DIABETES MELLITUS TYPE 2 PRESENTER Esther Mary Mathew

DIABETES MELLITUS TYPE 2 PRESENTER: Esther Mary Mathew M. Sc Nursing 1 st year MODERATOR : Ms. Ujjwal Dahiya Lecturer, CON AIIMS.

INTRODUCTION Type 2 diabetes is sometimes called a “life style” disease as it more common in people who don’t do enough exercise, have an unhealthy diet and obese. Type 2 Diabetes was previously seen mainly in older adults, however it is becoming more common in young children due to obesity and overweight children.

HISTORY The earliest known record of diabetes was written on 3 rd Dynasty Egyptian papyrus by physician ‘Hesy-Ra’. He stated recurring urination as a sign of this illness

HISTORY The Indian physician Sushruta in the 6 th century B. C. noticed the sweet nature of urine in such patients and termed the condition MADHUMEHA.
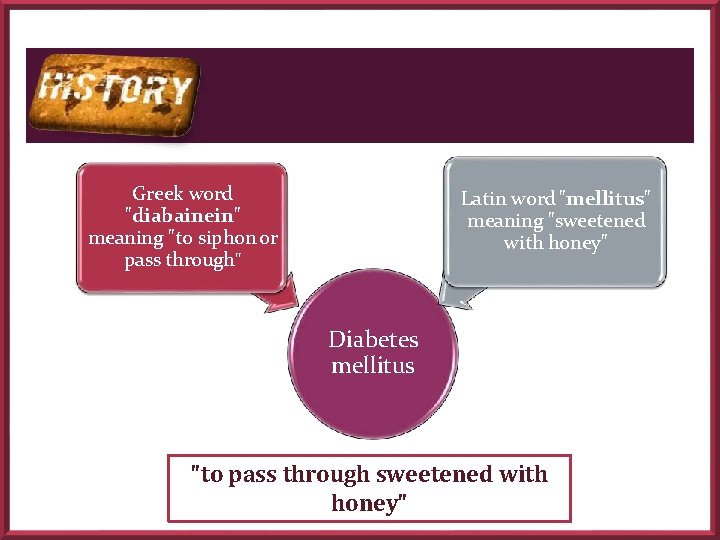
Greek word "diabainein" meaning "to siphon or pass through" Latin word "mellitus" meaning "sweetened with honey" Diabetes mellitus "to pass through sweetened with honey"

DIABETIC MELLITUS Diabetes is a group of metabolic diseases characterized by hyperglycemia resulting from defects in insulin secretion, insulin action, or both.

EPIDEMIOLOGY Globally 382 million people had diabetes in 2013 By 2035, this number will rise to 592 million In India 65. 1 million people had diabetes in 2013 By 2035, this number will increase by 70. 6%
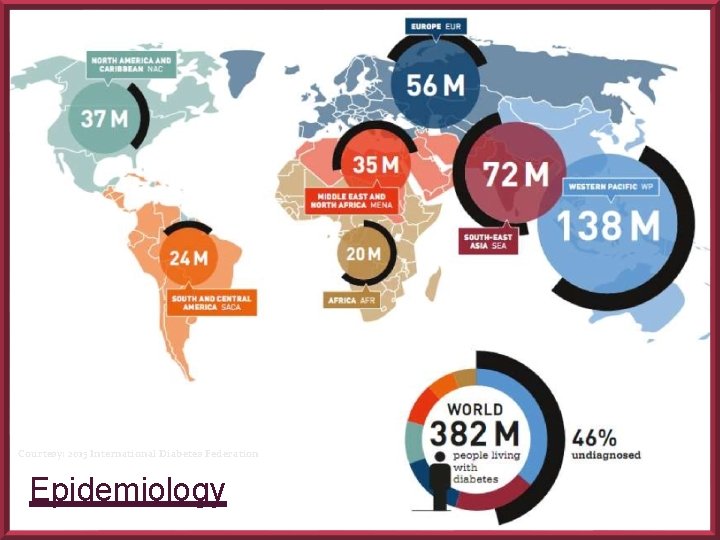
Courtesy: 2015 International Diabetes Federation Epidemiology

