Diabetes Mellitus in Children Dr Nadeem Zubairi D

Diabetes Mellitus in Children Dr Nadeem Zubairi
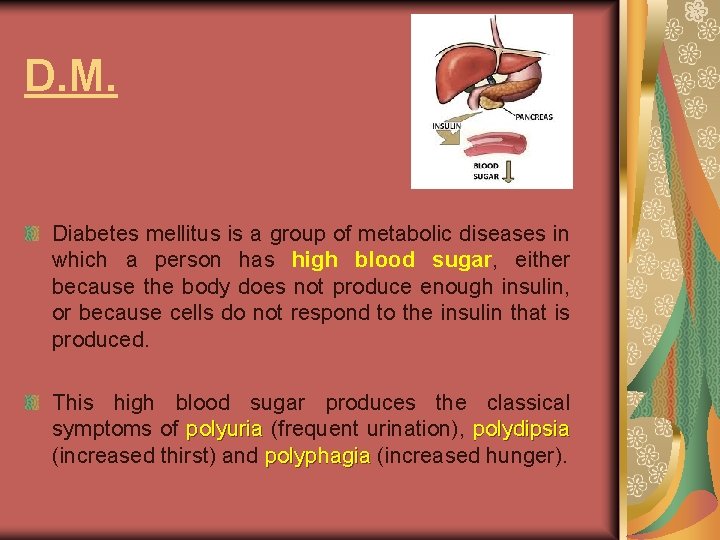
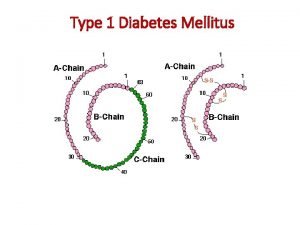
D. M. Diabetes mellitus is a group of metabolic diseases in which a person has high blood sugar, either because the body does not produce enough insulin, or because cells do not respond to the insulin that is produced. This high blood sugar produces the classical symptoms of polyuria (frequent urination), polydipsia polyuria polydipsia (increased thirst) and polyphagia (increased hunger). polyphagia

Before Insulin JL on 12/15/22 and 2 mos later Before insulin was discovered in 1921, everyone with type 1 diabetes died within weeks to years of its onset © 2004, John Walsh, P. A. , C. D. E.
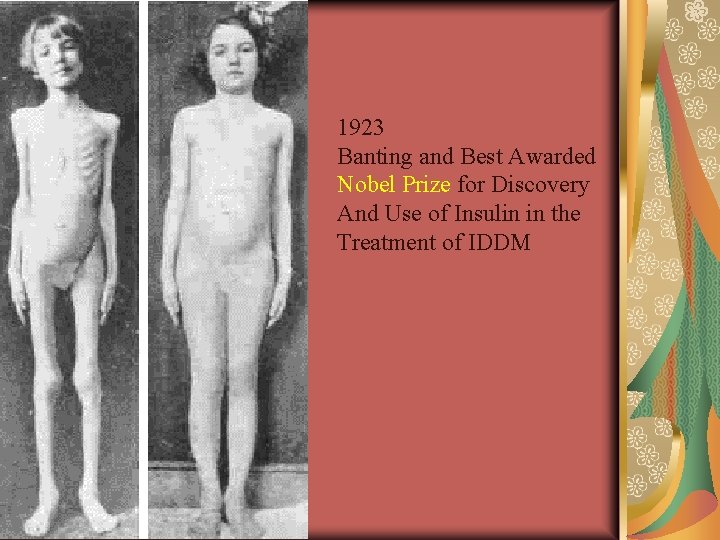
1923 Banting and Best Awarded Nobel Prize for Discovery And Use of Insulin in the Treatment of IDDM

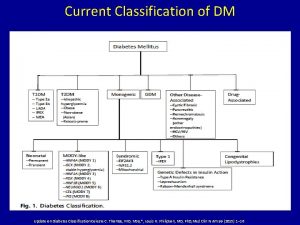
OLD CLASSIFICATION (1985) Type 1, Insulin-dependent (IDDM) Type 2, Non Insulin-dependent (NIDDM) obese non-obese MODY IGT Gestational Diabetes

© 2004, John Walsh, P. A. , C. D. E.

