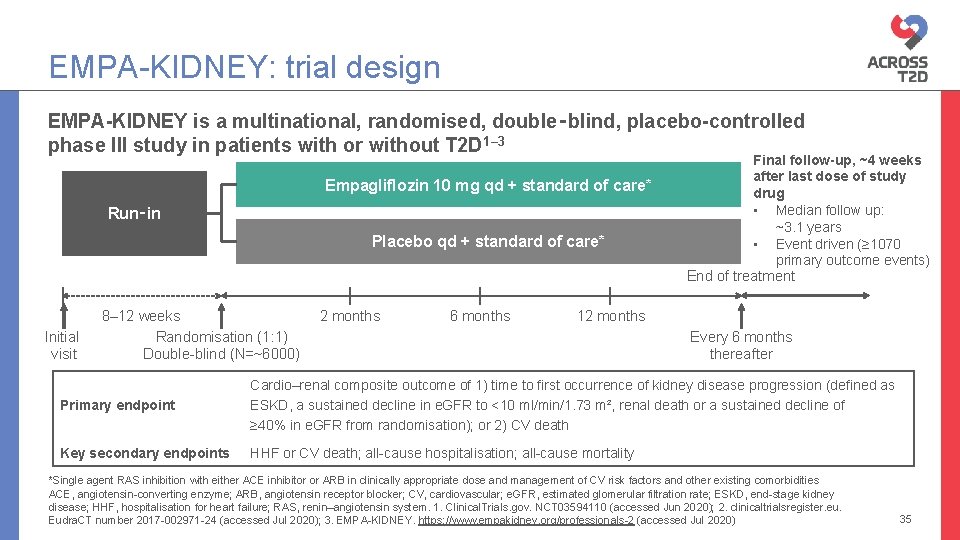
EMPA-KIDNEY: trial design EMPA-KIDNEY is a multinational, randomised, double‑blind, placebo-controlled phase III study in patients with or without T 2 D 1– 3 Empagliflozin 10 mg qd + standard of care* Run‑in Placebo qd + standard of care* Initial visit 8– 12 weeks Randomisation (1: 1) Double-blind (N=~6000) 2 months 6 months Final follow-up, ~4 weeks after last dose of study drug • Median follow up: ~3. 1 years • Event driven (≥ 1070 primary outcome events) End of treatment 12 months Every 6 months thereafter Primary endpoint Cardio–renal composite outcome of 1) time to first occurrence of kidney disease progression (defined as ESKD, a sustained decline in e. GFR to <10 ml/min/1. 73 m², renal death or a sustained decline of ≥ 40% in e. GFR from randomisation); or 2) CV death Key secondary endpoints HHF or CV death; all-cause hospitalisation; all-cause mortality *Single agent RAS inhibition with either ACE inhibitor or ARB in clinically appropriate dose and management of CV risk factors and other existing comorbidities ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CV, cardiovascular; e. GFR, estimated glomerular filtration rate; ESKD, end-stage kidney disease; HHF, hospitalisation for heart failure; RAS, renin–angiotensin system. 1. Clinical. Trials. gov. NCT 03594110 (accessed Jun 2020); 2. clinicaltrialsregister. eu. Eudra. CT number 2017 -002971 -24 (accessed Jul 2020); 3. EMPA-KIDNEY. https: //www. empakidney. org/professionals-2 (accessed Jul 2020) 35

EMPA-KIDNEY: key inclusion and exclusion criteria Key inclusion criteria Key exclusion criteria • Age ≥ 18 years • Currently receiving an SGLT 2 or SGLT 1/2 inhibitor • Evidence of CKD at risk of kidney disease progression defined by ≥ 3 months before and at the time of screening visit • T 2 D and prior atherosclerotic CV disease with e. GFR >60 ml/min/1. 73 m 2 – e. GFR ≥ 20 to <45 ml/min/1. 73 m 2, or – e. GFR ≥ 45 to <90 ml/min/1. 73 m 2 with UACR ≥ 200 mg/g • Clinically appropriate doses of single-agent RAS inhibition with either ACE inhibitor or ARB unless either not tolerated or not indicated • Receiving combined ACE inhibitor and ARB treatment • Any intravenous immunosuppression therapy in last 3 months; or anyone currently on >45 mg prednisolone (or equivalent) • Maintenance dialysis, functioning kidney transplant or scheduled living donor transplant • Polycystic kidney disease • Type 1 diabetes mellitus e. GFR calculated using CKD-EPI formula ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blockers; CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; CV, cardiovascular; e. GFR, estimated glomerular filtration rate; RAS, renin–angiotensin system; SGLT, sodium-glucose cotransporter; T 2 D, type 2 diabetes; UACR, urine albumin-to-creatinine ratio Clinical. Trials. gov. NCT 03594110 (accessed Jun 2020) 36