ANATOMY OF PANCREAS The adult pancreas is a transversely oriented retroperitoneal organ extending from the "C " loop of the duodenum to the hilum of the spleen EXOCRINE secretion pancreatic juice enzymes promote the digestion of carbohydrates, proteins and fats ENDOCRINE secretion Insulin and glucagon- enter portal vein – transported directly to the liver – regulate metabolism of carbohydrates, proteins and fats.

PANCREAS 15 - 20% α cells synthesize and secrete GLUCAGON 70 - 80% β cells synthesize and secrete INSULIN 1 -8% δ cells synthesize and secrete STOMATOSTATIN and GASTRIN 1 -2% F- cells secrete PANCREATIC POLYPEPTIDE which decreases the absorption of food from the GIT
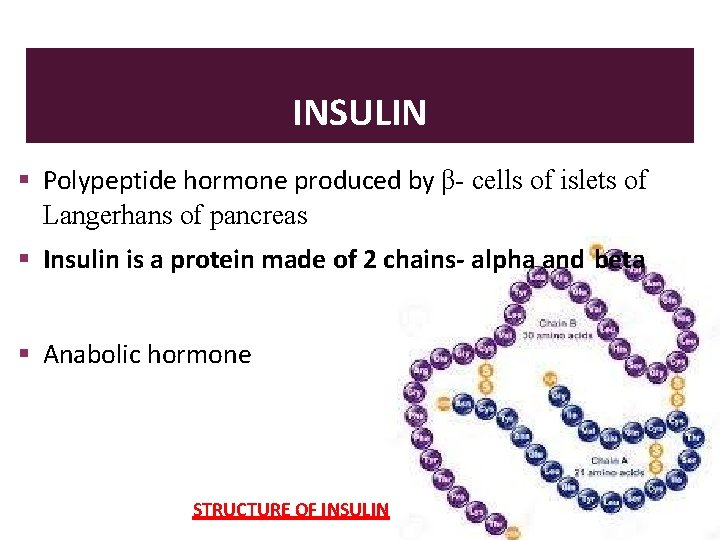
INSULIN Polypeptide hormone produced by β- cells of islets of Langerhans of pancreas Insulin is a protein made of 2 chains- alpha and beta Anabolic hormone STRUCTURE OF INSULIN

REGULATION OF INSULIN SECRETION Factors stimulating insulin secretion : Glucose: The effect is more predominant when glucose is administered orally. Arise in blood glucose level is a signal for insulin secretion. amino acids: gastrointestinal hormones: Gastrointestinal hormones (secretin, gastrin) enhance the secretion of insulin.

REGULATION OF INSULIN SECRETION Factors inhibiting insulin secretion • Epinephrine is the most potent inhibitor of insulin release. • In emergency situations like stress, extreme exercise and trauma, the nervous system stimulates adrenal medulla to release epinephrine. • Epinephrine suppresses insulin release and promotes energy metabolism by Mobilizing energy-yielding compounds-glucose from liver and fatty acids from adipose tissue