WHO CLASSIFICATION 2000 Is based on etiology not on type of treatment or age of the patient. Type 1 Diabetes ( autoimmune b-cell destruction) Type 2 Diabetes (defects in insulin secretion or action) Other specific types

Types of Diabetes in Children Type 1 diabetes mellitus accounts for >90% of cases. Type 2 diabetes is increasingly recognized in children with presentation like in adults. Permanent neonatal diabetes Transient neonatal diabetes Secondary diabetes e. g. in cystic fibrosis or Cushing syndrome.

– – – – EPIDEMIOLOGY Type 1 DM Most common metabolic disease in childhood Annual incidence 15 new cases per 100, 000 in children < 18 yrs Frequency increases with increasing age. 1: 1400 at age 5 yrs 1: 400 at age 16 yrs Males and females equally affected No correlation with socioeconomic status

Etiology Type 1 DM usually results from both an inherited risk and external triggers, Infection, diet. Type 1 diabetes is an autoimmune disorder in which the body attacks its pancreatic beta cells.

Genetic issues Clear evidence suggests a genetic component in type 1 diabetes mellitus. Monozygotic twins have a 60% lifetime concordance for developing type 1 diabetes mellitus, although only 30% do so within 10 years after the first twin is diagnosed. In contrast, dizygotic twins have only an 8% risk of concordance, which is similar to the risk among other siblings. The frequency of diabetes developing in children with a diabetic mother is 2 -3% and 56% if the father has type 1 diabetes mellitus. The risk to children rises to almost 30% if both parents are diabetic.

Type I – IDDM Two shared HLA haplotypes (DR 3 + DR 4) 12 -20% risk One shared HLA haplotype (DR 3 or DR 4) 5 -7% risk No shared HLA haplotypes 1 -2% risk HLA DQ(beta) Asp 57 virtual protection HLA DQ(beta) non-Asp 57 100 X increased risk

Environmental factors Viral infections (most important) probably by initiating or modifying an autoimmune process. ( Mumps, coxsackie virus, CMV, congenital rubella) Dietary factors are also relevant Breastfed infants have a lower risk for Type 1 DM. Some cow's milk proteins (e. g. , bovine serum albumin) have antigenic similarities to an islet cell antigen.

Diagnosis Symptoms of diabetes + casual plasma glucose concentration ≽ 11. 1 mmol/L (≽ 200 mg/dl). ¹ Casual is defined as at any time of the day without regard to time since the last meal. Or Fasting plasma glucose ≽ 7. 0 mmol/L (≽ 126 mg/dl). ² Fasting is defined as no caloric intake for at least 8 h. Or 2 h post load glucose ≽ 11. mmol/L (≽ 200 mg/dl) during an OGTT.
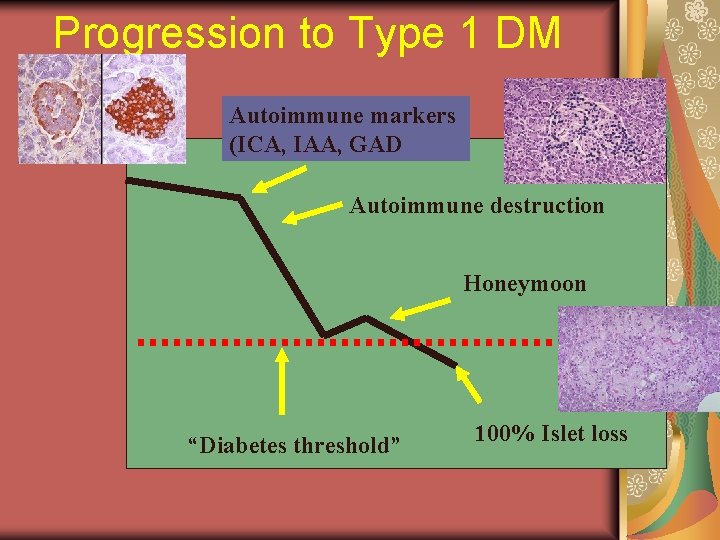
Progression to Type 1 DM Autoimmune markers (ICA, IAA, GAD Autoimmune destruction Honeymoon “Diabetes threshold” 100% Islet loss