Summary • Kidney disease in patients with T 2 D is associated with reduced life expectancy and higher CV mortality rates • Systemic effects of hyperglycaemia in patients with T 2 D ultimately lead to nephron loss, albuminuria and progression of kidney disease • Guidelines recommend that kidney function of patients with T 2 D is assessed based on glomerular filtration rate and albuminuria • Evidence from SGLT 2 inhibitor and GLP-1 receptor agonist outcomes trials has led to major updates in clinical recommendations and guidelines – Global clinical guidelines and societies now recommend the preferential use of SGLT 2 inhibitors in patients with T 2 D and CKD, chronic kidney disease; CV, cardiovascular; GLP-1, glucagon-like peptide-1; SGLT 2, sodium-glucose co-transporter-2; T 2 D, type 2 diabetes 37
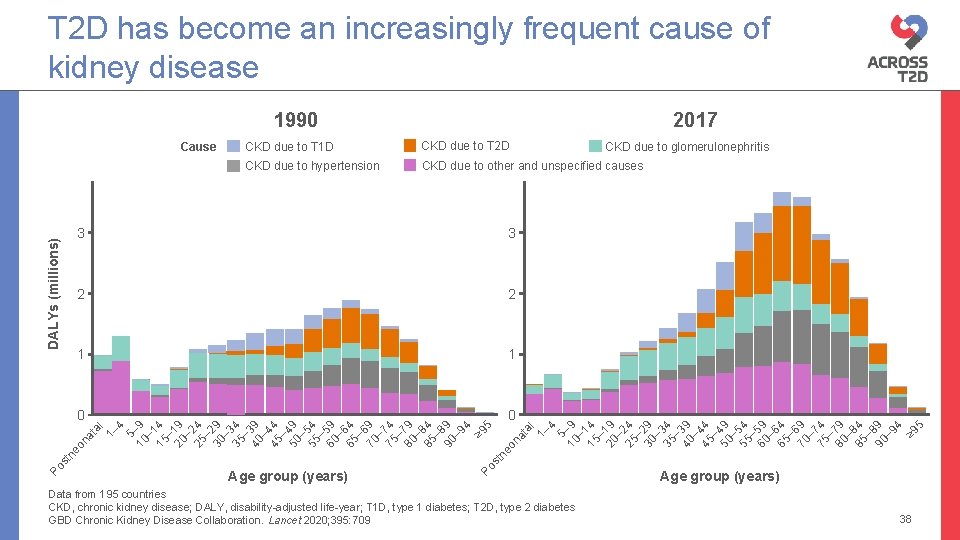
T 2 D has become an increasingly frequent cause of kidney disease 1990 CKD due to T 1 D CKD due to T 2 D CKD due to hypertension CKD due to other and unspecified causes 2 1 1 0 0 na eo tn Age group (years) Po s eo ta l 1– 4 5– 10 9 – 1 15 4 – 20 19 – 25 24 – 30 29 – 35 34 – 40 39 – 45 44 – 50 49 – 55 54 – 60 59 – 65 64 – 6 70 9 – 75 74 – 80 79 – 85 84 – 8 90 9 – 9 4 ≥ 9 5 2 ta l 1– 4 5– 10 9 – 1 15 4 – 20 19 – 25 24 – 30 29 – 35 34 – 40 39 – 45 44 – 50 49 – 55 54 – 60 59 – 65 64 – 6 70 9 – 75 74 – 80 79 – 85 84 – 8 90 9 – 9 4 ≥ 9 5 3 tn Po s CKD due to glomerulonephritis 3 na DALYs (millions) Cause 2017 Data from 195 countries CKD, chronic kidney disease; DALY, disability-adjusted life-year; T 1 D, type 1 diabetes; T 2 D, type 2 diabetes GBD Chronic Kidney Disease Collaboration. Lancet 2020; 395: 709 Age group (years) 38
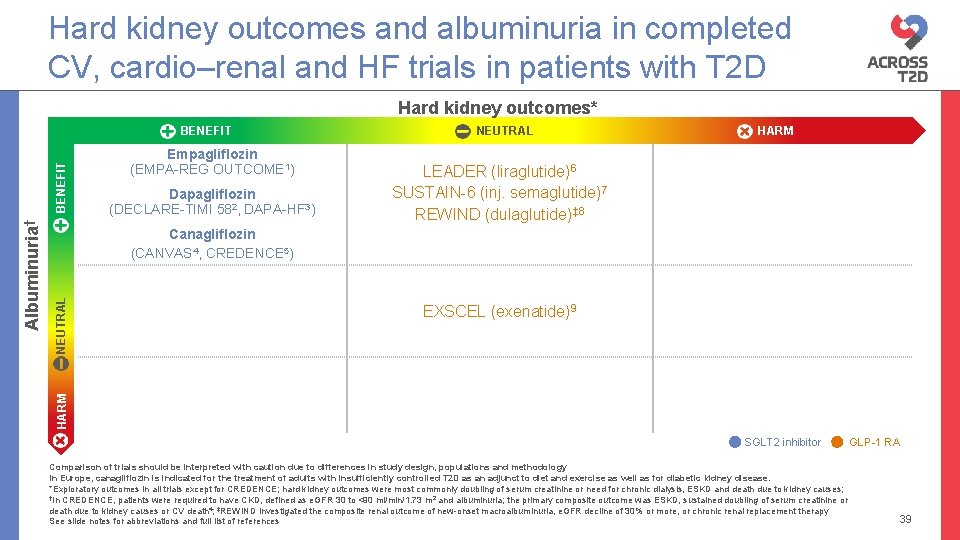
Hard kidney outcomes and albuminuria in completed CV, cardio–renal and HF trials in patients with T 2 D Hard kidney outcomes* Empagliflozin (EMPA-REG OUTCOME 1) Dapagliflozin (DECLARE-TIMI 582, DAPA-HF 3) NEUTRAL HARM LEADER (liraglutide)6 SUSTAIN-6 (inj. semaglutide)7 REWIND (dulaglutide)‡ 8 NEUTRAL Canagliflozin (CANVAS 4, CREDENCE 5) EXSCEL (exenatide)9 HARM Albuminuria† BENEFIT SGLT 2 inhibitor Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology In Europe, canagliflozin is indicated for the treatment of adults with insufficiently controlled T 2 D as an adjunct to diet and exercise as well as for diabetic kidney disease. *Exploratory outcomes in all trials except for CREDENCE; hard kidney outcomes were most commonly doubling of serum creatinine or need for chronic dialysis, ESKD and death due to kidney causes; †In CREDENCE, patients were required to have CKD, defined as e. GFR 30 to <90 ml/min/1. 