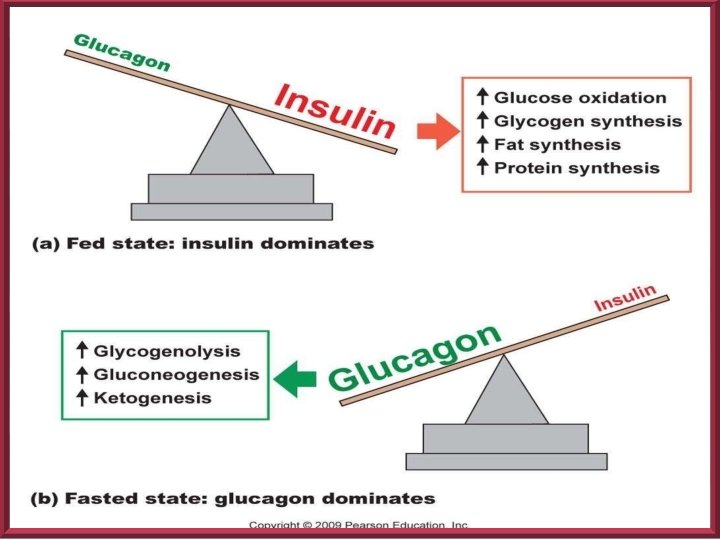
Gluconeogenesis : The synthesis of glucose from noncarbohydrate precursors( e. g. amino acids, glycerol) Glycogenesis: The formation of glycogen from glucose. Glycogenolysis : The breakdown of glycogen to glucose
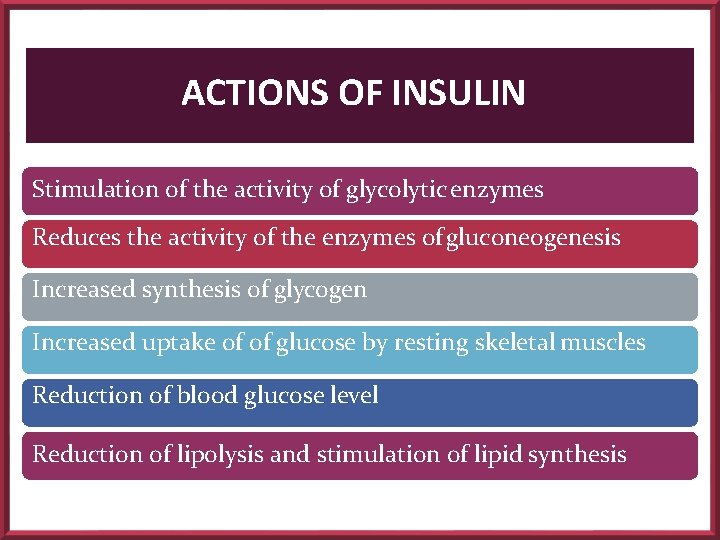
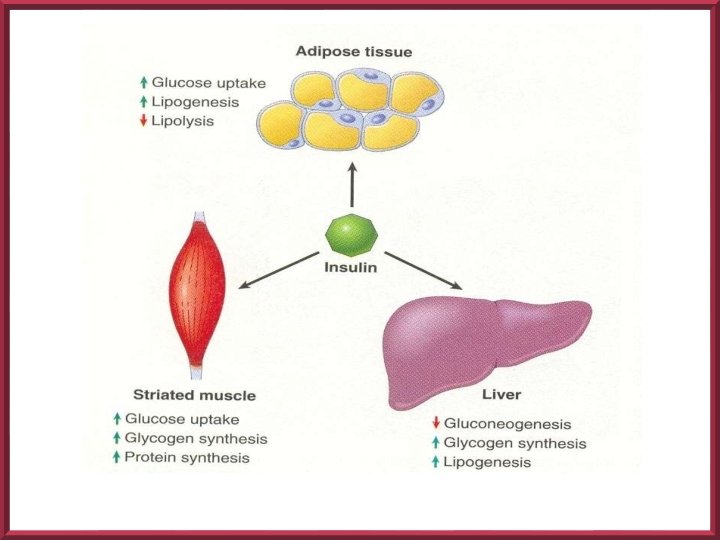
ACTIONS OF INSULIN Stimulation of the activity of glycolytic enzymes Reduces the activity of the enzymes of gluconeogenesis Increased synthesis of glycogen Increased uptake of of glucose by resting skeletal muscles Reduction of blood glucose level Reduction of lipolysis and stimulation of lipid synthesis