Honeymoon Period In patients with new onset of DM 1 who do not have DKA, the beta cell mass has not been completely destroyed. The remaining functional beta cells seem to recover with insulin treatment, and they are again able to produce insulin. When this occurs, insulin requirements decrease, and there is a period of stable blood glucose control, often with nearly normal glucose concentrations. This phase of the disease, known as the honeymoon period, usually starts in the first weeks of therapy and usually continues for a few months at most, but can last 2 years

CLINICAL PRESENTATIONS Classical symptom triad: ü polyuria, loss polydipsia and weight DKA Accidental diagnosis

COMPLICATIONS OF DIABETES Acute: Ø DKA Ø Hypoglycemia Late-onset: q. Retinopathy q. Neuropathy q. Nephropathy q. Ischemic heart disease & stroke

DCCT And Other Studies Research studies between 1970 and 2000 showed that complications could be prevented by lowering high glucose levels Studies Results DCCT 1984 -1992 EDIC 1996 UKPDS 1978 -1998 Kumamoto 1992 -2000 Better health Fewer complications Improved sense of well-being More flexible lifestyle © 2004, John Walsh, P. A. , C. D. E.

TREATMENT ELEMENTS Education Insulin therapy Diet and meal planning Exercise Monitoring ü Hb. A 1 c every 2 -months ü Home regular BG monitoring ü Home urine ketones tests when indicated

EDUCATION Educate child & care givers about: q Diabetes q Insulin q Life-saving skills q Recognition of Hypo & DKA q Meal plan q Sick-day management

Management Principles of insulin therapy Varies between individuals and changes over time The correct dose of insulin is the dose that achieves the best glycemic control without causing obvious hypoglycemia problems and achieving normal growth(height and weight) Dosage depends on, Age, weight, stage of puberty, duration and phase of diabetes, state of injection sites, nutritional intake and distribution, exercise pattern, daily routine

Hyperglycemia: Microangiopathic complications Hypoglycemia: Neuronal loss Poor school performance seizures

TYPES OF INSULIN Short acting (neutral, soluble, regular) ü Peak 2 -3 hours & duration up to 8 hours Intermediate acting Isophane (peak 6 -8 h & duration 16 -24 h) ü Biphasic (peak 4 -6 h & duration 12 -20 h) ü Semilente (peak 5 -7 h & duration 12 -18 h) ü Long acting (lente, ultralente & PZI) ü Peak 8 -14 h & duration 20 -36 h

INSULIN ANALOGS Ultra short acting Insulin Lispro q Insulin Aspart q Long acting without peak action to simulate normal basal insulin q Glargine

FREQUENTLY USED REGIMES Twice daily regimes 2 daily injections of a mixture of a short or rapid insulin with an intermediate acting insulin (before meal and before main evening meal). Approximately 1/3 of the total insulin dose is short acting and 2/3 is intermediate. 2/3 of the total daily insulin is given in the morning and 1/3 in the evening. Three injections daily Before breakfast – mixture of rapid or short with intermediate Before afternoon snack or evening meal – rapid or short Before bed – intermediate acting

Insulin Dosage Prepubertal children usually require 0. 7 -1. 0 IU/kg/day During puberty, requirements may rise above 1 and even up to 2 IU/kg/day

INSULIN CONCENTRATIONS Insulin is available in different concentrations 40, 80 & 100 Unit/ml. WHO now recommends U 100 to be the only used insulin to prevent confusion. Special preparation for infusion pumps is soluble insulin 500 U/ml.

ADVERSE EFFECTS OF INSULIN Hypoglycemia Lipoatrophy Lipohypertrophy Obesity Insulin allergy Insulin antibodies Insulin induced edema

PRACTICAL PROBLEMS injection sites & technique Insulin storage & transfer Mixing insulin preparations Insulin & school hours Adjusting insulin dose at home Sick-day management Recognition & Rx of hypo at home

TREATMENT MADE EASY Insulin pens & new delivery products Handy insulin pumps fine micro needles Simple accurate glucometers Free educational material computer programs for comprehensive management & monitoring

Diet On the standard twice daily regime, food intake is divided into 3 main meals with snacks between meals and before going to bed. Diet should be high in fibre which will provide a sustained release of carbs.