73 m 2 and albuminuria; the primary composite outcome was ESKD, sustained doubling of serum creatinine or death due to kidney causes or CV death 4; ‡REWIND investigated the composite renal outcome of new-onset macroalbuminuria, e. GFR decline of 30% or more, or chronic renal replacement therapy See slide notes for abbreviations and full list of references GLP-1 RA 39

Evidence from SGLT 2 inhibitor and GLP-1 RA outcomes trials has led to major updates in clinical recommendations and guidelines Timeline of outcomes trial data release and updates to clinical recommendations and guidelines Diabetes CVD/HF DKD EMPA-REG OUTCOME 1 Empagliflozin LEADER 2 SUSTAIN-63 Liraglutide Semaglutide CANVAS Program 4 Canagliflozin 2015 2016 2017 Empagliflozin Feb 201610 Empagliflozin May 201620 Liraglutide Sep 201611 Empagliflozin May 201621 Empagliflozin Liraglutide Jan 201712 Four* CVOTs Oct 201713 Empagliflozin Aug 201722 DECLAREHarmony CREDENCE 7 REWIND 8 DAPA-HF 9 TIMI 586 Outcomes 5 Canagliflozin Dulaglutide Dapagliflozin Albiglutide Dapagliflozin 2018 Semaglutide Canagliflozin Jan 201814 Canagliflozin Mar 201823 Semaglutide Canagliflozin Apr 201815 2019 SGLT 2 i, GLP-1 RA benefits Dec 2019/Jan 202018, 19 Four*/five† CVOTs Oct 2018/Jan 201916, 17 Four CVOTs Nov 2018*24 Six CVOTs Mar 2019§ 25 2020 Six CVOTs Sep 2019¶ 26 SGLT 2 i benefits Sep 2019 17, 24, 27 SGLT 2 i CVOTs Feb 2019‡ 28 *EMPA-REG OUTCOME, LEADER, SUSTAIN-6 and CANVAS Program; †EMPA-REG OUTCOME, LEADER, SUSTAIN-6, CANVAS Program and Harmony Outcomes; ‡GLP-1 RA are also recommended but lack evidence of direct benefit on hard kidney outcomes; §EMPA-REG OUTCOME, LEADER, SUSTAIN-6, CANVAS Program, Harmony Outcomes and DECLARE TIMI-58; ¶EMPA-REG OUTCOME, LEADER, SUSTAIN-6, CANVAS Program, REWIND and DECLARE TIMI-58 See slide notes for full list of abbreviations and references 40

ADA guidelines have evolved to recommend SGLT 2 inhibitors and GLP-1 RAs with proven CV and kidney benefits Jan 2016 Jan 2017 Jan 2018 Jan 2019 Jan 2020 Glucose lowering Consider empagliflozin or liraglutide to reduce mortality (Level B) Incorporate an agent proven to reduce CV events and mortality (Level A) SGLT 2 inhibitors or GLP-1 RAs with demonstrated CVD benefit are recommended independent of Hb. A 1 c (Level A) “…incorporate an agent proven to reduce major adverse CV events and CV mortality (currently empagliflozin and liraglutide)” 3 “Among patients with ASCVD at high risk of heart failure or in whom heart failure