INSULIN Pancreas secretes 40 -50 units of insulin daily in two steps: • Secreted at low levels during fasting ( basal insulin secretion • Increased levels after eating (prandial) • An early burst of insulin occurs within 10 minutes of eating • Then proceeds with increasing release as long as hyperglycemia is present
GLUCOSE HOMEOSTASIS

CLASSIFICATION Classification by American diabetic association 2009 : • Type 1 diabetes • Type 2 diabetes • Gestational diabetes mellitus (GDM) • Secondary DM: Hormonal problems, pancreatic disorders, drugs

TYPE 1 DM Juvenile / IDDM (5 to 10%) Autoimmune destruction of pancreatic beta cells. Individual has an absolute insulin deficiency and no longer produces insulin. Such patients are absolutely dependent on exogenously administered insulin for survival.
TYPE 2 DIABETES MELLITUS

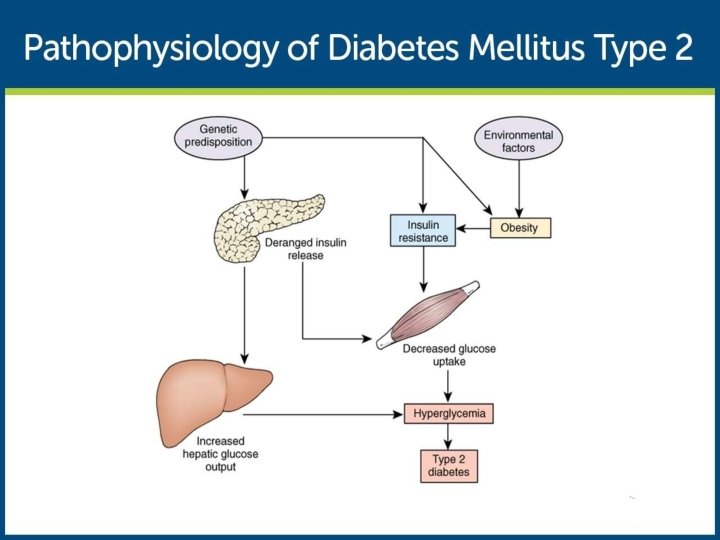
TYPE 2 DM Most common type Comprises 90 to 95% of DM cases Most type 2 DM patients are overweight, and most are diagnosed as adults. Approximately half of the patients are unaware of their disease
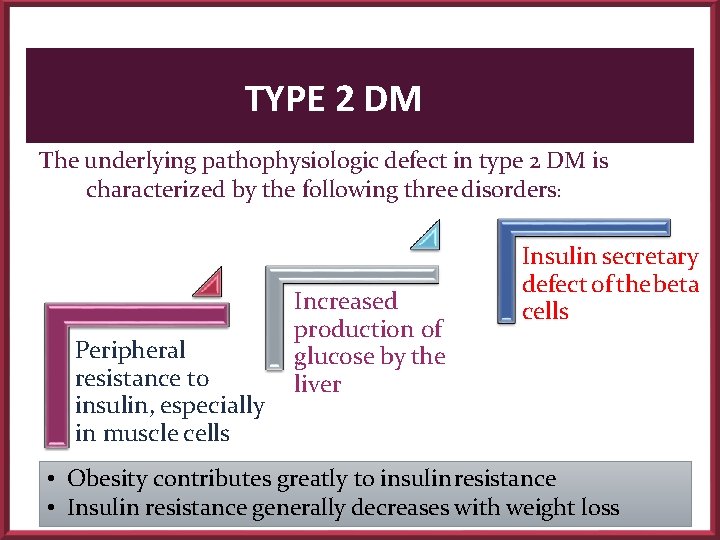
TYPE 2 DM The underlying pathophysiologic defect in type 2 DM is characterized by the following three disorders: Peripheral resistance to insulin, especially in muscle cells Increased production of glucose by the liver Insulin secretary defect of the beta cells • Obesity contributes greatly to insulin resistance • Insulin resistance generally decreases with weight loss
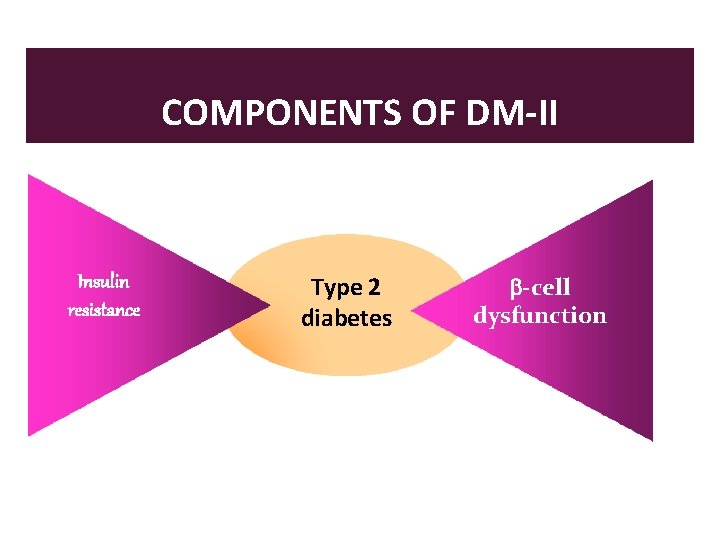
COMPONENTS OF DM-II Insulin resistance Type 2 diabetes -cell dysfunction
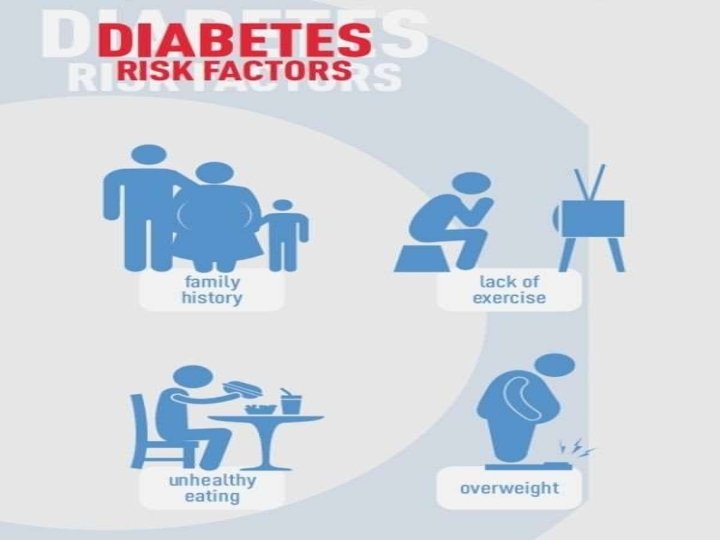