Type of Food The following are among the most recent consensus recommendations: Carbohydrates should provide 50 -55% of daily energy intake. (No more than 10% of carbohydrates should be from sucrose or other refined carbohydrates. ) Fat should provide 30 -35% of daily energy intake. Protein should provide 10 -15% of daily energy intake.

Activity Type 1 diabetes mellitus requires no restrictions on activity; exercise has real benefits for a child with diabetes. Most children can adjust their insulin dosage and diet to cope with all forms of exercise. The current guidelines are increasingly sophisticated and allow children to compete at the highest levels in sport.

Dreams are the seedlings of realities

FUTURE PROMISES The cure for IDDM is successful islet cell transplantation, which will be available in the near future. Primary prevention by a vaccine or drug will be offered to at risk subjects identified by genetic studies. Gene modulation therapy for susceptible subjects is a promising preventive measure.

DIABETES MELLITUS - TYPE 1 MONITORING STRATEGIES Self Blood Glucose Monitoring – 2 to 4 / day Urine Testing – Ketones Glycosylated Hemoglobin - Hb. A 1 C - quarterly Blood lipids - annually Thyroid function – annually Urine micro albumin – quarterly after 5 yr Dilated fundoscopy – age 10 yr + 3 -5 yr

Diabetic ketoacidosis Ketoacidosis is a state of uncontrolled catabolism associated with insulin deficiency Diabetic ketoacidosis tends to occur in individuals younger than 19 years, but it may occur in patients with diabetes at any age. Criteria Hyperglycemia: blood glucose >11 mmol/L(200 mg/d. L) Venous p. H<7. 3 or bicarbonate <15 mmol/L Ketonemia and ketonuria

Diabetic ketoacidosis In the absence of insulin, hepatic glucose production accelerates, and peripheral uptake by tissues such as muscle is reduced. Rising glucose levels lead to an osmotic diuresis, loss of fluid and electrolytes, and dehydration. Plasma osmolality rises and renal perfusion falls. In parallel, rapid lipolysis occurs, leading to elevated circulating free fatty-acid levels. The free fatty acids are broken down to fatty acyl-Co. A within the liver cells, and this in turn is converted to ketonebodies within the mitochondria

Diabetic ketoacidosis Accumulation of ketone bodies produces a metabolic acidosis. Vomiting leads to further loss of fluid and electrolytes. The excess ketones are excreted in the urine but also appear in the breath, producing a distinctive smell similar to that of acetone. Respiratory compensation for the acidosis leads to hyperventilation, graphically described as 'air hunger'. Progressive dehydration impairs renal excretion of hydrogen ions and ketones, aggravating the acidosis

Diabetic ketoacidosis Mostly the causes are Previous undiagnosed diabetes Interruption of insulin therapy The stress of intercurrent illness Goals of therapy Correct dehydration Correct acidosis and reverse ketosis Restore blood glucose to near normal Avoid complications of therapy Identify and treat precipitating event

History Classic symptoms of hyperglycemia Thirst Polyuria, polydipsia Nocturia Other symptoms Generalized weakness Malaise/lethargy Nausea/vomiting Decreased perspiration Fatigue Anorexia or increased appetite Confusion Symptoms of associated infections and conditions Fever Dysuria Chills Chest pain Abdominal pain Shortness of breath

Physical General signs Ill appearance Dry skin Labored respirations Dry mucous membranes Decreased skin turgor Decreased reflexes Vital signs Tachycardia Hypotension Tachypnea Hypothermia Fever, if infection Specific signs Ketotic breath (fruity, with acetone smell) Confusion Coma Abdominal tenderness

Management 1. Fluids If in shock, initial resuscitation with normal saline. Dehydration should then be corrected gradually over 48 to 72 hour using 0. 45% Saline Monitor : - - - Fluid input and output Electrolytes, creatinine and acid-base status regularly Neurological states 2. Insulin - insulin infusion is started, titrating the dose according to the blood glucose. Monitor blood glucose regularly. aim for gradual reduction of blood glucose.