coexists, SGLT 2 inhibitors are preferred. For patients with T 2 D and CKD, consider use of an SGLT 2 inhibitor or GLP-1 RA shown to reduce risk of CKD progression, CV events, or both” 4 “In patients with T 2 D and established HF, an SGLT 2 inhibitor may be considered to reduce risk of HHF. For patients with T 2 D and DKD, consider use of an SGLT 2 inhibitor to reduce risk of CKD progression, CV events or both. In patients with CKD who are at increased risk for CV events, use of a GLP-1 RA may reduce risk of progression of albuminuria, CV events or both” 5 “SGLT 2 inhibitors provide insulinindependent glucose lowering…These agents provide modest weight loss and blood pressure reduction” 1 “…empagliflozin or liraglutide should be considered as they have been shown to reduce CV and allcause mortality when added to standard care” 2 See slide notes for abbreviations and full list of references The Standards of Care is a “living” document where notable updates are published online, should new evidence merit immediate inclusion 6 41
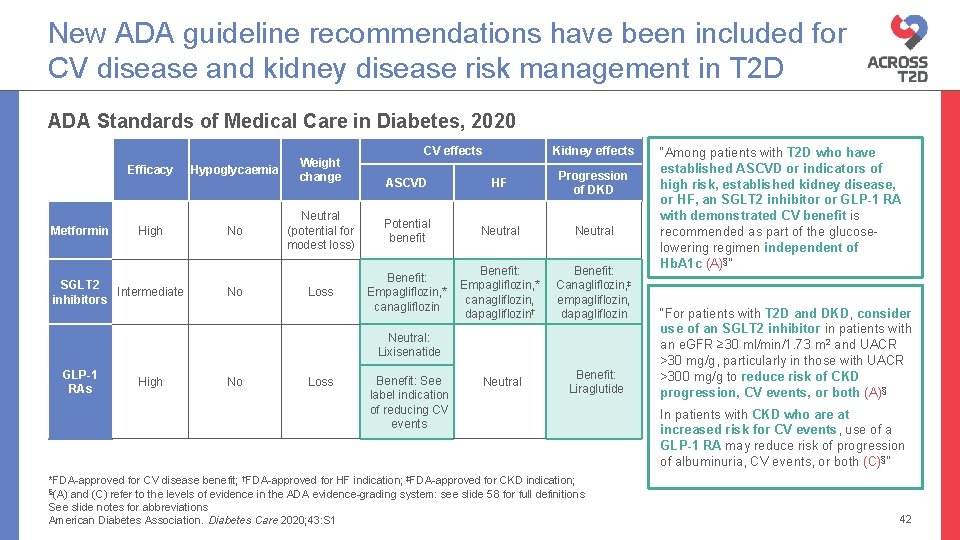
New ADA guideline recommendations have been included for CV disease and kidney disease risk management in T 2 D ADA Standards of Medical Care in Diabetes, 2020 Metformin Efficacy Hypoglycaemia Weight change High No SGLT 2 Intermediate inhibitors No CV effects Kidney effects ASCVD HF Progression of DKD Neutral (potential for modest loss) Potential benefit Neutral Loss Benefit: Empagliflozin, * canagliflozin, dapagliflozin† Benefit: Canagliflozin, ‡ empagliflozin, dapagliflozin Neutral Benefit: Liraglutide Neutral: Lixisenatide GLP-1 RAs High No Loss Benefit: See label indication of reducing CV events *FDA-approved for CV disease benefit; †FDA-approved for HF indication; ‡FDA-approved for CKD indication; §(A) and (C) refer to the levels of evidence in the ADA evidence-grading system: see slide 58 for full definitions See slide notes for abbreviations American Diabetes Association. Diabetes Care 2020; 43: S 1 “Among patients with T 2 D who have established ASCVD or indicators of high risk, established kidney disease, or HF, an SGLT 2 inhibitor or GLP-1 RA with demonstrated CV benefit is recommended as part of the glucoselowering regimen independent of Hb. A 1 c (A)§” “For patients with T 2 D and DKD, consider use of an SGLT 2 inhibitor in patients with an e. GFR ≥ 30 ml/min/1. 73 m 2 and UACR >30 mg/g, particularly in those with UACR >300 mg/g to reduce risk of CKD progression, CV events, or both (A)§ In patients with CKD who are at increased risk for CV events, use of a GLP-1 RA may reduce risk of progression of albuminuria, CV events, or both (C)§” ( 42
- Slides: 42