RISK FACTORS NON-MODIFIABLE: Age: 45 or more Race : African American, Asian American, Hispanic or Latino. Familial history : a parent, or siblings with diabetes.

RISK FACTORS MODIFIABLE: �Pre diabetes �Heart and blood disease �Hypertension �Low HDL cholesterol and high triglycerides. �Obesity �Polycystic ovary syndrome �Physical inactivity

RESEARCH INPUT High Bone Mineral Density and Fracture Risk in Type 2 Diabetes as Skeletal Complications of Inadequate Glucose Control. Ling Oei, Abbas, Karol Estrada et al Journal : Diabetes Care 2013 Jun; 36(6) Objective: To examine the influence of glucose control on skeletal complications.
RESEARCH INPUT RESEARCH DESIGN AND METHODS: prospective population-based study 420 participants with type 2 diabetes were classified by glucose control - according to Hb. A 1 c �adequately controlled diabetes (ACD: n = 203; Hb. A 1 c <7. 5%) �inadequately controlled diabetes (ICD; n = 217; Hb. A 1 c≥ 7. 5%) �no diabetes (n = 3, 715)
RESEARCH INPUT RESULTS : The ICD group had 1. 1– 5. 6% higher BMD, and 1. 2 to − 1. 8% narrower femoral necks than ACD and ND, respectively. Participants with ICD had 47– 62% higher fracture risk than individuals without diabetes whereas those with ACD had a risk similar to those without diabetes. CONCLUSIONS : Poor glycemic control in type 2 diabetes is associated with fracture risk.
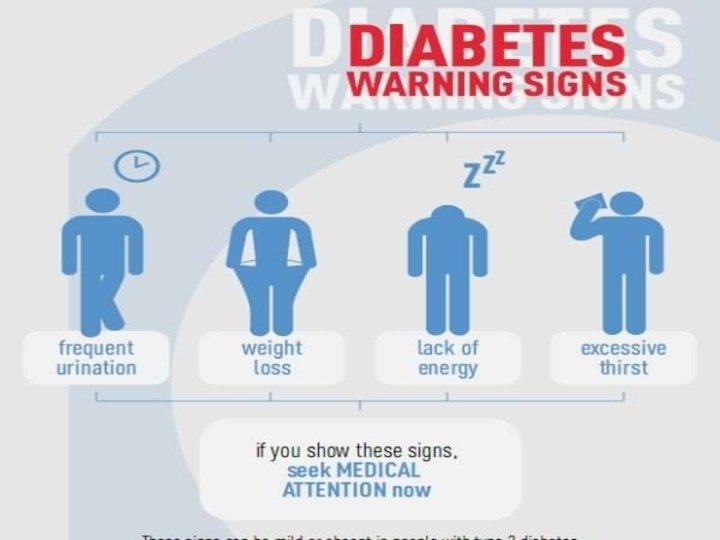

CLINICAL PRESENTATION Patients can be asymptomatic Polyuria Polydipsia Polyphagia Fatigue Weight loss Most patients are discovered while performing urine glucose screening
DIAGNOSIS Fasting Plasma Glucose Oral Glucose Tolerance Test (OGTT) Glycoselated Hemoglobin (Hb. A 1 c) Urinalysis • Glycosuria • Ketone bodies

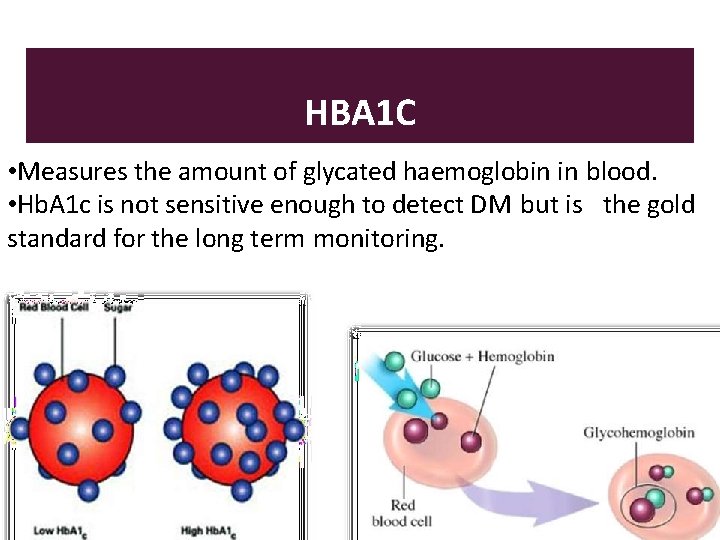
HBA 1 C • Measures the amount of glycated haemoglobin in blood. • Hb. A 1 c is not sensitive enough to detect DM but is the gold standard for the long term monitoring.