3. Potassium Although the initial plasma concentration may be high, it will fall following treatment with insulin and rehydration High doses (20 to 40 m. Eq/L) to be added to drips 4. Sodobicarb(used only in selected patients) -severe acidemia(arterial p. H<6. 9)in whom decreased cardiac contractility and peripheral vasodilation can further impair tissue function - life threatening hyperkalemia - cautiously give 1 -2 mmol/kg over 60 min
COMPLICATIONS OF THERAPY Main complication is BRAIN EDEMA © 2004, John Walsh, P. A. , C. D. E.

TYPE 2 DM IN CHILDREN Among 10 – 19 yr olds T 2 DM accounts for 33% of all new cases African Americans – 42% Caucasians – 10%
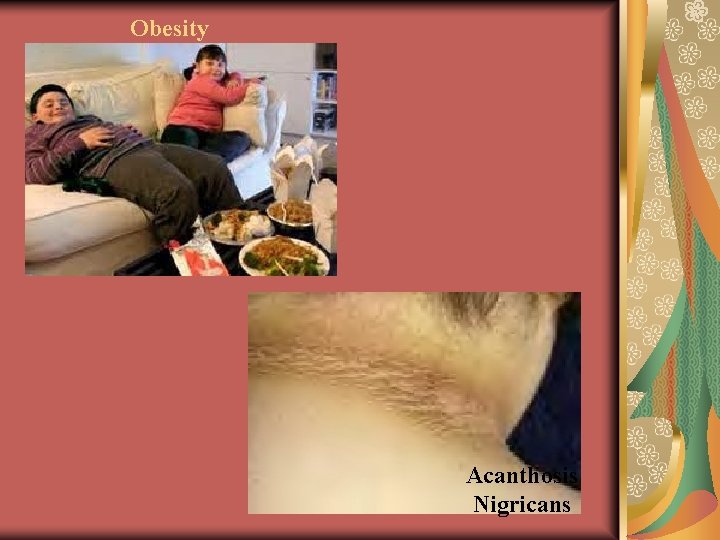
Obesity Acanthosis Nigricans
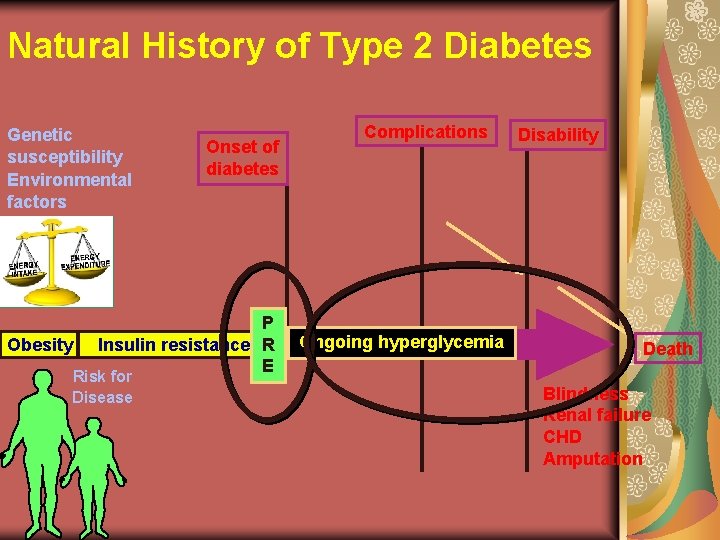
Natural History of Type 2 Diabetes Genetic susceptibility Environmental factors Onset of diabetes Complications Disability P Obesity Insulin resistance R Ongoing hyperglycemia Death E Risk for Metabolic Retinopathy Blindness Disease Atherosclerosis Syndrome Hyperglycemia Nephropathy Renal failure Neuropathy CHD Hypertension Amputation