ADDITIONAL INVESTIGATIONS; Lipid profile Fundoscopic examination LFT , Urine analysis ECG Test to assess other complications

DIAGNOSTIC CRITERIA • Classic signs of HYPERGLYSEMIA with CPG ≥ 200 mg/ • OGTT ≥ 200 mg/d. L • FPG ≥ 126 mg/d. L • A 1 C ≥ 6. 5% Any one test should be confirmed with a second test, most often fasting plasma glucose (FPG).
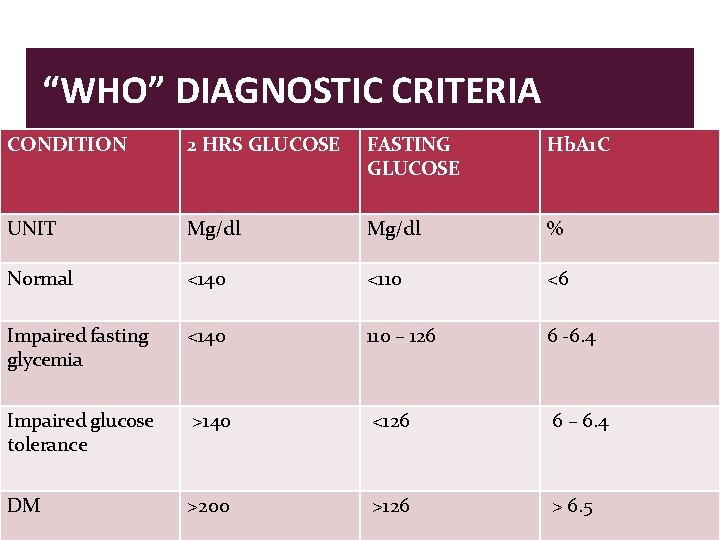
“WHO” DIAGNOSTIC CRITERIA CONDITION 2 HRS GLUCOSE FASTING GLUCOSE Hb. A 1 C UNIT Mg/dl % Normal <140 <110 <6 Impaired fasting glycemia <140 110 – 126 6 -6. 4 Impaired glucose tolerance >140 <126 6 – 6. 4 DM >200 >126 > 6. 5

MANAGEMENT OF DIABETES MELLITUS
MANAGEMENT OF DM The major components of the treatment of diabetes are: A • Medical Nutrition Therapy(Diet and Exercise) B • Oral hypoglycaemic therapy C • Insulin

A. DIET �Dietary treatment should aim at: Ensuring weight control. Providing nutritional requirements. Allowing good glycaemic control with blood glucose levels as close to normal as possible. Correcting any associated blood lipid abnormalities.

DIETARY MANAGEMENT Follow individualized meal plan and snacks as scheduled Balanced diabetic diet – 50% CHO, 30% fats, 20% CHON, vitamins and minerals diet based on pts. size, wt. , age, occupation and activity. Meal should include more fiber and starch and fewer simple or refined sugars.

DIETARY MANAGEMENT If taking insulin, eat extra food before periods of vigorous exercise Routine blood glucose testing before each meal and at bedtime is necessary during initial control, during illness and in unstable pts. Excessive salt intake is to be avoided. It should be particularly restricted in people with hypertension and those with nephropathy

How to eat low GI food Eat grains in the least processed state possible. Limit potatoes and refined grain products. Avoid concentrated sweets (jellies, jams, cakes, ice cream) Choose foods with healthy fats. Have 3 meals and one or two snacks each day Eat slowly and stop when full. Avoid periods of fasting and feasting, Do not skip meals

EXERCISE �Physical activity promotes weight reduction and improves insulin sensitivity, thus lowering blood glucose levels. �Exercise same time and duration of day. �People should, however, be educated about the potential risk of hypoglycaemia and how to avoid it. �Avoid during poor metabolic control. �Avoid trauma to extremities.


EXERCISE PRECAUTIONS Patients who have BS >250 mg/dl and who have urine ketones should not begin exercise until urine tests are NEGATIVE. Use of proper footwear. Avoid exercise in extreme heat or cold Have snacks after the exercise , to avoid post exercise hypoglycemia.
- Slides: 50