Therapy of T 2 DM in Children Reduce calories – weight loss NOT T 1 DM diet with high complex CHO No Between meal snacks NOT T 1 DM where hypoglycemia is frequent Reduce CHO intake Reduce fat intake Exercise – increase healthy life style Drug treatment early x
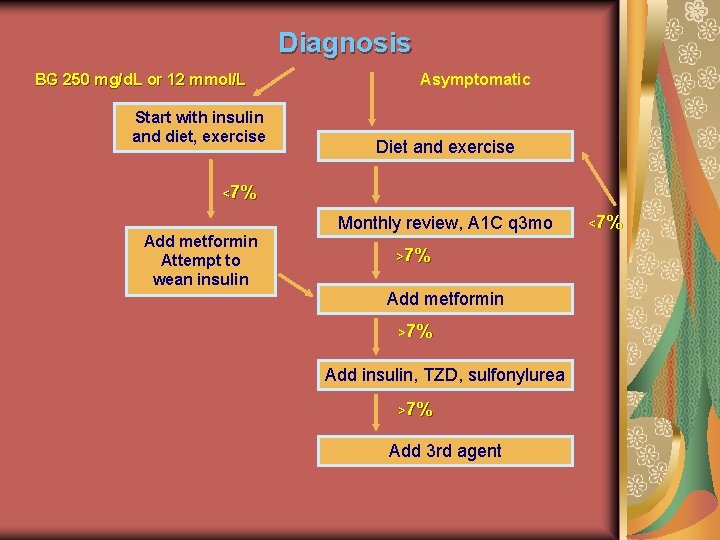
Diagnosis BG 250 mg/d. L or 12 mmol/L Start with insulin and diet, exercise Asymptomatic Diet and exercise <7% Add metformin Attempt to wean insulin Monthly review, A 1 C q 3 mo >7% Add metformin >7% Add insulin, TZD, sulfonylurea >7% Add 3 rd agent TZD = thiazolidinedione Silverstein JH, Rosenbloom AL. J Pediatr Endcrinol Metab. 2000; 13 Suppl 6: 1406 -1409. <7%

T 2 DM vs T 1 DM T 2 DM T 1 DM Obesity 95% > 85%tile Not Common + Family Hx T 2 72 – 85% Not Common Acanthosis Nigricans Maternal Gestational DM 60 – 86% + 7% all school aged children Not Common IUGR + Not common

T 2 DM vs. T 1 DM T 2 - DM T 1 – DM Polyuria/dypsia + + Ketonuria +/- DKA +/- Autoimmunity +/- + Initial insulin Often required Required Honeymoon + + Worse @ illness + +
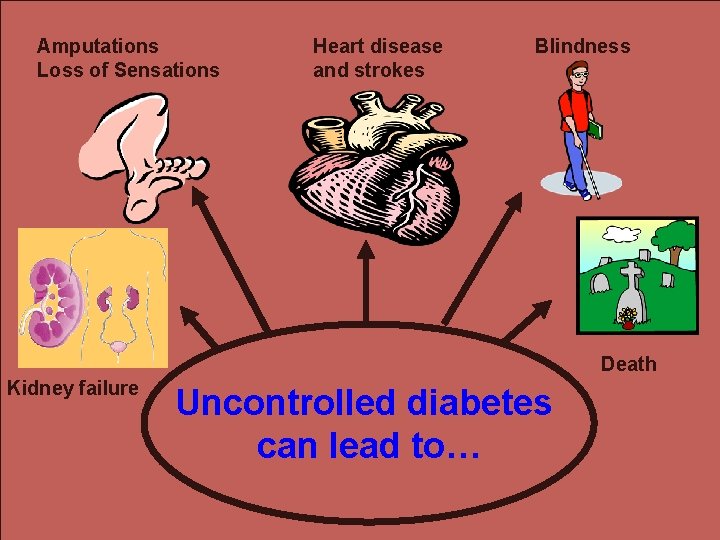
Amputations Loss of Sensations Heart disease and strokes Blindness Death Kidney failure Uncontrolled diabetes can lead to…

By learning you will teach; By teaching you will learn. Latin Proverb © 2004, John Walsh, P. A. , C. D. E.

Research tells us 14 out of any 10 individuals likes Candy Thanks Amigos
- Slides: 56